01 - introduction
1/38
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
39 Terms
What are the 3 categories of primary materials? What is the 4th one?
Metals
Ceramics
Polymers
Composites
What is special about composites?
They're formed by combining 2 or more of the 3 primary materials (metals, ceramics, polymers)
What kind of material is located between polymers and ceramics?
Silicones
What kinds of materials are located between metals and ceramics?
Semi-conductors
What kind of material is located between metals and polymers?
Conducting polymers
How many properties the polymers have?
12
Why are the 4 physical & thermal properties of polymers?
Low density : little mass, for BIG SIZE
Low thermal conductivity : don't conduct heat well
High thermal expansion : expand/shrink significantly under temperature changes
Temperature dependence : very sensitive to heat (soften, melt, degrade...)
What are the 4 electrical & chemical properties of polymers?
Good electrical insulation capacity : don't allow electricity to flow through them easily
High resistance to diffusion : don't allow fluids to seep (s'infiltrer) through them easily
Good chemical resistance : mostly don't react with acids, bases, salts
Flammability : catch fire easily
What are the 3 mechanical properties of polymers?
Low Young's modulus (elastic) : flexible, can bent easily
Low tensile strength : break under less pulling force than other materials
Good malleability : easy to shape and mold
What is the aesthetic & manufacturing property of polymers?
Dyeable : can easily absorb pigments
What are the 4 sectors using the most plastics?
Packaging
Construction
Vehicles
Electronics
What can we say about the oil consumption for the plastic production?
Plastic production requires less total oil than other materials
How has the plastics production been evolving since the end of the 20th century worldwide? (1975)
It has been multiplied by 8 from 1975 to 2023 (50 millions tons to 400 millions tons per year)
Which continent produces most plastics? Which country? What proportion?
Asia : >50%
China : >30%
How has the plastics production been evolving these past few years in Europe?
It has decreased
Give the definition of plastics
Plastics are high-molecular organic compounds (very large molecules, mostly of carbon)
formed either by modifying high-molecular natural products (like rubber from trees, cellulose...)
OR by chemical deposits of low-molecular basic building blocks (monomers) through chemical reactions
What is a monomer? What are their properties?
Small, simple molecules
Can bond together to form larger structures
Building blocks of polymer
Low molecular weight
Can exist independently OR as a part of a polymer chain
Often gases/liquids at room temperature
What are polymers? What are their properties?
Large, complex molecules
Made up of repeating units (monomers) linked together by chemical bonds
Basis of plastics
High molecular weight
Solids/liquids/gels, depending on their structure
Their properties depend on the type of monomer AND how they're linked together
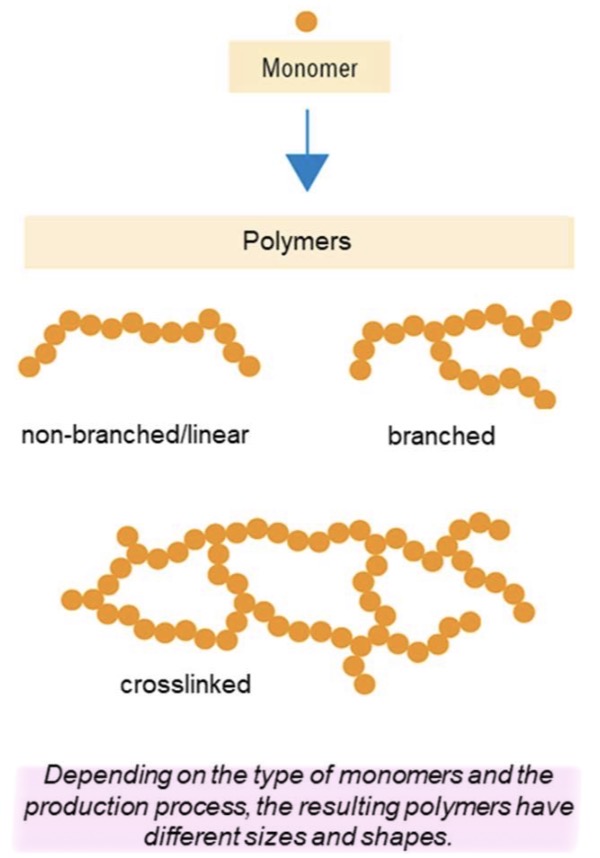
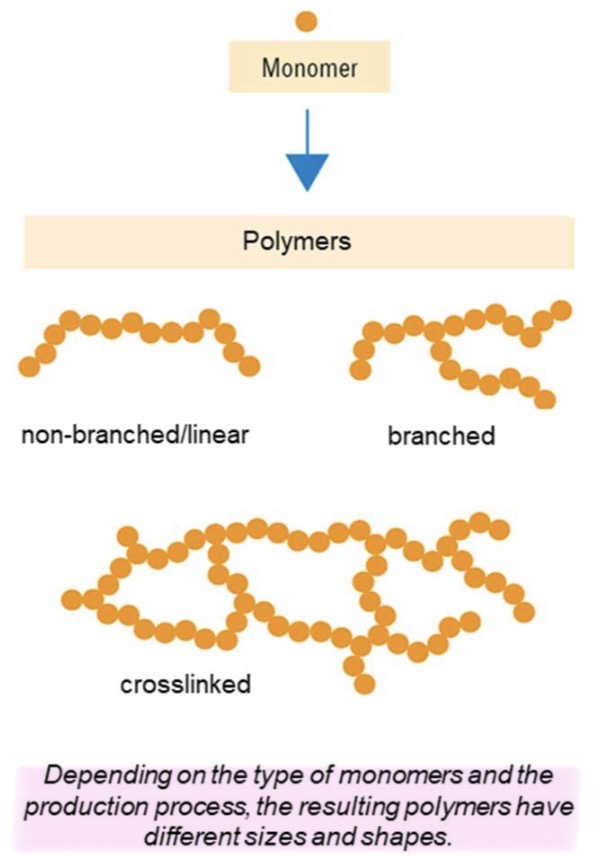
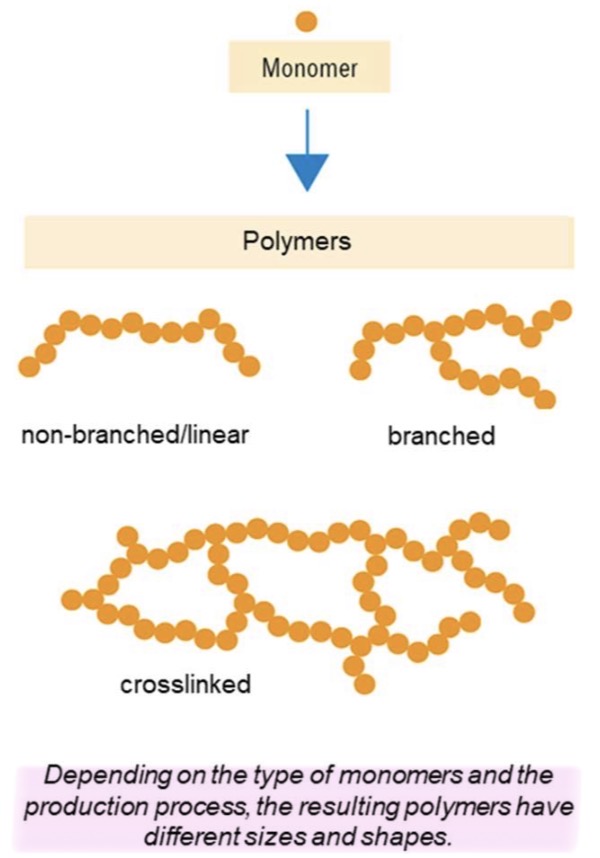
What are the 3 types of polymers chains? What are their differences?
Non-branched/ linear : no branches or connections between chains
Branched : linear chains with side branches attached to the main chain
Crosslinked : chains are interconnected by chemical bonds
What are the 3 different types of chemical bonds? Between what kind of material do they occur?
Metallic bonds : metal/metal
Atomic bonds : non-metal/non-metal
Ionic bonds : metal/non-metal
What kinds of bonds occur in plastics? (4)
Chemical bonds : within the polymer chain, between monomers (high bonding force, require a lot of energy to break)
Cross linking : chemical bonds between different polymer chains
Interactions between the molecules : secondary interactions (van der Waals forces, hydrogen bonds, dipole/dipole interactions...) between different polymer chains
Mechanical bonds : polymer chains are physically entangled with each other (like a spaghetti bowl)
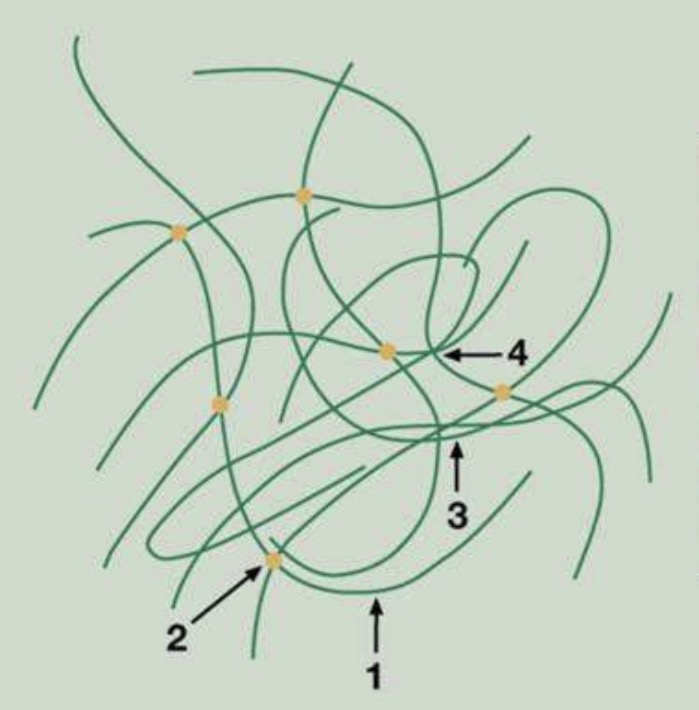
What is the type of high-bonding energy bonds? Low-bonding energy? Give examples for each category
High-bonding energy : chemical bonds (covalent, metallic, ionic)
Low-bonding energy : intermolecular interactions (Van der Waals forces, hydrogen bonds)
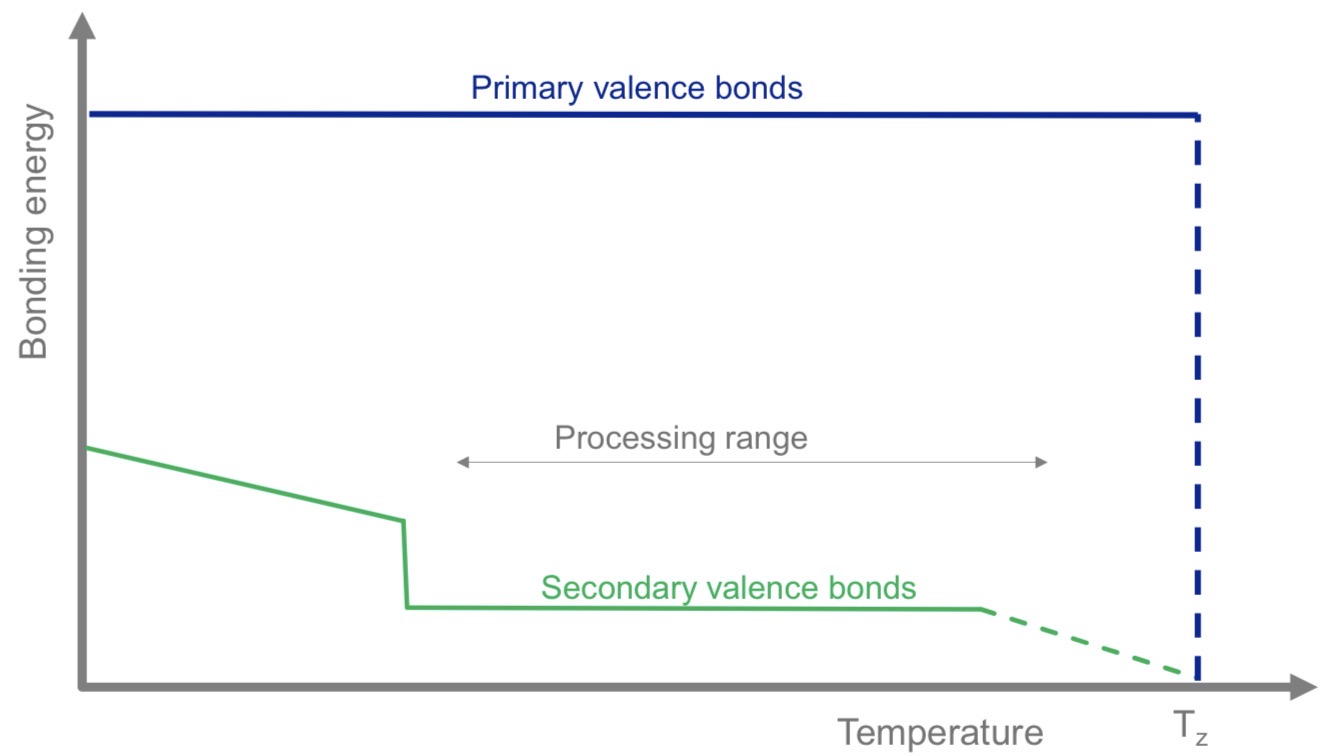
What's the influence of the temperature on the bonding energy of primary and secondary valence bonds?
Primary valence bonds : highly stable, do not break with temperature changes (until the decomposition temperature is reached)
Secondary valence bonds : weaken as temperature increases until it reaches the decomposition temperature
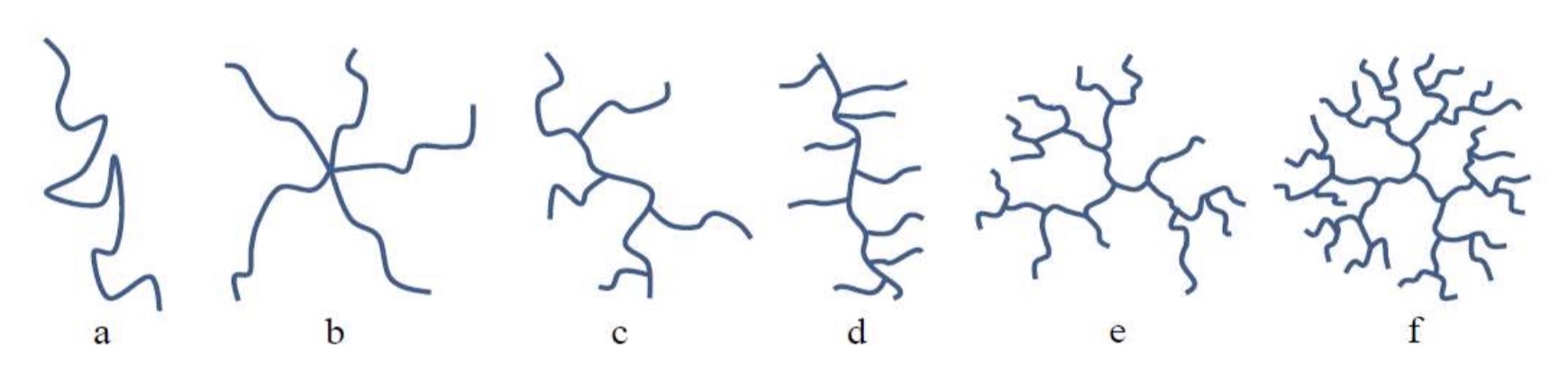
Give 6 names of polymers topologies
Linear
Star polymer
Chain branched
Comb polymer
Hyper branched
Dendrimer
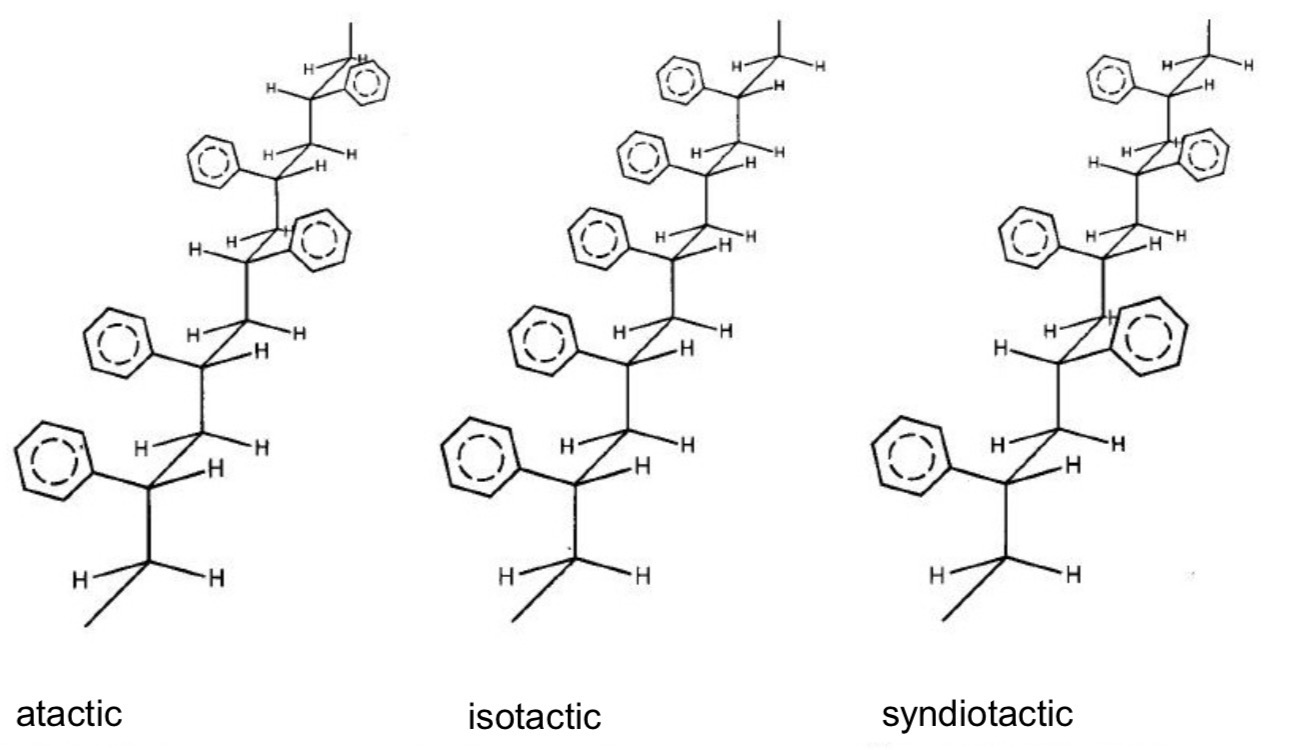
What’s the tacticity in macromolecules?
Give the 3 types of tacticity
Spatial arrangement of components/substituents along the polymer chain (sequence)
Atactic : random distribution (generally amorphous)
Isotactic : all substituents are located on the same side of the polymer chain (crystalline structure)
Syndiotactic : the substituents alternate regularly from one side to the other (crystalline structure)
What are the major steps and what do they use to product plastic?
Crude oil —> DISTILLATION —> naphta —> CRACKING —> monomers —> POLYMERISATION —> plastics
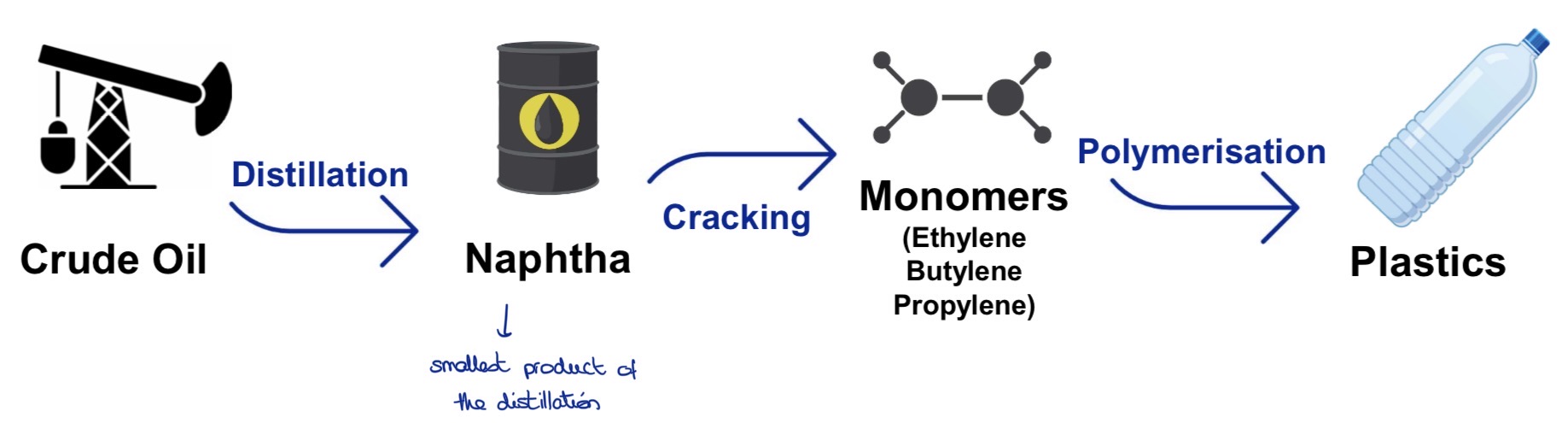
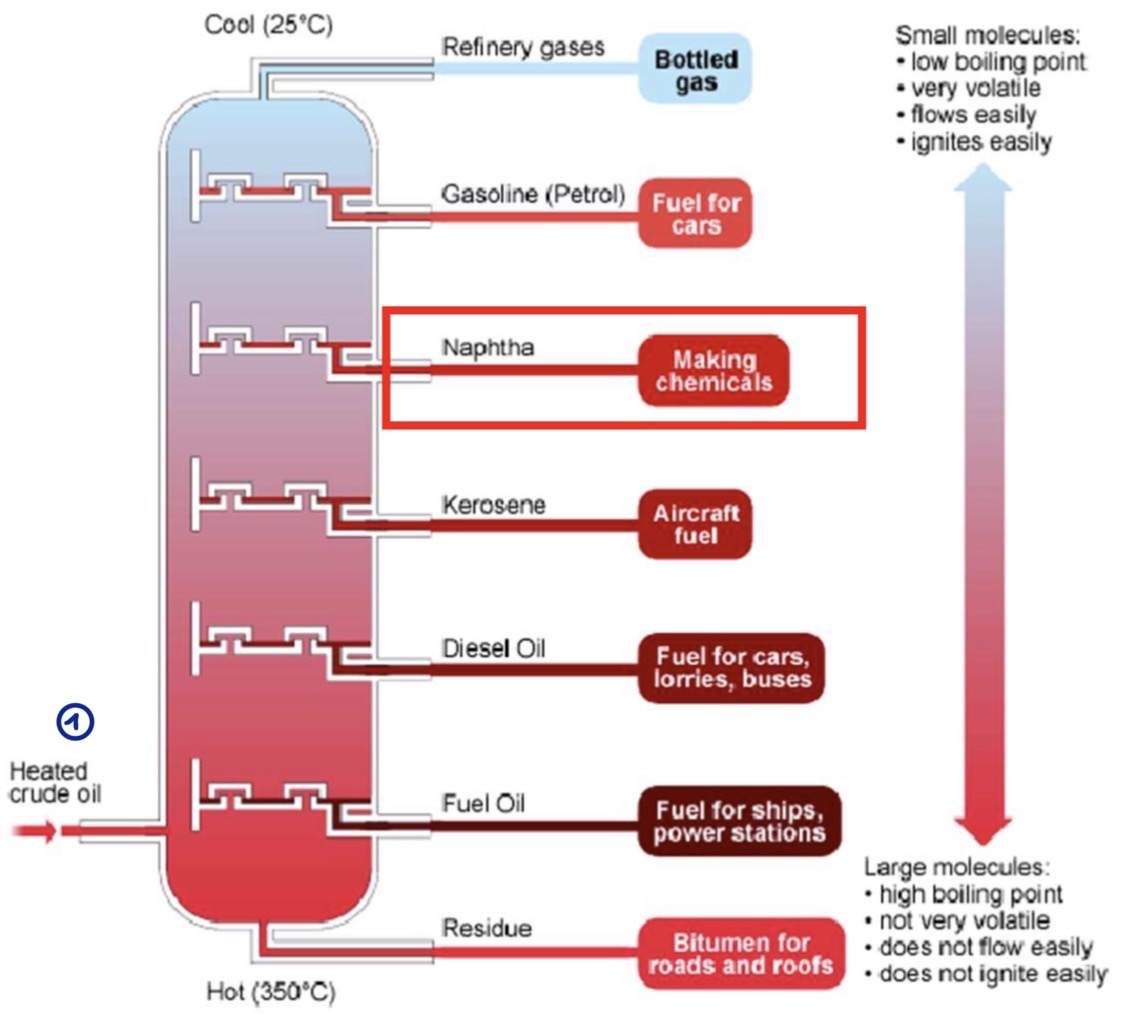
What happens during the distillation?
The crude oil is heated up to 350°
It’s turned into vapor
The vapor rises through the distillation column and cools down until it becomes naphta
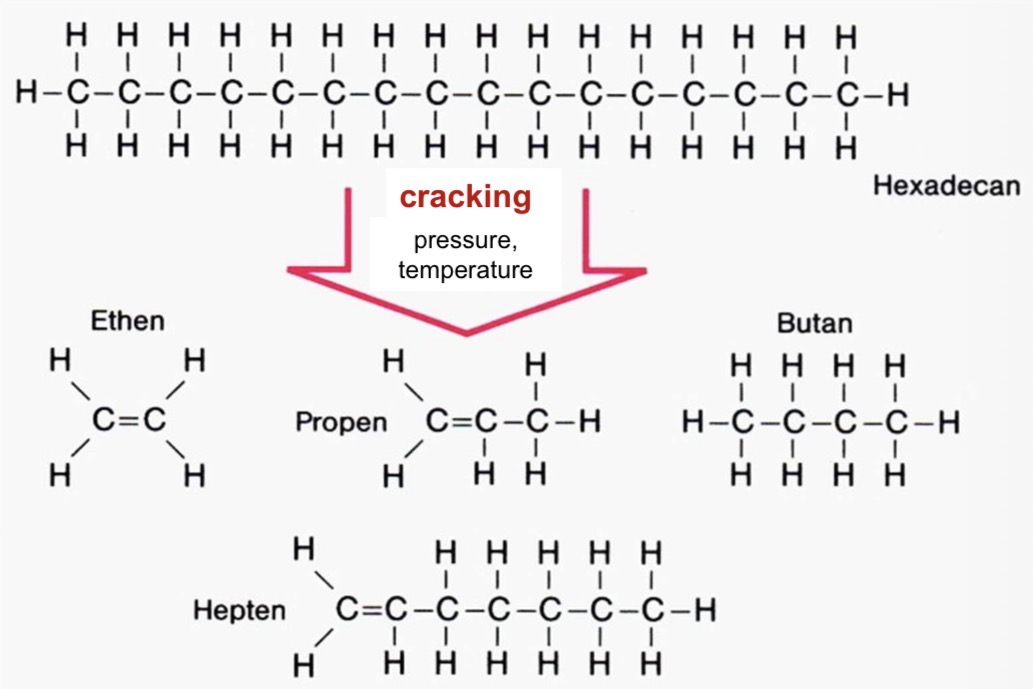
What’s the cracking step?
What happens during this?
Process used to break down large hydrocarbon molecules into smaller (more useful) molecules
large hydrocarbon molecules (naphta from the distillation) are under high pressure and temperature to break the carbon-carbon bonds
In the case of plastic production, we get monomers out of the cracking
What’s the polymerisation? (In general)
What are the 3 different types of polymerisation?
Process (chemical reaction) by which monomers react together to form polymer chains
polymerisation
Polycondensation
Polyaddition
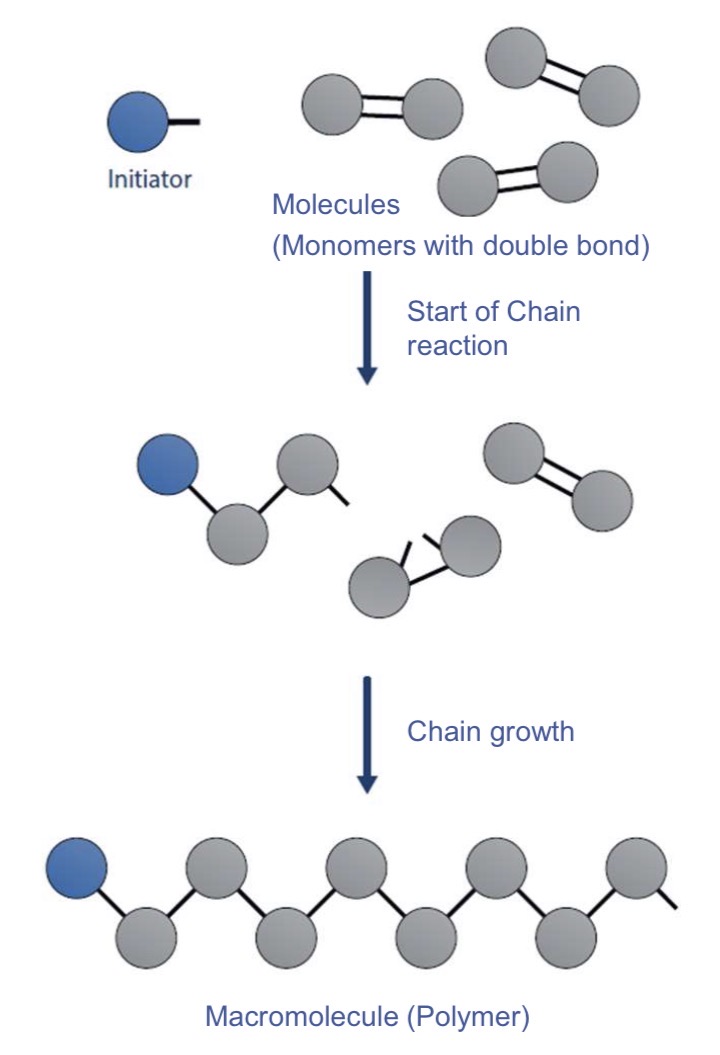
What’s the polymerisation?
What are the key features of this mechanism?
Chain growth reaction where monomers (small molecules) add to a growing polymer chain at one time, rapidly forming long chains
no by-products
Rapid chain growth
Homopolymerisation or copolymerisation are possible
Describe the process of polymerisation more in details
Activation : a initiator (radical, ion, catalyst) breaks the double bond of a monomer of which the center becomes active (=active center : radical, cation, anion)
Growth reaction : This monomer attacks another monomer by breaking its double bond and by binding with it forming a single bond, and leaving a free single bond
Termination : This process repeats itself until a terminator is encountered or until 2 growing chains combine (no active centres anymore, neutral centres)
What are the 4 different types of polymerisation? And how do they work?
radical polymerisation
—> uses chemical radicals to break double bonds
Thermal polymerisation
—> increasing the temp until radicals are formed
Ionic polymerisation
—> uses cations or anions as active centers
Coordination polymerisation
—> uses catalysts to control tacticity and monomer activation
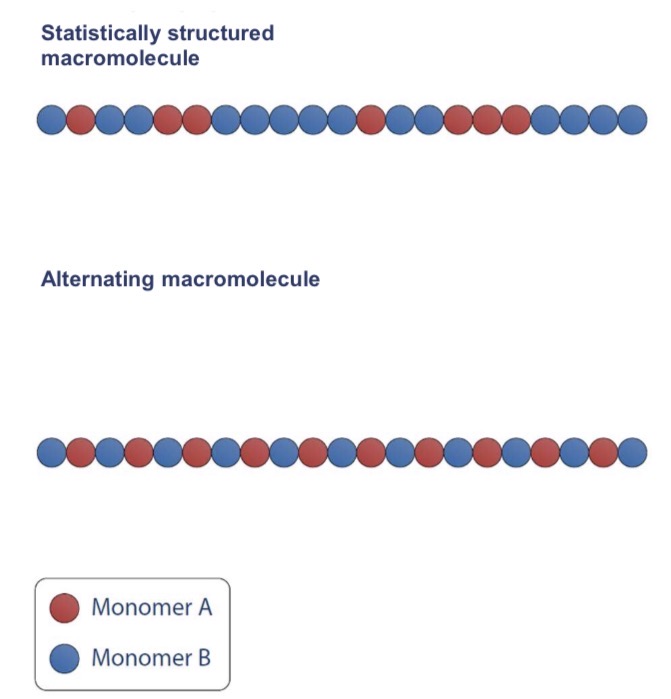
What’s the homopolymerisation? Copolymerisation?
Homo- : one type of monomer
Co- : multiple types of monomers (cf photo)
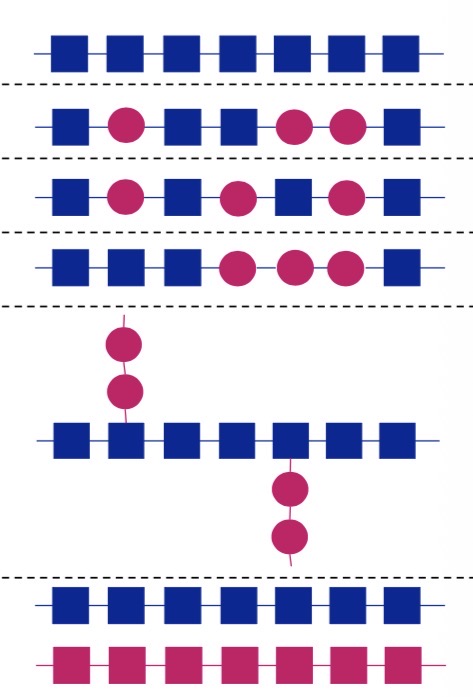
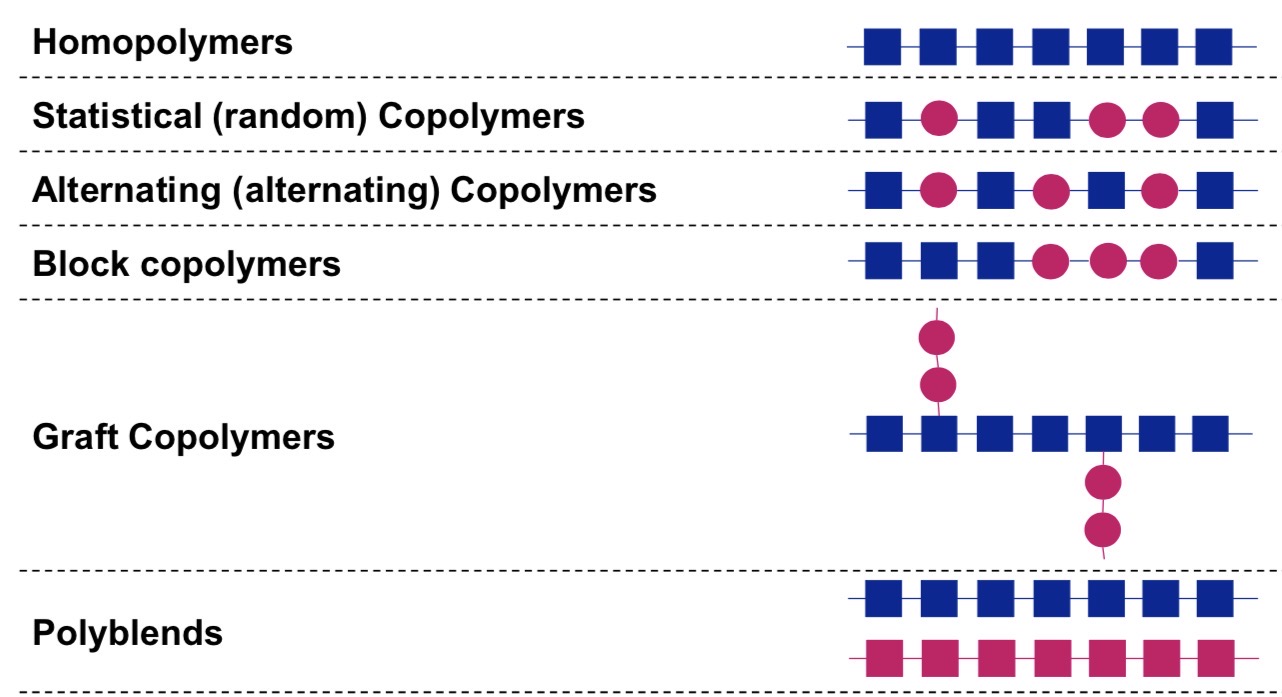
Name these different polymers structures
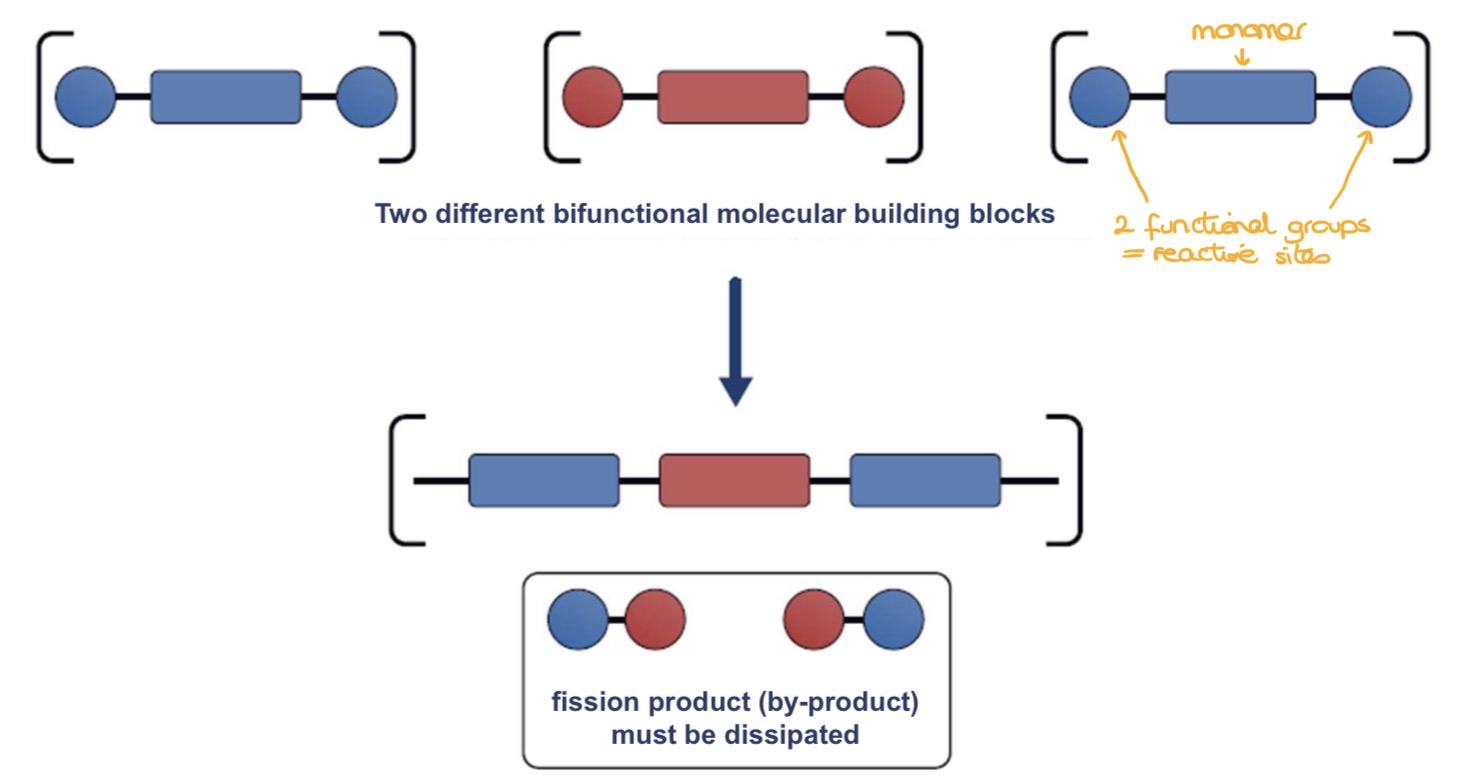
What’s the polycondensation?
What are the key features of this mechanism?
Step growth reaction where monomers with functional groups react to form polymers, releasing small molecules as by-products
starts with a catalyst (acid or base)
By-products (water, hydrochloric acid, alcohols, ammonia)
Slower reaction compared to polymerisation
Polyamides, polyesters, phenoplastics, aminoplastics, silicones
What are the 3 types of polycondensation? How do they work?
melt polycondensation
—> solvent-free reaction
Solvent polycondensation
—> the reaction takes place in a homogeneous solution (solvent)
Interfacial polycondensation
—> the reaction takes place at the interface of 2 immiscible liquids
Describe the process of polycondensation more in details
Monomers have 2 functional groups (reactive sites)
Functional groups react, forming covalent bonds and releasing by-products (formed from parts of the functional groups)
Chains grow slowly as monomers react step-by-step
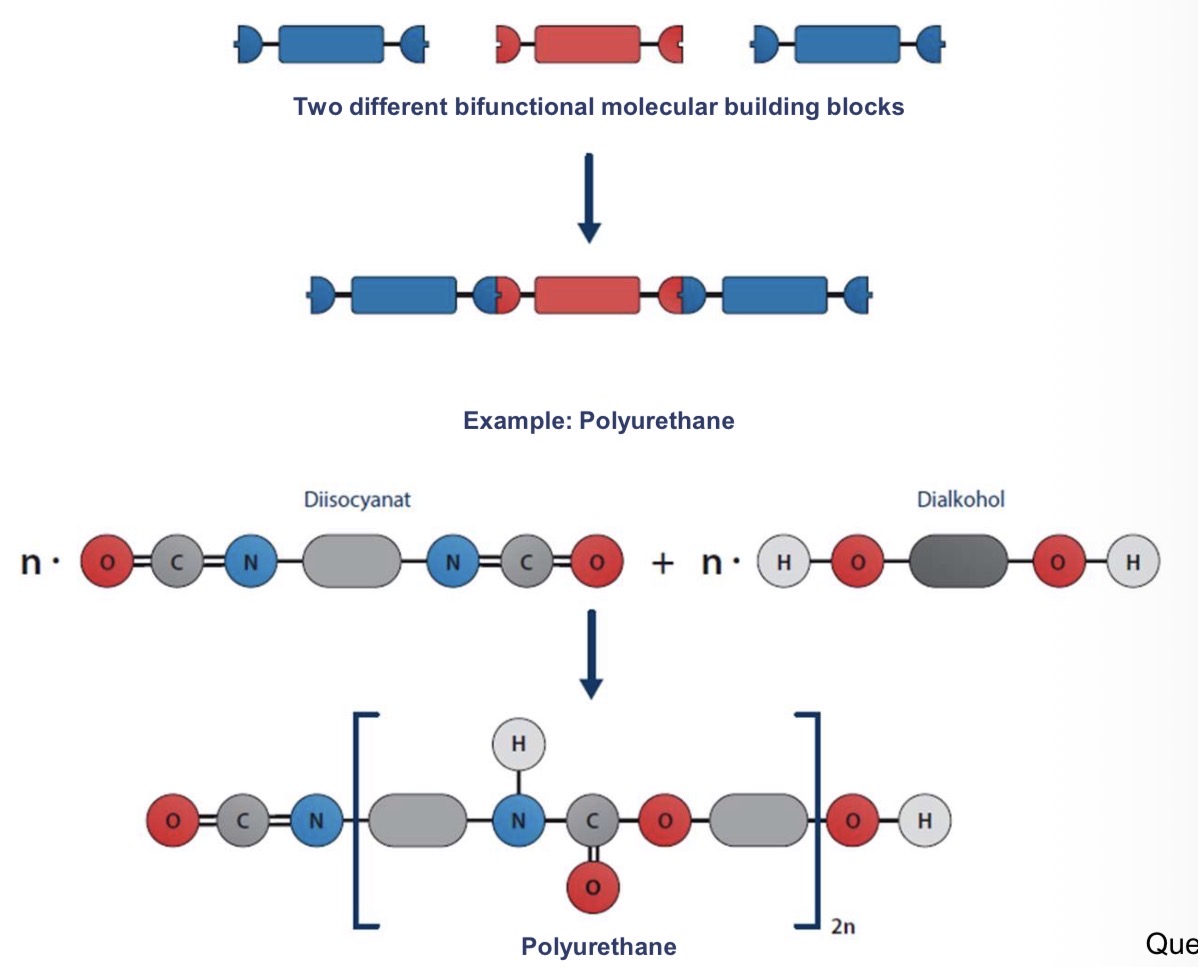
What’s the polyaddition?
What are the key features of this mechanism?
Step growth reaction where monomers add to each other without releasing by-products, forming polymers
no by-products
Catalysts required to initiate the process
Shift of hydrogen atoms from one functional group to another (facilitates the covalent bonds formation)
Polyurethanes, polyueras, epoxy resins
Describe the process of polyaddition more in details
monomers have functional groups (reactive sites) that can directly react without the need to release any part of them (by-products)
Monomers add directly to each other, forming covalent bonds without by-products
Chains grow slowly, similar to polycondensation