Module 2 Organic Chemistry Portage
1/119
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
120 Terms
Hydrocarbons
molecules composed only of carbon and hydrogen
Saturated hydrocarbons
Alkanes are considered ______, because they only have single C-C bonds
Linear, Branched, or Cyclic
what are the possible carbon skeleton shapes of alkanes?
Cycloalkanes
alkanes with cyclic carbon structures
Methane
CH4
CnH2n
What is the molecular formula for Alkenes
C2H6
Ethane
C3H8
Propane
C4H10
Butane
C5H12
Pentane
C6H14
Hexane
C7H16
Heptane
C8H18
Octane
C9H20
Nonane
C10H22
Decane
the larger molecular weight, the more constitutional isomers possible
How does molecular weight affect the number of constitutional isomers?
-ane
what is the suffix indicating a saturated hydrocarbon (alkane) ?
all acrylic saturated alkanes
The suffix -ane is used for _________
by combining the prefix and the suffix
how are linear (continuous) alkanes named in accordance with IUPAC?
Longest continuous chain of carbon in the skeleton
how is the parent name of the molecular in a branched alkane determined?
substituent
any group attached to the parent chain
alkyl groups
saturated hydrocarbon substituents are known as _______
-yl
The name of an alkyl substituent is determined by using a prefix corresponding to the number of carbons present and changing the -ane suffix to “______”
start at the position to the closest substituent
in a basic sense, how do you know where to start numbering and placing locusts?
Locant-(prefix)substituentparent
order for systematic names
they are listed alphabetically in the final name of the compound
what do you do if there are two or more different substituents on a parent chain?
-o
Halogen atoms as substituents are named by changing the -ine ending of the halogen element name to ____
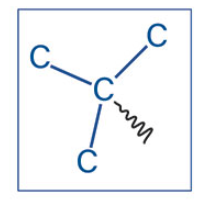
Tert-Butyl or t-Butyl
What is this akyl group?
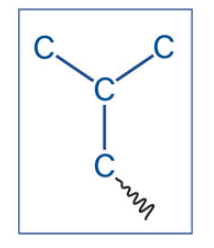
Isobutyl
What is the name of this akyl group?
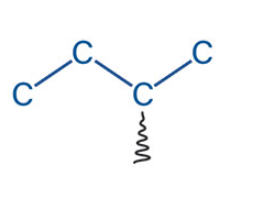
Sec-Butyl
What is the name of this akyl group?
butyl or n-Butyl
What is the name of this akyl group?
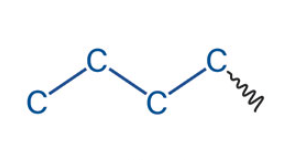
isopropyl
what is the name of this akyl group?
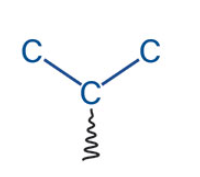
Propyl or n-Propyl
What is the name of this akyl group
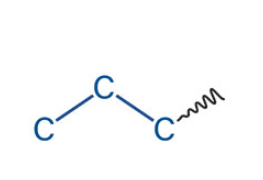
Water-insoluble
when in reference to Alkanes solubility in water, they are ________ _______
hydrogen bonding
when hydrogen is electronically attracted to another atom, like in water, this is called
increases
as molecular weight increases, MP and BP ________
Van Der Waals Forces (VDWFs)
the attractive forces between non-polar molecules
nonbonding intermolecular forces
attractive forces between molecules that are not a true bond in sense of atom sharing or donating electrons to each other.
Conformations
Alkanes can adopt ______ which are different orientations of atoms or groups of atoms
Conformers
different shapes of molecules derived by rotations of single bonds are referred to as _______ of one another
Newman Projections
shows the confirmation of molecules when looking down a particular bond axis. Allow us to visualize the relationships between atoms.
CnH2n
Formula for cycloalkanes with one ring
Cyclopropane
What is the name of this cycloalkane?

Cyclobutane
What is the name of this cycloalkane?

Cyclopentane
What is the name of this cycloalkane?
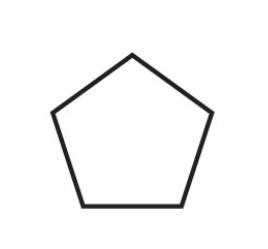
Cyclohexane
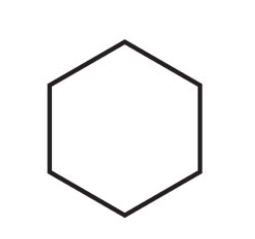
What is the name of this cycloalkane?
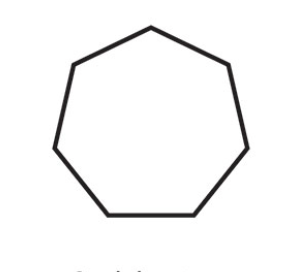
Cycloheptane
What is the name of this cycloalkane?
begin with the highest alphabetical priority substituent
how do you begin to number the substituents on a cycloalkane
angle strain
deviation from ideal bond angles in cycloalkanes
torsional strain
eclipsing interactions in cycloalkanes that contribute to the instability of the molecule
ring strain
the total amount of strain energy in a cycloalkane
chair conformation
the most stable conformation that cyclohexane can adopt
non-planar conformation
slight rotation around the cycloalkane C-C bonds, include puckered or enveloped
Stereoisomers
different compounds that have the same connectivity between atoms but differ in positioning of certain atoms or substituent groups in a 3D space.
Exothermic
because the combustion reaction produces heat as a product, the reactions are ______
Combustion reaction
what type of reaction is this?
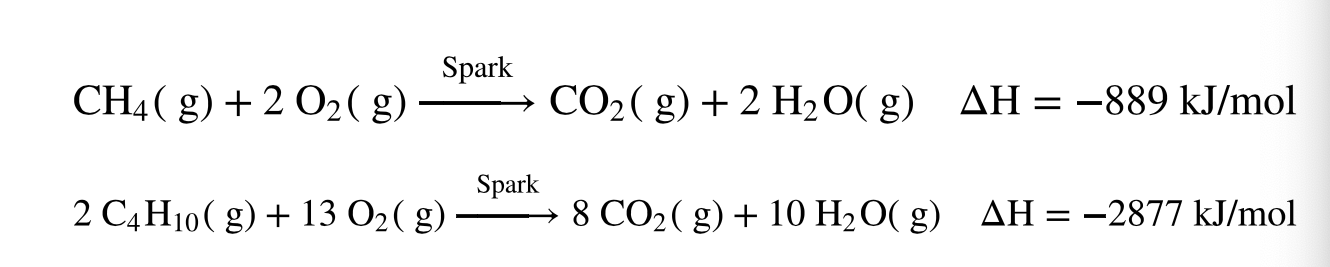
Carbon Monoxide
If a fossil fuel is combusted in a partial oxygen atmosphere, incomplete oxidation of the carbon atoms produces ________
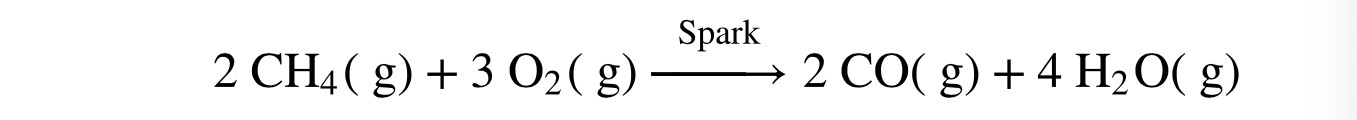
Radical Halogenation (substitution)
halogen atoms (typically Br or Cl) are substituted and take the place of hydrogen atoms on an alkane molecule
Alkyl Halide (Haloalkane)
the product of radical halogenation of an alkane is an __________
Radical Halogenation (Substitution)
what kind of reaction is this?
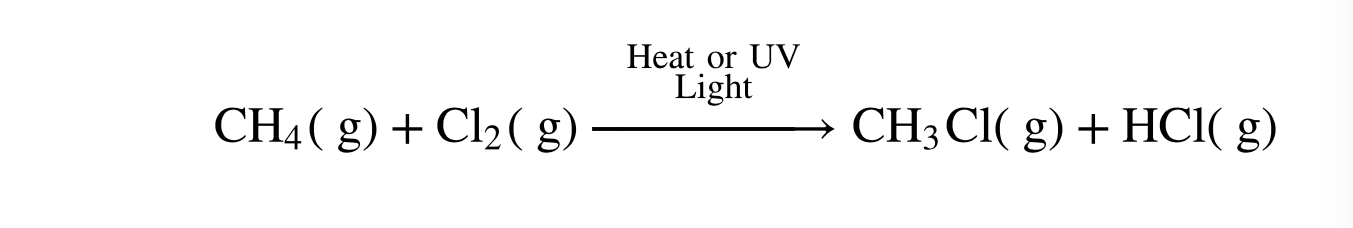
Halogen Radicals
species with a single unpaired electrons that attack the alkane substrate to pull off hydrogen atoms and substitute themselves in their place.
polyhalogenated products
If the halogen is in stoichiometric excess to the alkane substrate, the reaction can continue further to give _________ products, where more than one hydrogen atom is replaced by a halogen.
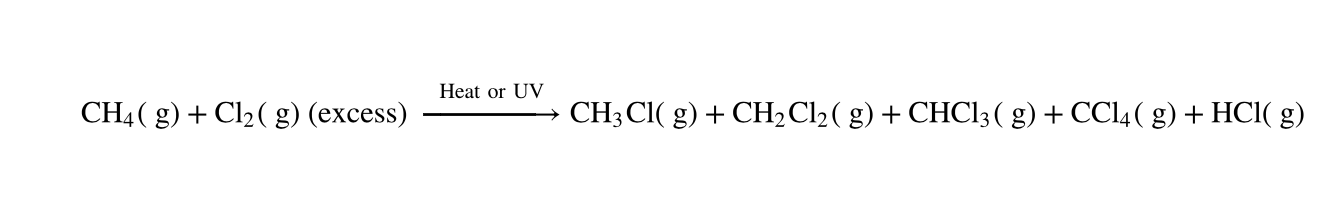
Alkenes
Hydrocarbons molecules that contain one or more C=C bonds in their structure.
CnH2n
formula for an alkene containing a single C=C bond
alkynes
hydrocarbon molecules that contain one or more C≡C bonds in their structure
CnH2n-2.
what is the formula for alkynes that have a single C≡C
olefins
alkenes are sometimes referred to as ______
Acetylenes
alkynes are sometimes referred to as _______
Unsaturated
in reference to their carbon to hydrogen ration, alkenes and alkynes are considered _______ _______
polyenes
compounds with more than one C=C bonds
polyynes
compound with more than one C≡C bond (more than one triple bond)
-ene
suffix used in alkene naming
-yne
suffix used in alkyne naming
by using di, tri, tetra, etc in addition to the suffix. to indicate the number of bonds
when more than one double or triple bond is present, how is this indicated?
Ethylene
what is the common name for this alkene (1-ethene)
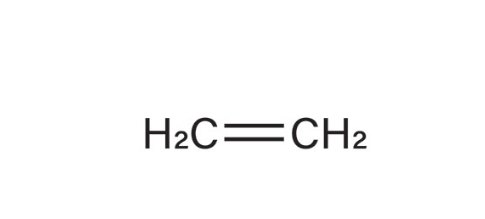
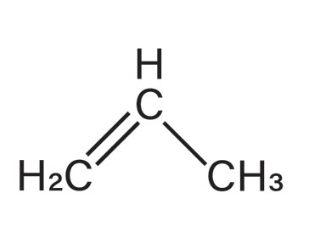
Propylene
What is the name of this alkene (1-propene)
Acetylene
What is the name of this alkyne (1-ethyne)
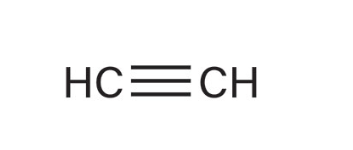
trigonal planar
the geometry around each carbon atom in a double bond is __________
addition reaction
the most common type of reaction for alkenes and alkynes
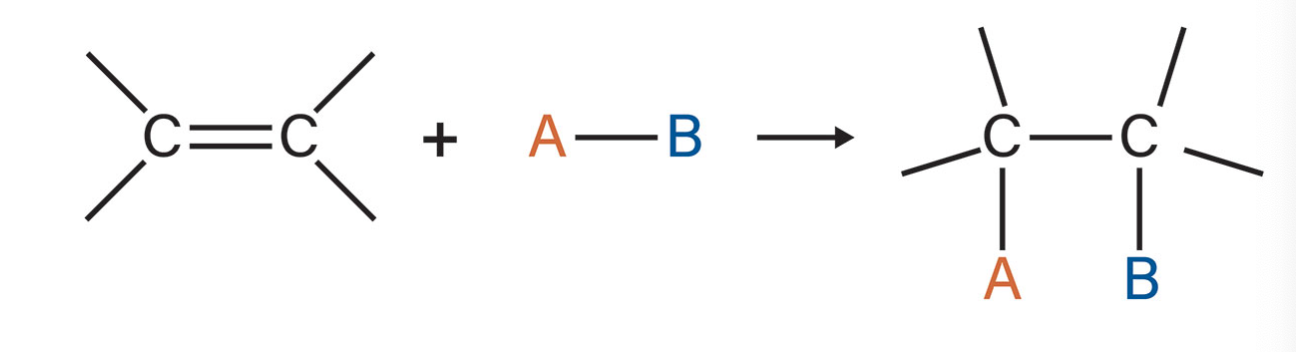
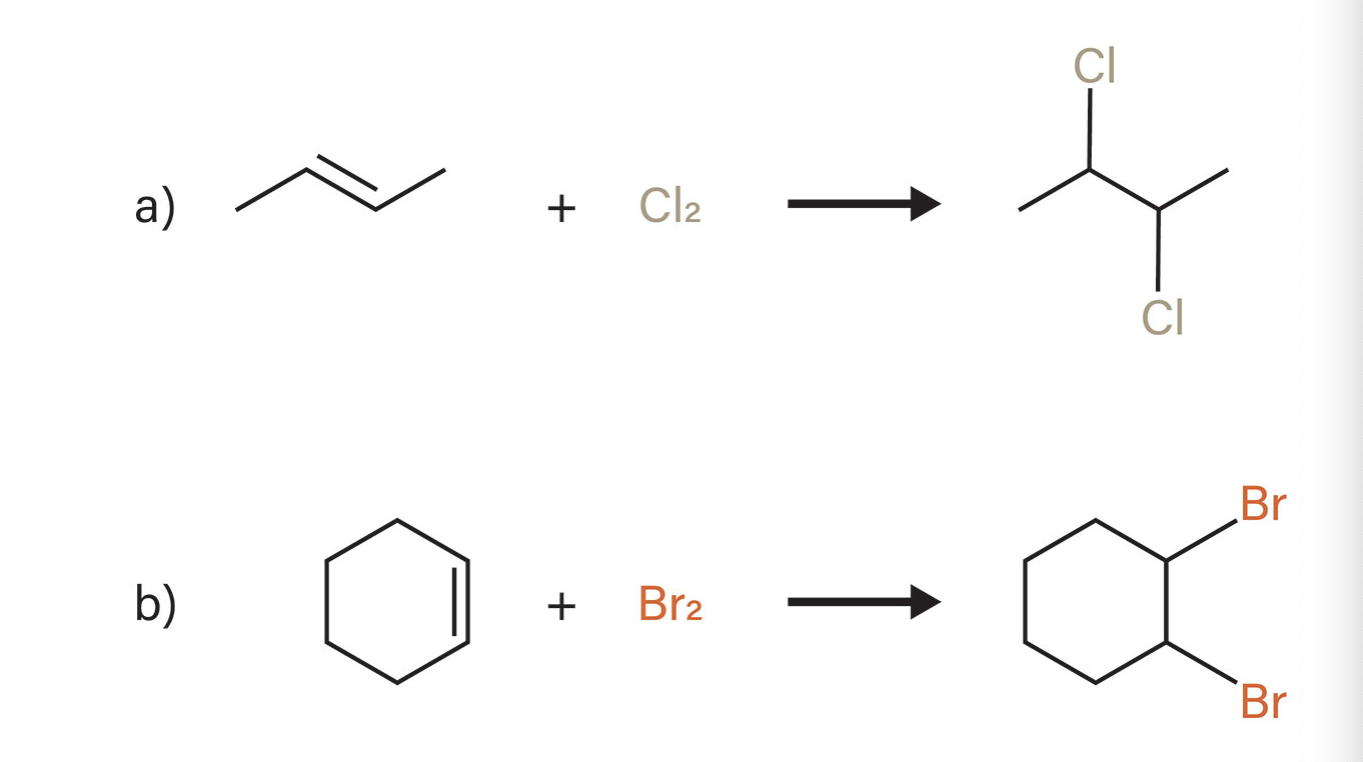
Halogenation (chlorination and bromation)
what type of reaction is this?
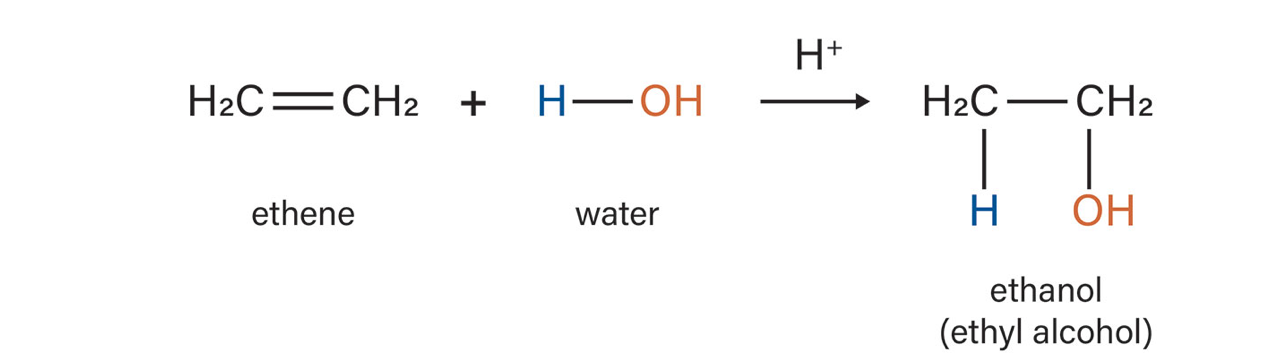
Hydration
What reaction is this?
addition of an acid
what type of reaction is this?
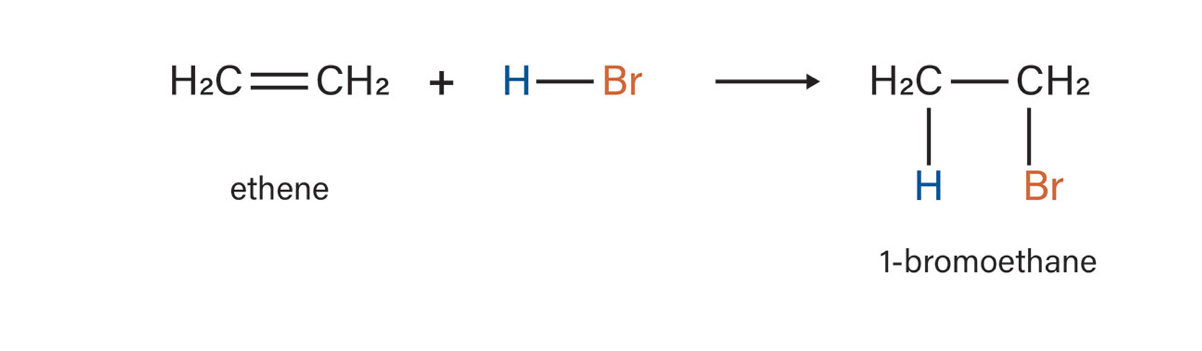
hydrogenation
what type of reaction is this?
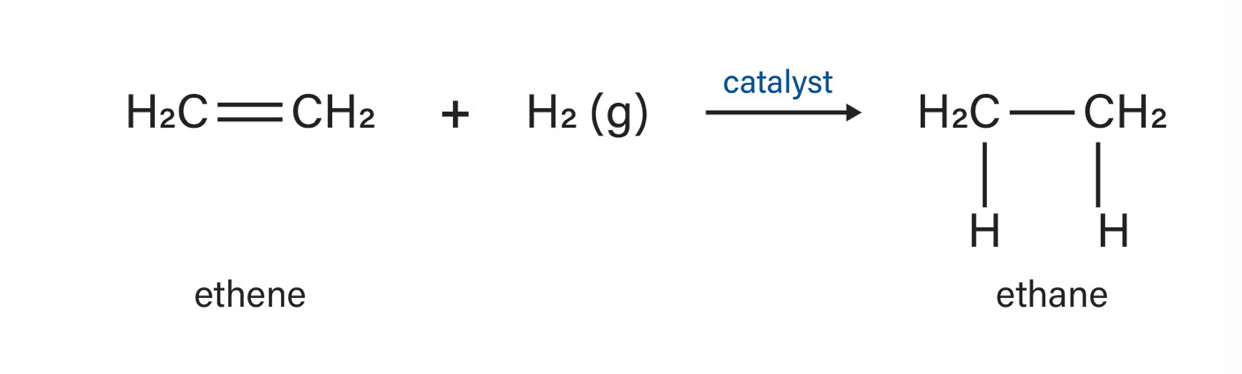
cis
Hydrogenation produces mainly _____ steroisomeres
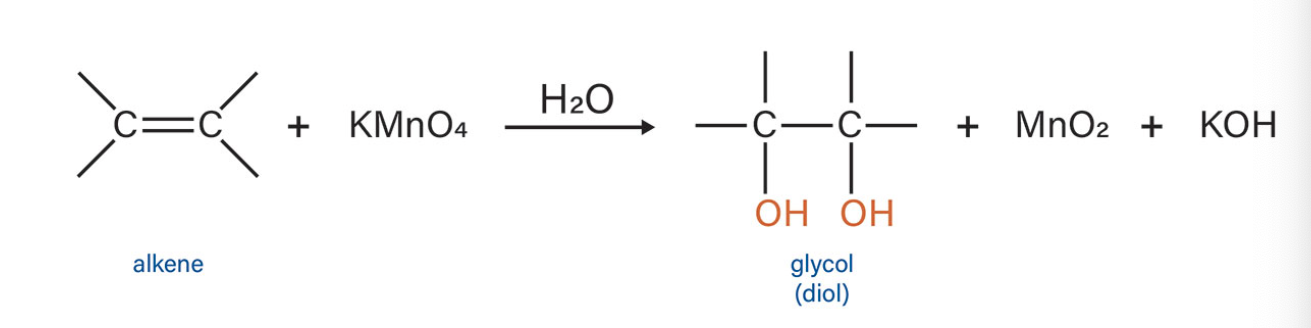
Oxigenation
what type of reaction is this?
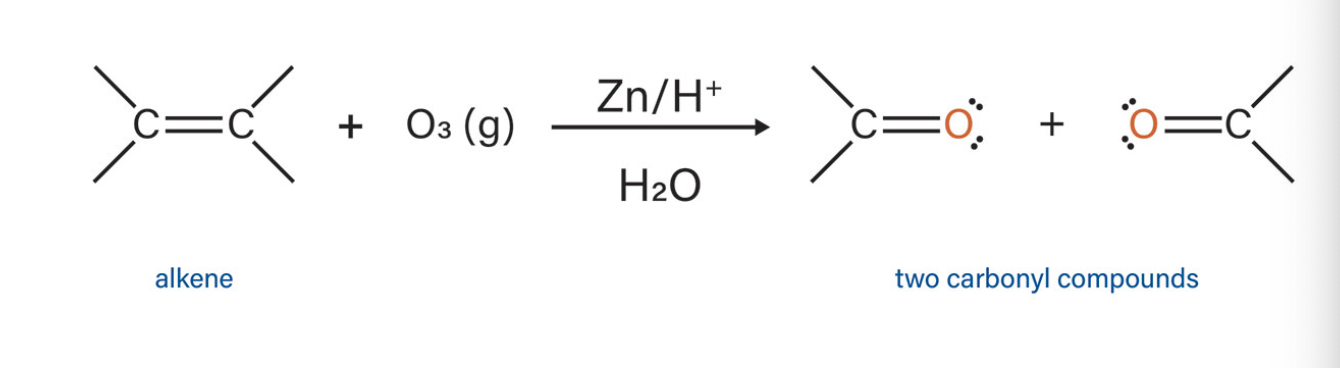
onzonolysis
what type of reaction is this?
aliphatic hydrocarbons
One of the major classes of hydrocarbons represented by alkanes, alkenes, and alkynes
aromatic hydrocarbons
one of the major groups of hydrocarbons represented by benzene. s
-arenes
as a class, aromatic hydrocarbons are also referred to as _____
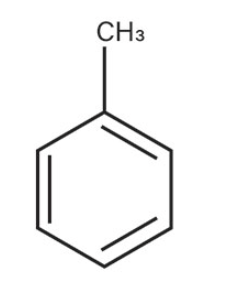
Toluene
what is the common name for this benzene structure?
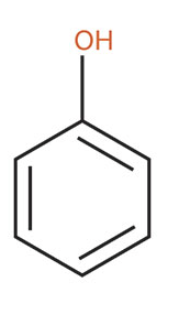
Phenol
what is the common name of this benzene structure?
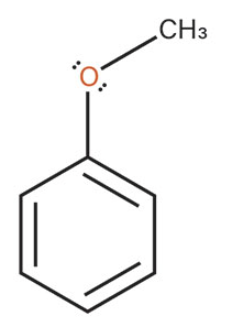
Anisole
what is the common name of this benzene structure?
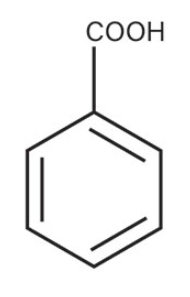
Benzoic Acid
what is the common name of this benzene structure?
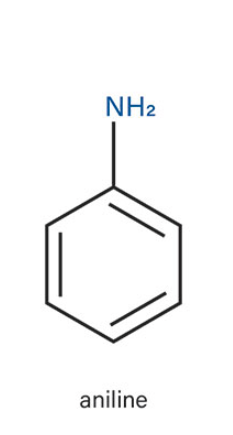
aniline
what is the common name of this benzene structure?
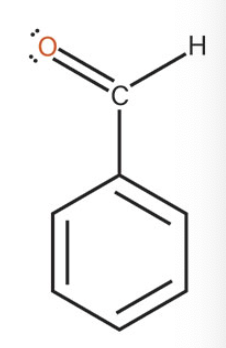
benzaldehyde
what is the common name of this benzene structure?
aryl group
functional groups of aromatic rings
poly cyclic aromatic hydrocarbons
molecules composed of more than one benzene ring fused together
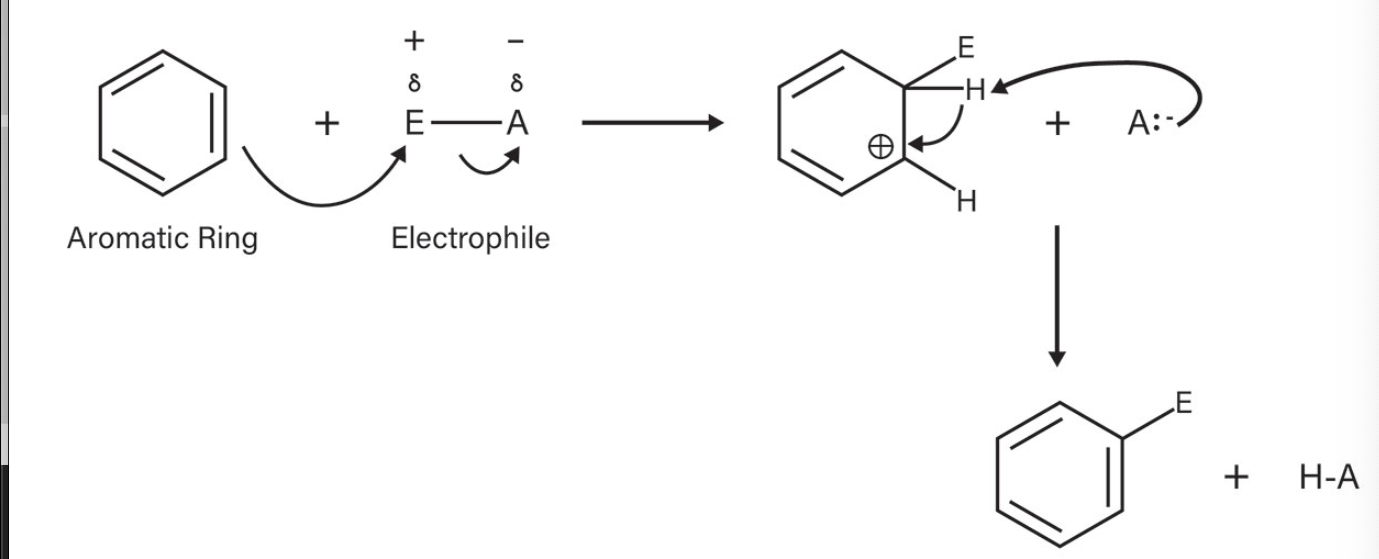
Electrophilic aromatic substitutions reactions (EAS)
benzene derivatives react by substitution in reactions called _______
Electrophiles
chemical species that seeks electrons
EAS Reaction
What is the reaction called?