rganic chem
1/7
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
8 Terms
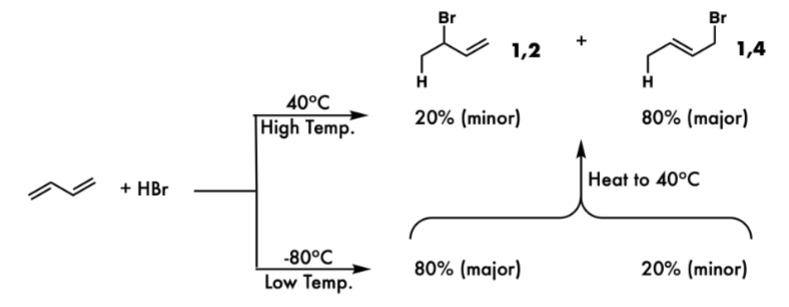
Difference between kinetic and Thermodynamic Product
The kinetic product is the product formed faster, usually at lower temperatures and with less activation energy, while the thermodynamic product is formed more slowly but is more stable, typically at higher temperatures.
Diels-Alder Reactions
A cycloaddition reaction involving a diene and a dienophile that forms a six-membered ring. It is a key reaction in organic chemistry for constructing complex molecules.
Huckles Rule for Aromatic, nonaromatic, and antiaromatic
Huckels Rule: Must be planar and cyclic, have continuous P-orbitals, total number of pi electrons that equals 4n+2. Typically shows unusual stability from aromaticity.
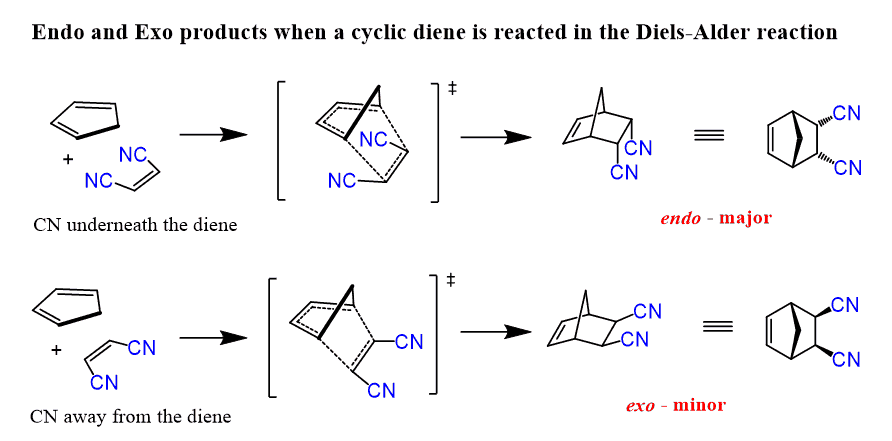
Difference between endo and exo product
Endo = Kinetically favoured (low temp), Exo = thermodynamically favoured (high tempt)
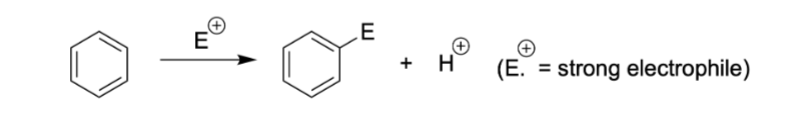
EAS, what is it and what are some common forms:
Electrophilic aromatic substitution
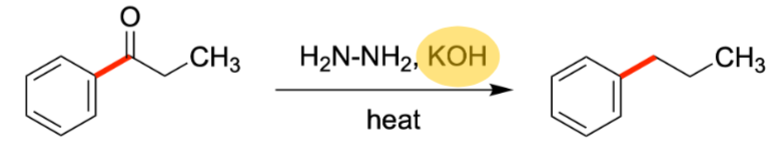
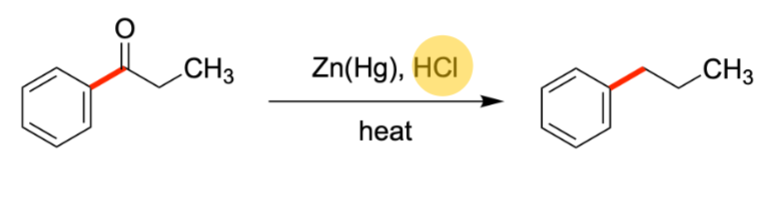
Clemmenson Reduction
Replacement of Carbonyl with H
Wolff-kishner reduction
Alternate form of Clemmenson reduction