1) Metal-Induced Radicals and Radical Reactions
1/88
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
89 Terms
What are three features of reactive intermediates?
Species that are not isolated (transient species, normally low conc in reactions since at high concs a lot of side reactions will occur)
Can be detected by spectroscopic methods, or trapped chemically
Presence often confirmed indirectly through mechanistic understanding
What is a free radical?
Contains an unpaired e-
therefore electron deficient
but normally uncharged
How do you form a radical (radical pair)?
By direct homolysis of a weak sigma bond
Bond order
BO = (bonding e-s - antibonding e-s)/2
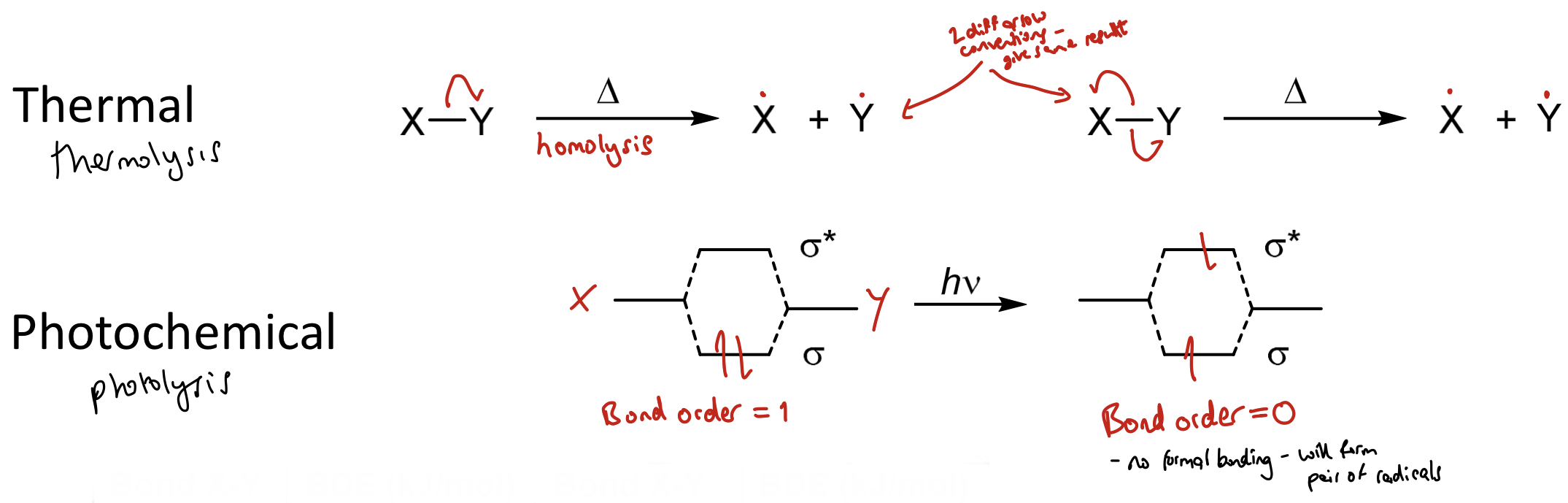
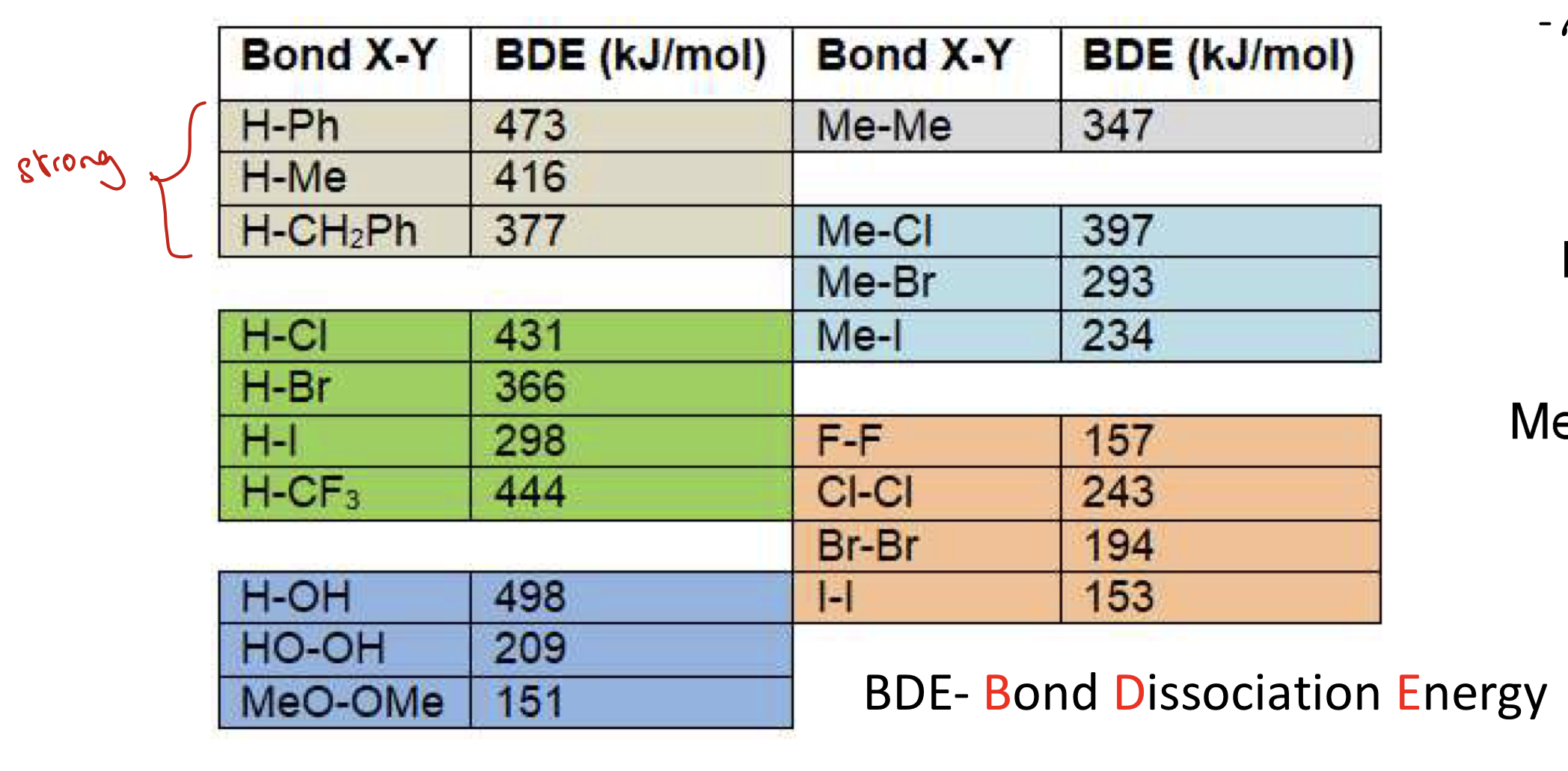
Table of BDE
What’s a radical initiator?
Species to propagate desired chemical process - weak bonds which readily undergo thermolysis as precursors.
What are 2 features of a good thermally activated radical initiator?
stable at room temperature for practicality
rapidly fragment at a controlled rate under usable reaction temperatures (>60ºC → <150ºC
What is the decomposition rate and what is it dependent on
kd predicts the rate of decomposition
Dependent on temperature and the solvent used
(If the radicals react with each other of the solvent then it is an unproductive reaction (the cage effect)) The cage effect causes unproductive “wastage” reactions including recombination of radicals to reform the initiator.
What is the initiator half life (t1/2)
The time required for the reacting species to reduce to half its initial conc at a given temperature and in a particular solvent
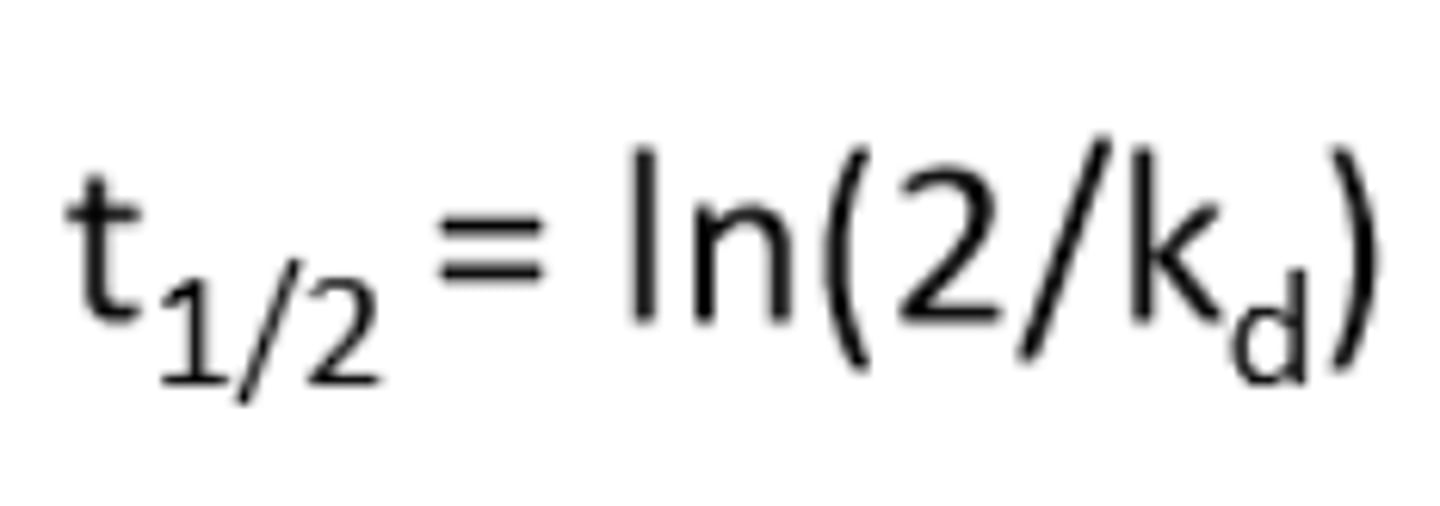
What is the relationship between bond strength and half life?
Weaker bond = shorter half life
Relationship between temperature, kd and t1/2
higher temp = lower half life and higher kd
Name three common radical initiators
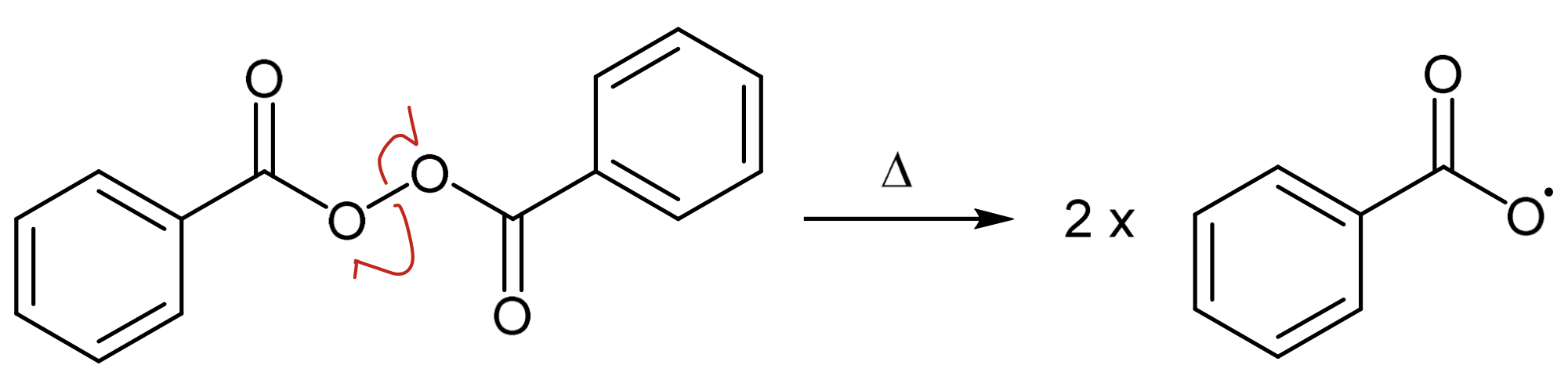
Benzoyl peroxide
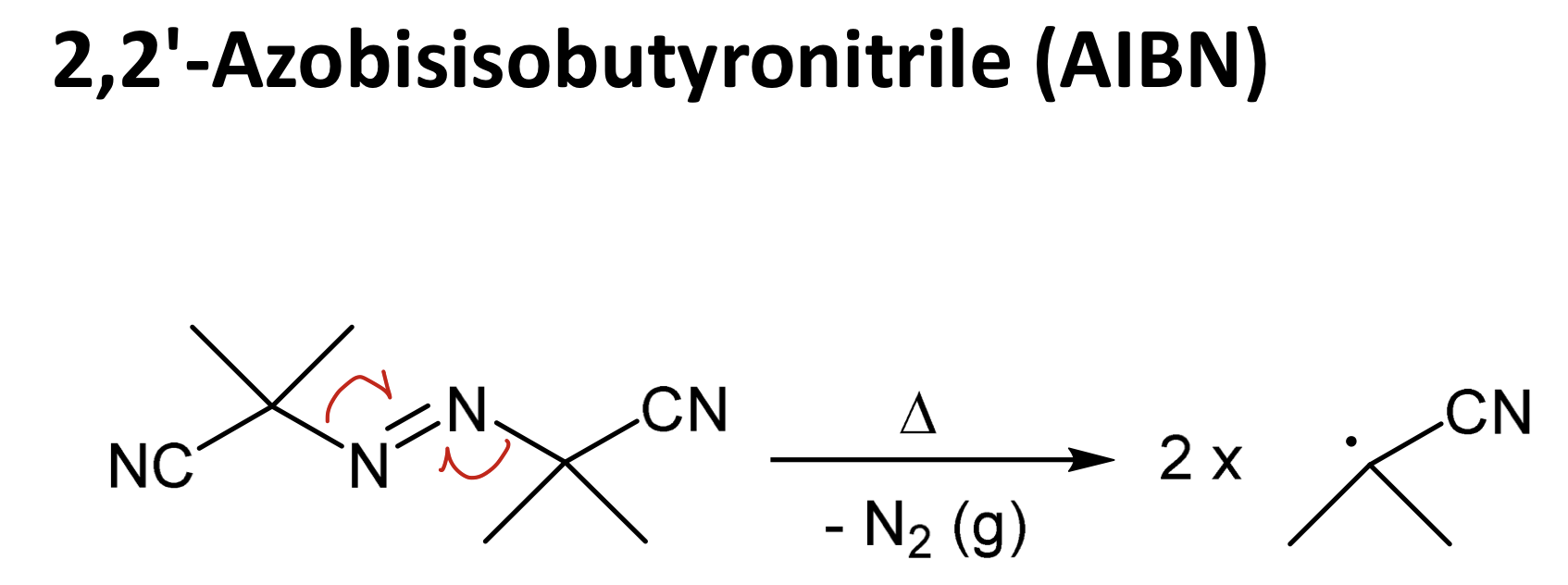
2',2’-azobisisobutyronitrile (AIBN)
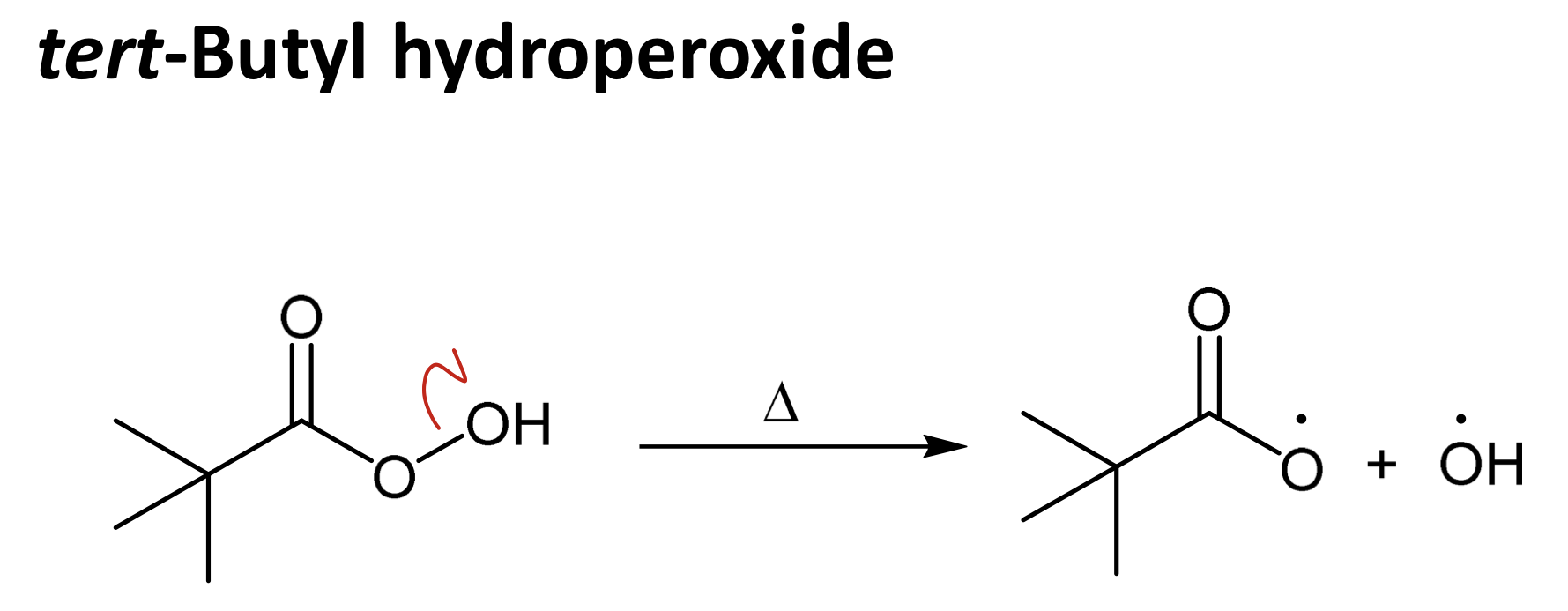
tert-butyl hydroperoxide
Draw benzoyl peroxide and its homolysis
Draw AIBN and its homolysis
Draw tert-butyl hydroperoxide and its homolysis
What is another way to form radicals (other than weak bond homolysis)?
By single electron reduction or oxidation
(addition or removal of one e-)
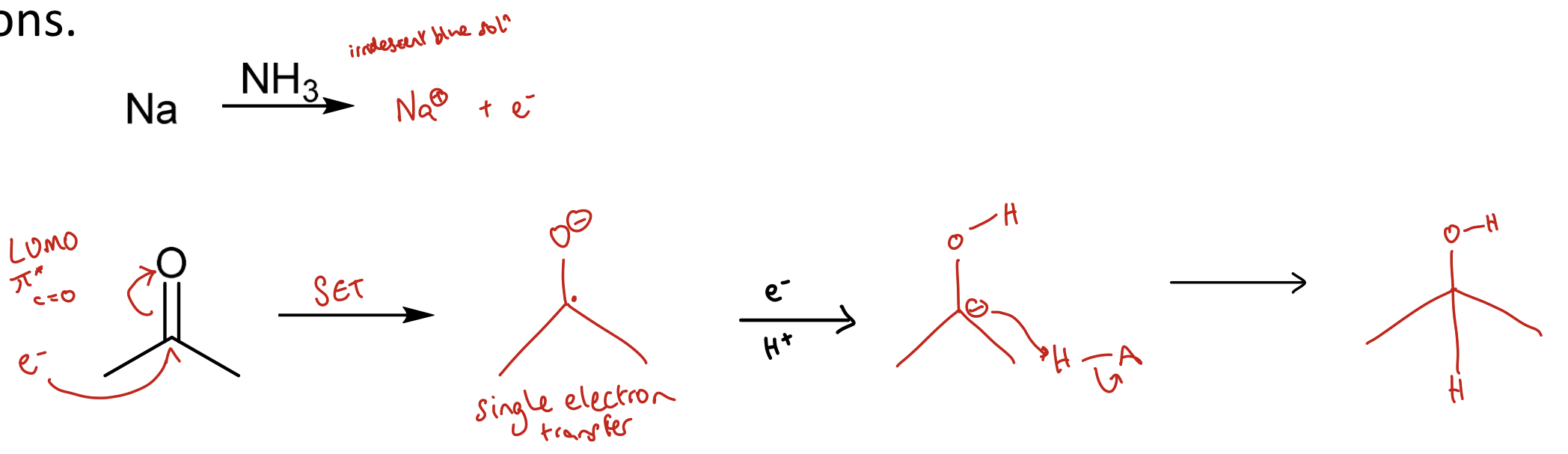
How can reductive metals produce radical anions?
Reductive metals i.e. Li, Na, K and Mg are readily oxidised.
They dissolve in certain solvents (like ammonia or alcohol)
Can produce solvated e-s that occupy pi orbitals (e.g. pi* of carbonyls or aromatics) → prod radical anions
Which metals are reductive and produce radical anions
Li, Na, K, and Mg
also known as the dissolving metal reduction process
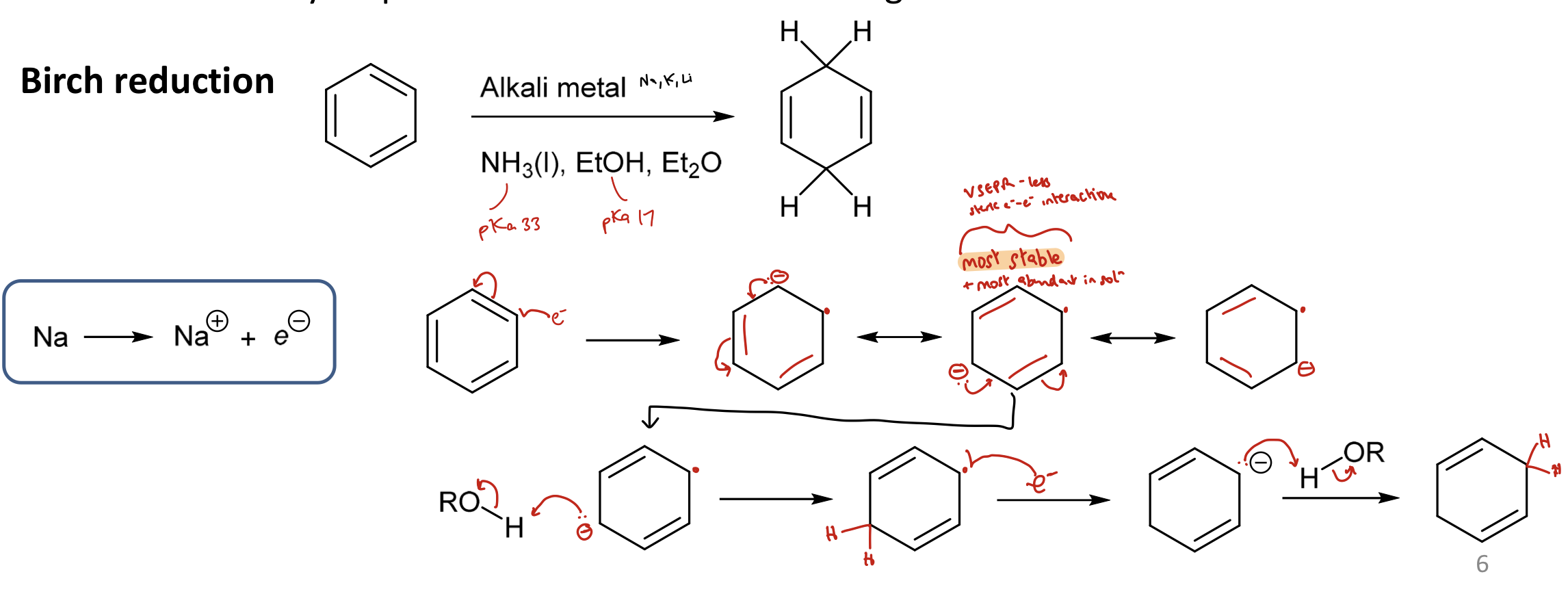
What is the Birch reduction? Show the mechanism.
Reduction of arene to cyclic diene. Reagents: alkali metal, NH3, EtOH and Et2O
During dissolving metal reductions, where does an EDG sit in relation to the double bonds?
EDG always sits on the double bond
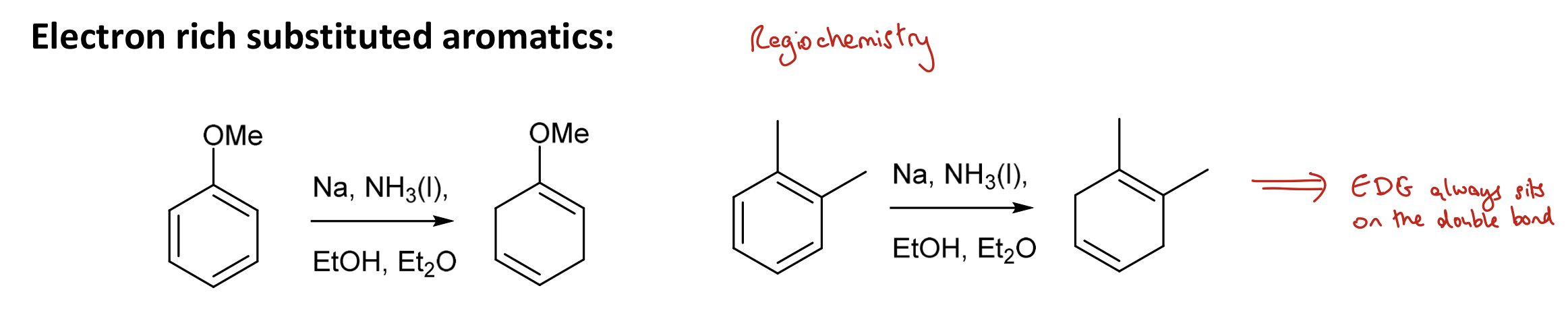
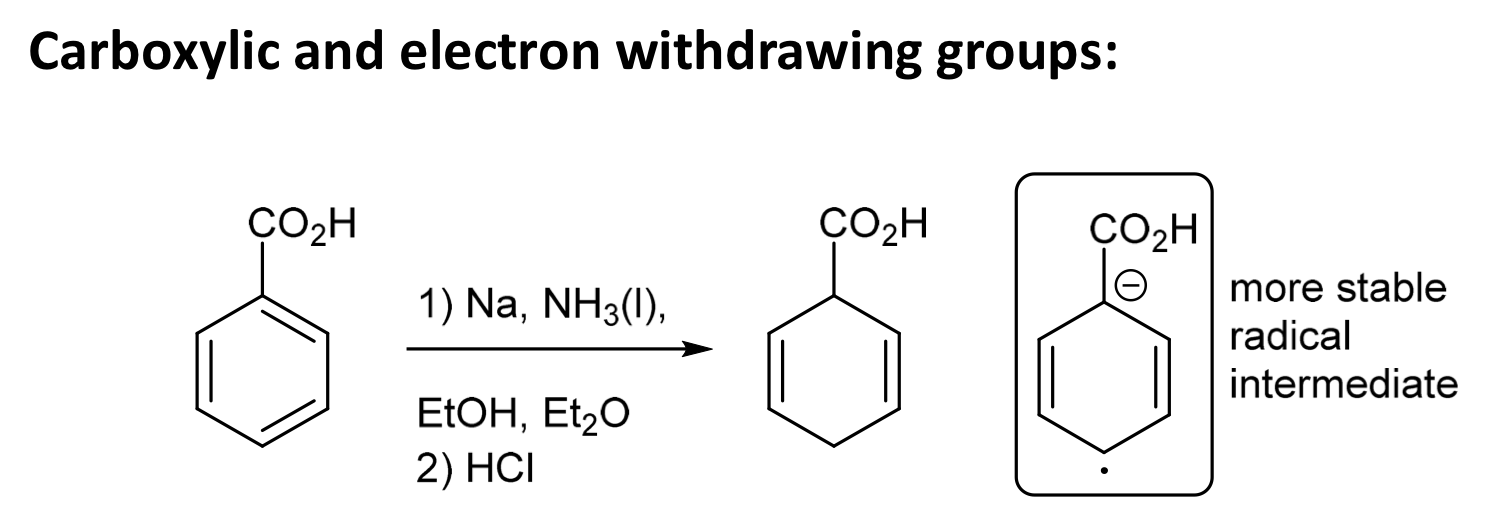
During dissolving metal reductions, where does an EWG sit in relation to the double bonds?
EWG avoids the double bond
e.g. withdraws = avoids (EWG sits between diene units)
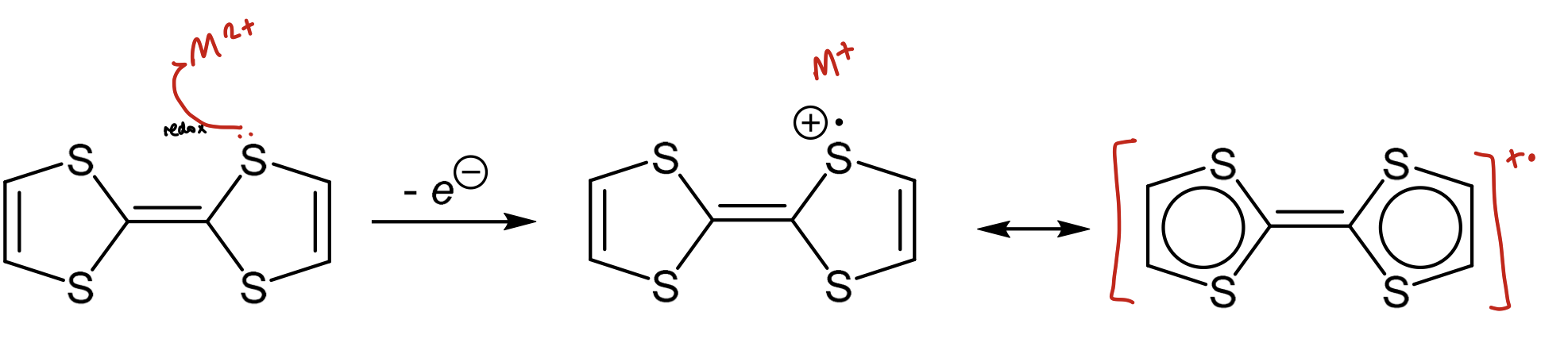
How can Cu2+, Fe3+, Ce4+ be used to generate a radical cation?
They’re one electron oxidising agents
used to remove an electron from high energy HOMO (pi orbitals of aromatics)
What are the three useful synthetic radical processes (excluding polymerisation)?
1) Radical Combination: union of 2 free radicals → new sigma bonded structure
2) Atom Abstraction: Typically radical attacks a H atom, forming a new radical species (displacement process)
3) Addition: The radical can add to a double bond. Regioselectivity is controlled by the resulting stability of the radical generated
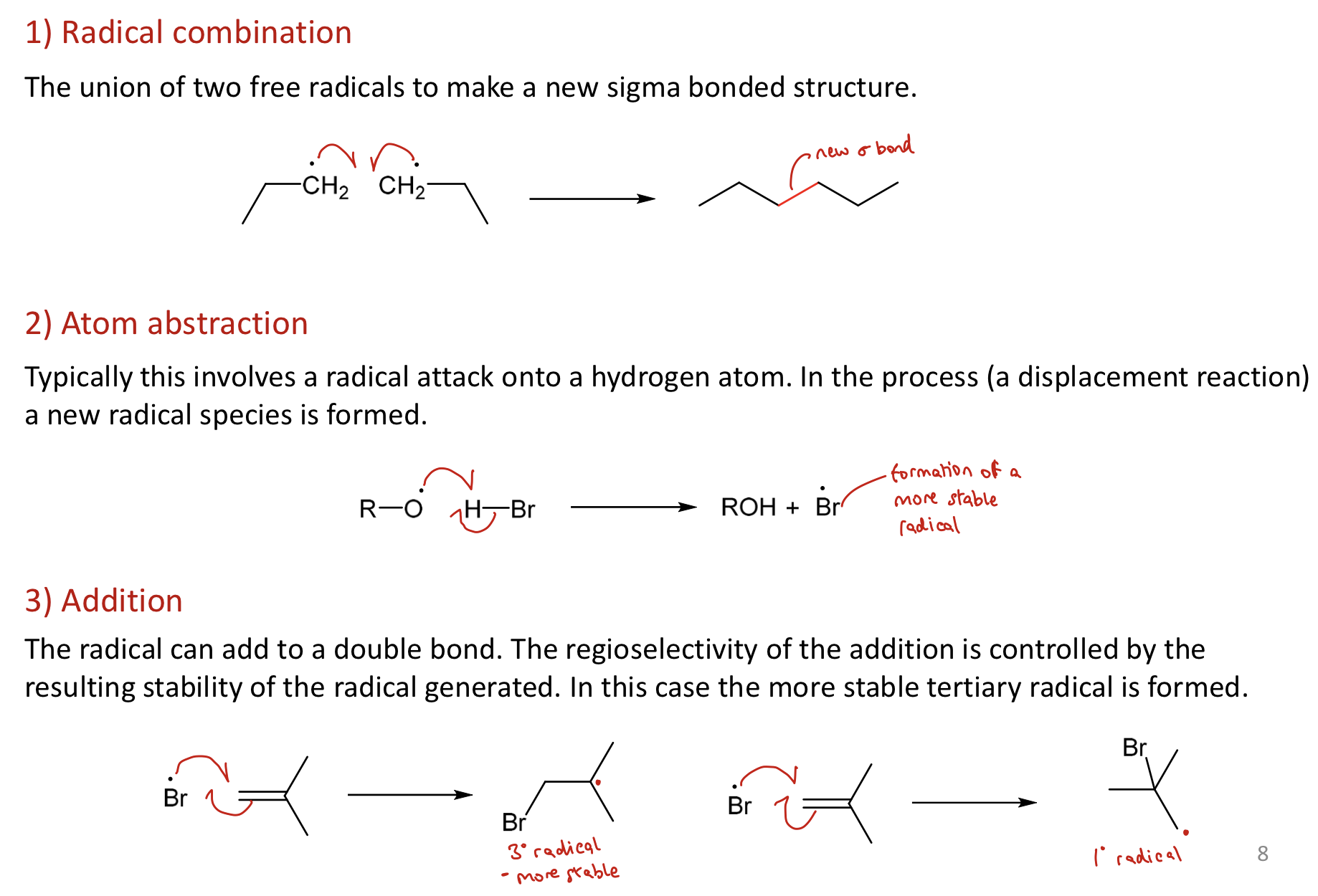
What are simple alkyl radicals like?
Unstable, reactive and v short-lived,
Essentially electron poor species.
What are e- poor species stabilised by?
Stabilised by neighbouring atoms that can donate e- density (think CC stability and hyperconjugation of alkyl grps)
Also by resonance
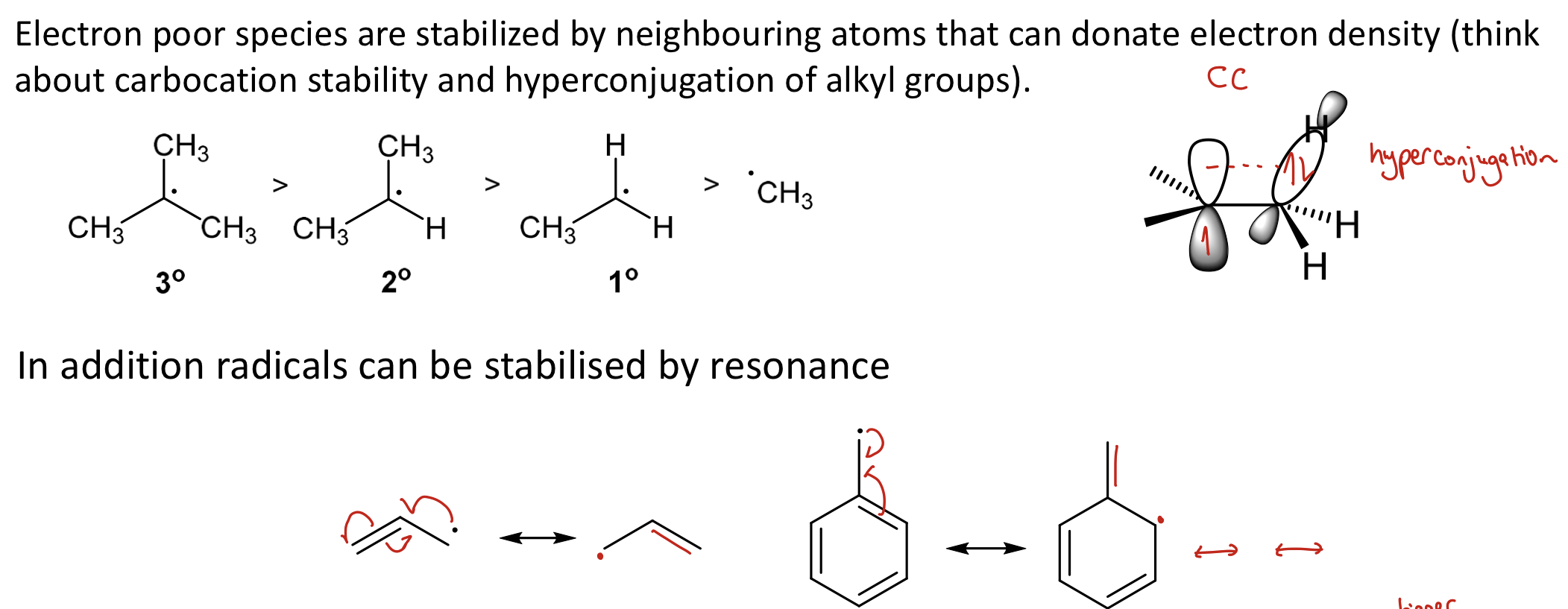
What does radical stability depend on?
More substituted radicals are more stable due to electron donation and hyperconjugation
radicals can be stabilised by resonance
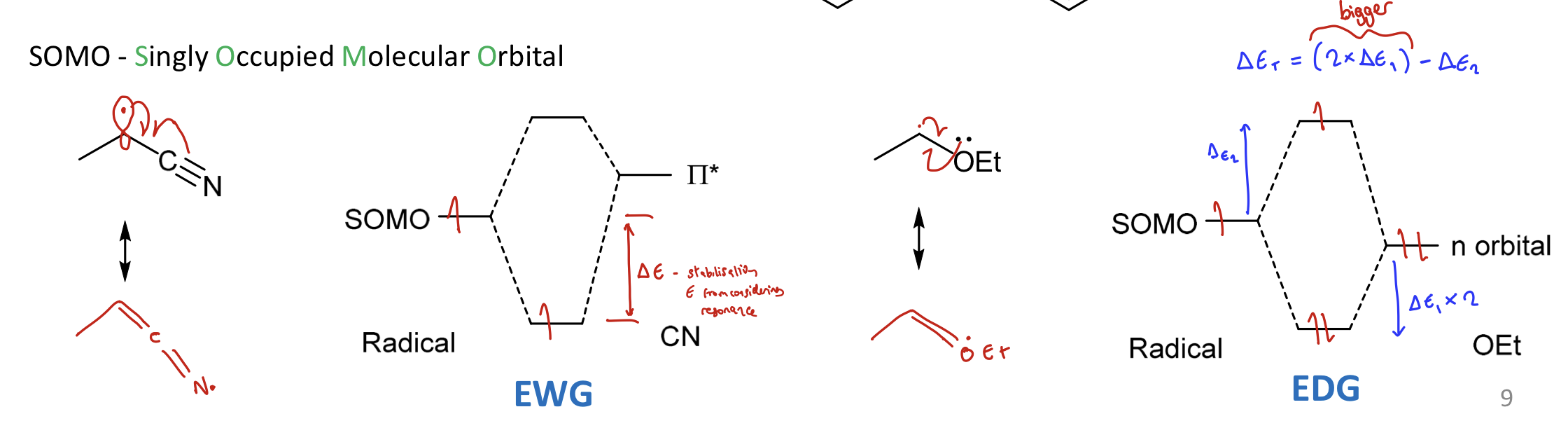
What is the SOMO? How do EWG and EDG differ with this?
Singly Occupied Molecular Orbital
Greater stabilisation energy for EDG radicals
because LP contributing the the MO diagram → 2 electrons go down in E and one electron goes up in E.
Overall ΔE = 2ΔE1 - ΔE2
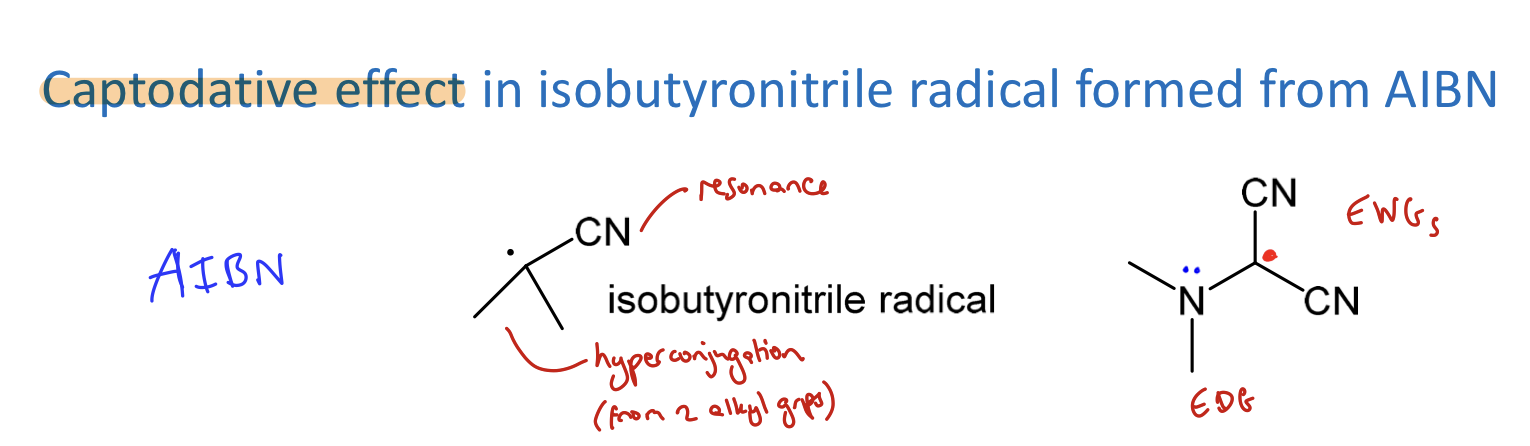
What is the captodative effect?
Alkyl radicals very reactive. Can be stabilised by:
delocalisation into pi systems e.g. allyl or benzyl
adjacent EWG or EDG systems
combo of these factors → further stabilisation
= captodative effect
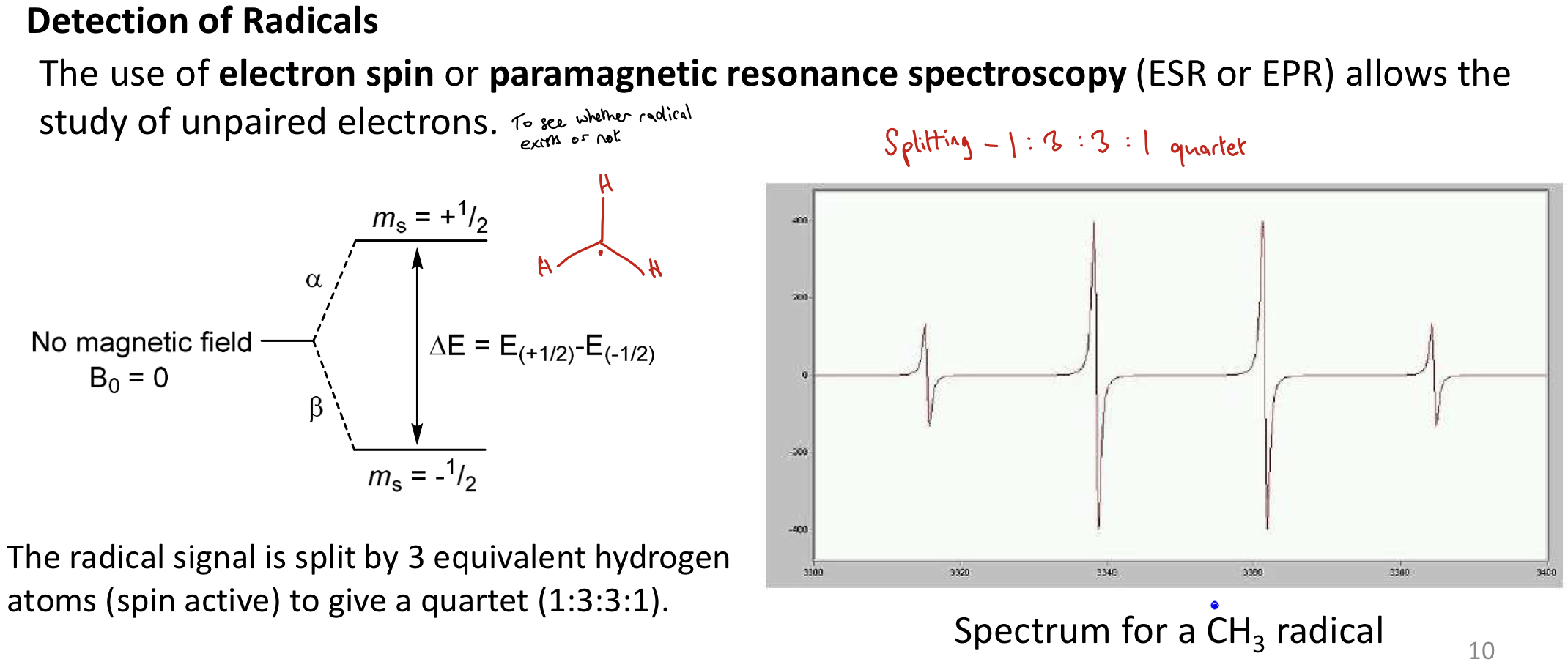
How are radicals detected and why is detection hard?
Detected via e- spin or paramagnetic resonance spectroscopy (ESR or EPR) - allow the study of unpaired electrons (to see if radical exists or not)
For CH3 radical, the 3 Hs (spin active) give a 1:3:3:1 quartet
Detection hard because radicals have short half lives.
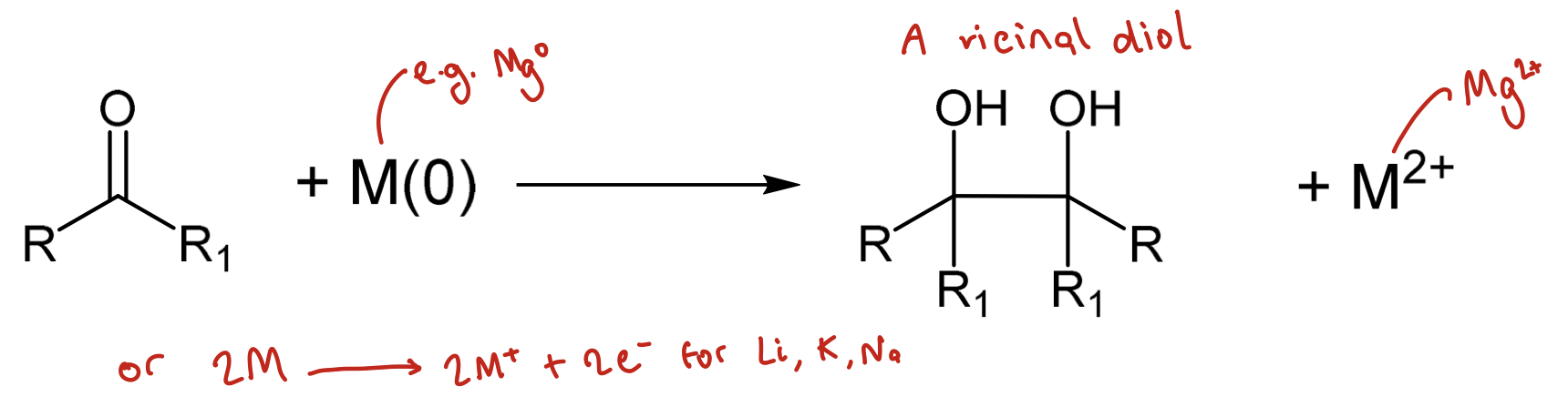
What is the pinacol coupling reaction?
Generates new covalent bond between 2 carbonyl units forming a 1,2-diol (pinacol)
Uses an M(0) metal such as Mg
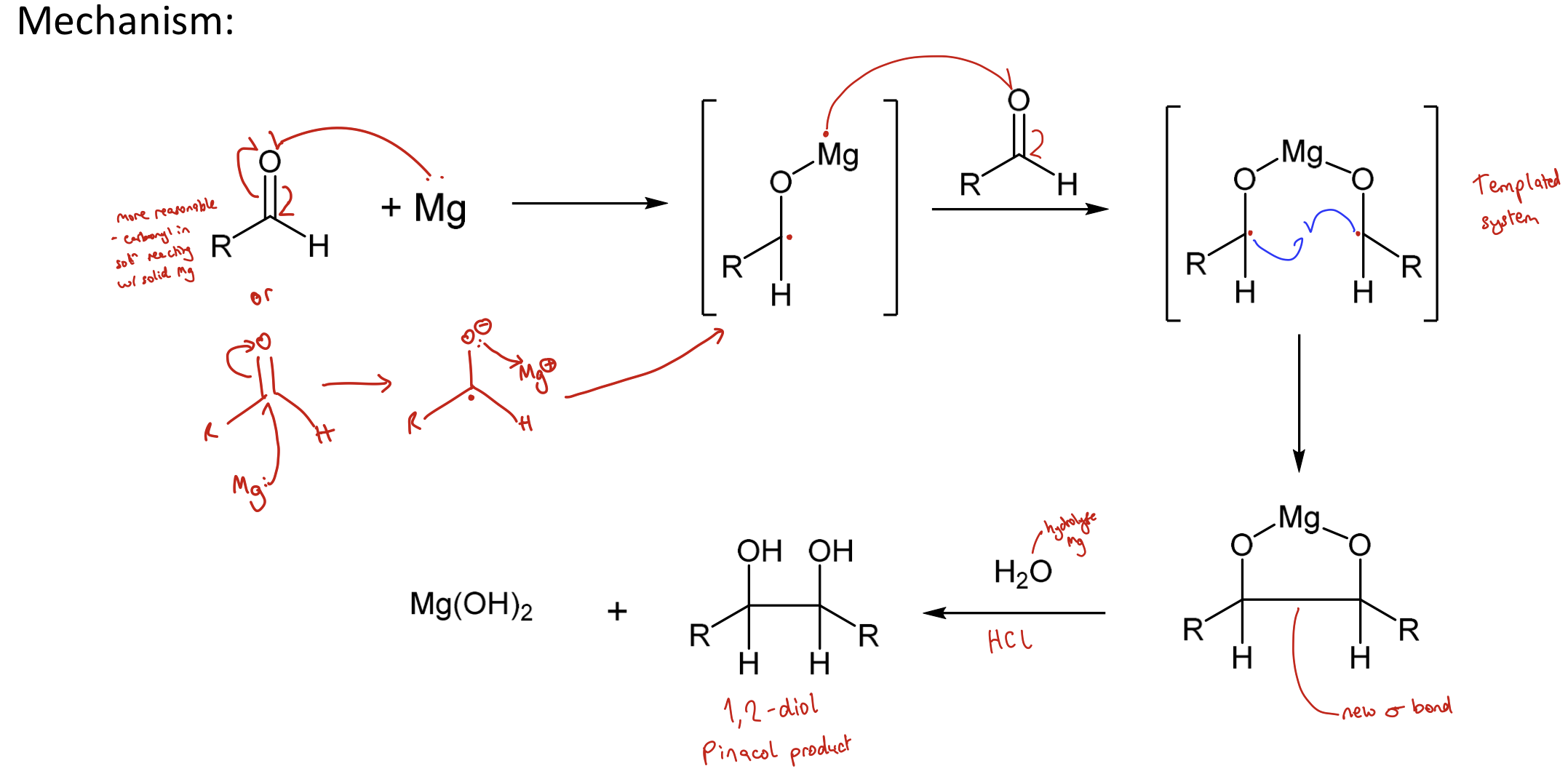
Draw pinacol coupling reaction mechanism.
An electron on Mg attacks the O of a carbonyl which breaks to form a diradical. The radical on the Mg repeats on another carbonyl. The two radicals combine to form a bond. Mg is removed using acid hydrolysis.
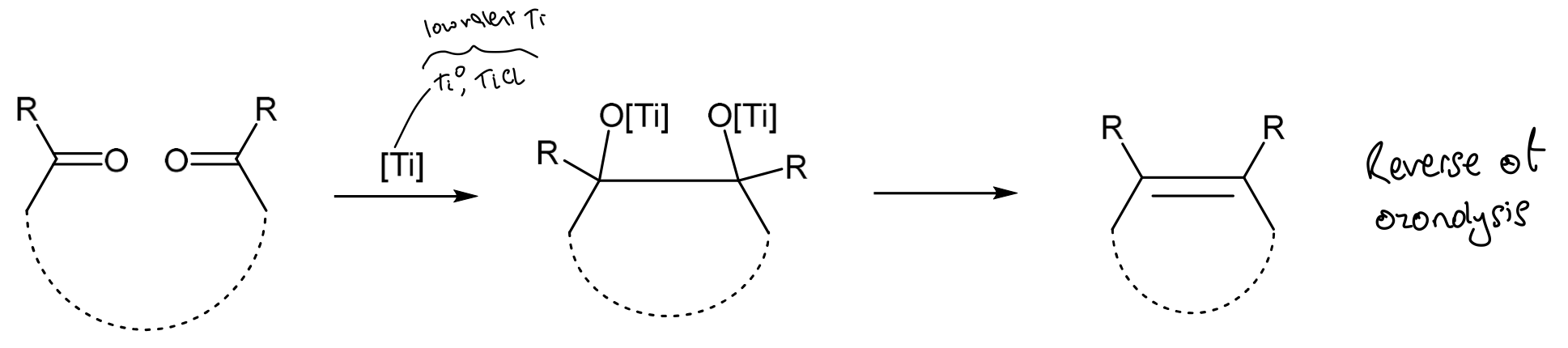
What is the McMurray Reaction?
Combines two carbonyl species to form an alkene using low valent titanium (e.g. TiCl3) and Na/Zn.
Low valent titanium can couple aldehydes and ketones to the corresponding alkenes
The low valent titanium species is prepared by the reduction of a TiXn species, where X is a halide, with K, Na, Li, Zn, Mg, C8K, LiAlH4 (electron donors)
How is TiCl3 reduced by Zn (to make a low valent titanium for McMurray reaction?)
Active [Ti] generated in presence of carbonyl species with zinc as reductant - instant method.
Zn donates its lone pair into the sigma* orbital on Ti-Cl, breaking one of the Ti-Cl bond and kicking a Cl out of the complex
Makes TiCl and Zn(+)Cl
The floating Cl- attacks the Zn+ to form TiCl and ZnCl2
![<p>Active [Ti] generated in presence of carbonyl species with zinc as reductant - instant method.</p><p>Zn donates its lone pair into the sigma* orbital on Ti-Cl, breaking one of the Ti-Cl bond and kicking a Cl out of the complex</p><p>Makes TiCl and Zn(+)Cl</p><p>The floating Cl- attacks the Zn+ to form TiCl and ZnCl2</p>](https://assets.knowt.com/user-attachments/b6955545-deb5-47cf-a3bf-cdf49d963a6e.png)
What are the steps of the McMurray reaction? Show mechanism.
Low valent Ti donates one electron to the carbonyl oxygen, forming Ti-O and breaking C=O
Forms a free radical centre around the carbonyl
Does it again for the other carbonyl
2 radicals come together to form a new sigma bond
One electron on titanium goes into the Ti-O bond, along with an electron from the C-O bond. The other electron from the C-O bond goes onto the new sigma bond (exact same for the other site)
The Ti-O bond is taken off and the new sigma bond is now a double bond
i.e. Ti+ radical attacks O of a carbonyl, breaking the bond to form a radical. This repeats and the two carbonyl radicals combine. The C-O bonds breaks to form an alkene and 2 TiClO
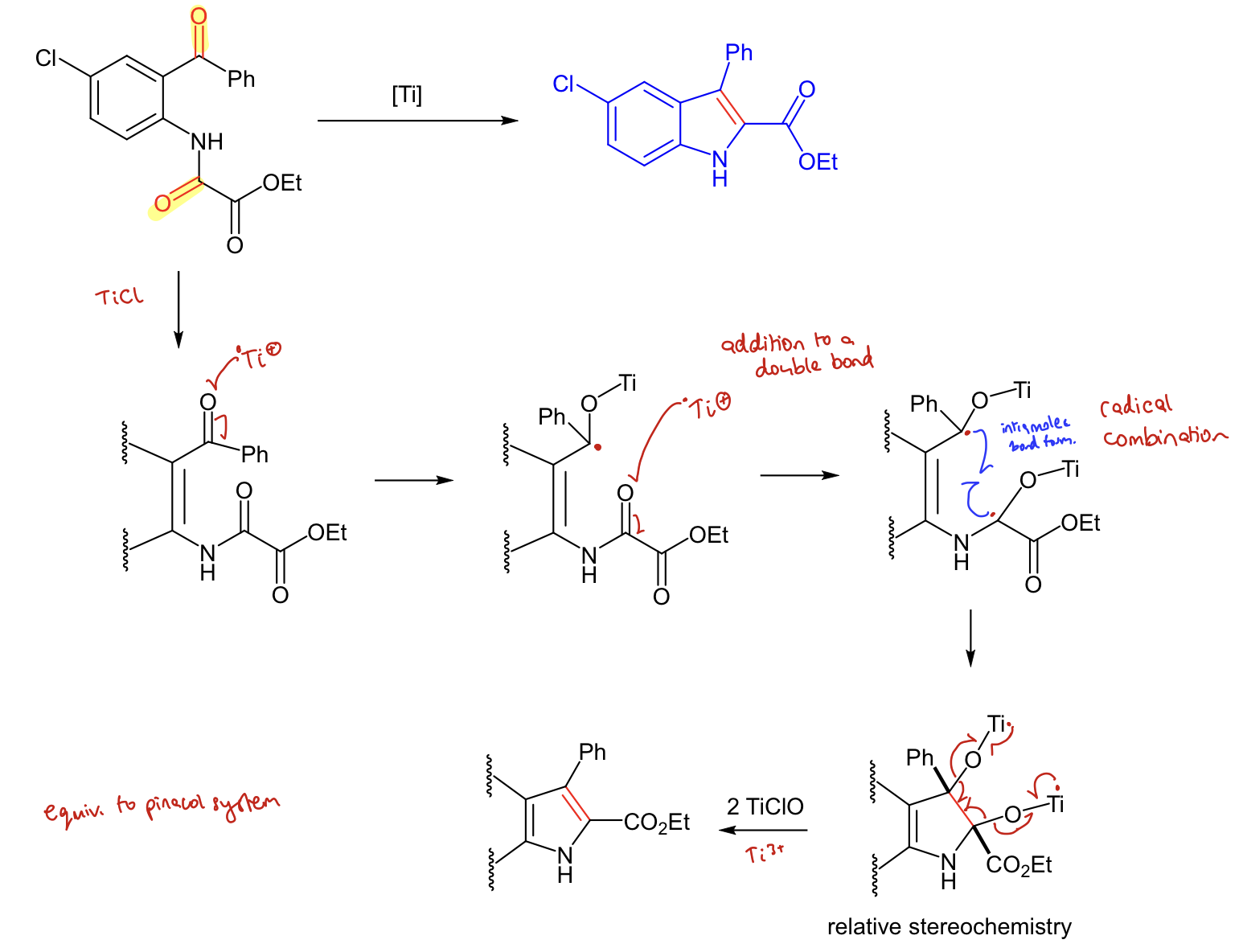
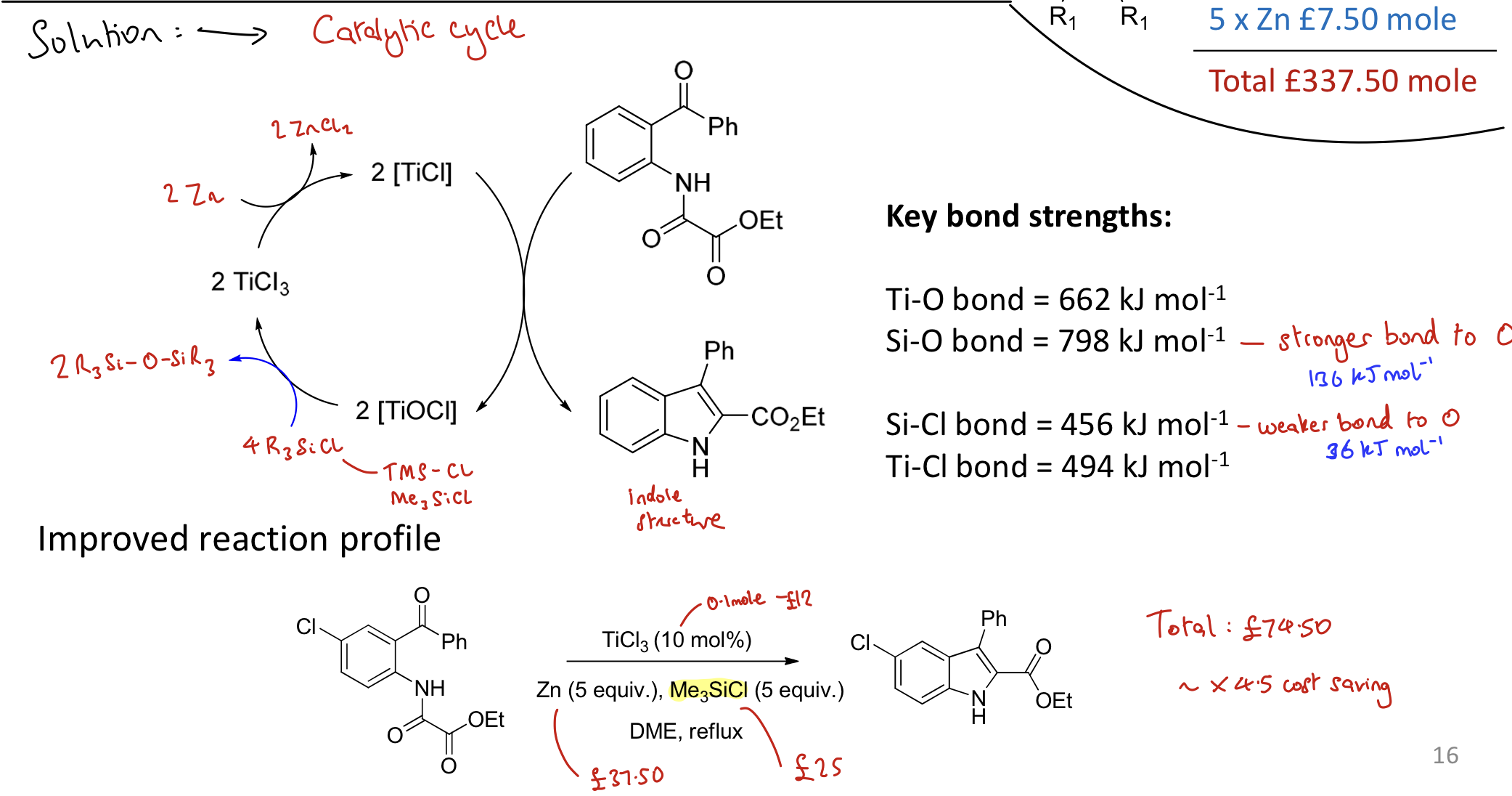
How can the McMurray reaction be improved? What are the benefits
Creating catalytic process. Reduces costs.
A replenishing species (Me3SiCl) is introduced → allows low valent Ti species to reform and be reused.
Using 4 Me3SiCl to oxidise 2 TiOCl back to 2 TiCl3 to recover the catalyst, removing 2 (Me3Si)2O
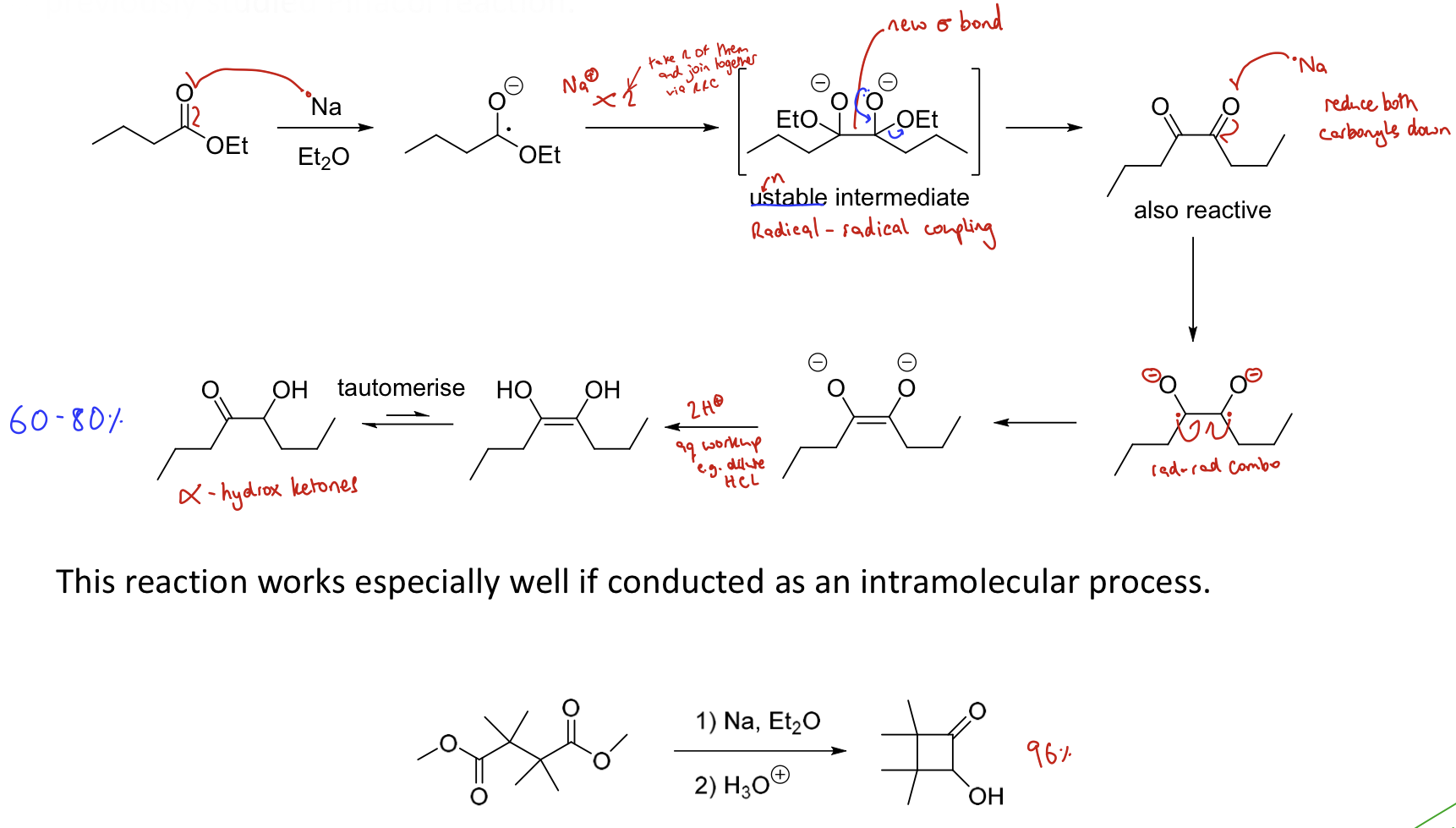
What is the Acyloin reaction? Draw the mechanism.
Reductive coupling of esters (similar mechanism to pinacol reaction)
This converts two esters to an α-hydroxy ketone using Na
Metal reduces carbonyl to form a radical
Radical coupling to join two former carbonyls together
Metal reduces carbonyls again
Radical coupling to form a double bond
Add H+ to form a dihydroxy
Tautomerisation forms α-hydroxy ketones
Works v. well as intramolecular process.
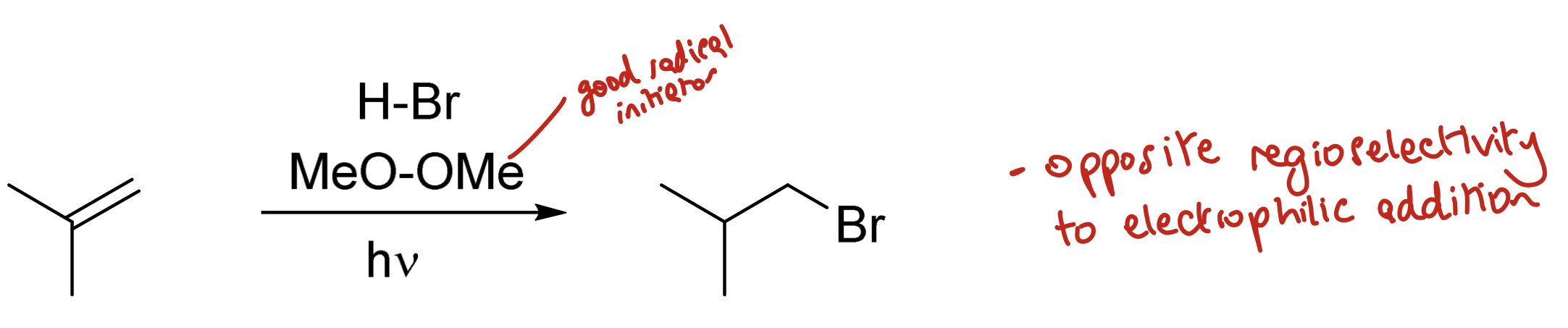
How can HBr be added across a double bond to the least substituted carbon?
Using HBr through a radical process, activating using MeO-OMe (good radical initiator) and hν
(opposite regioselectivity to electrophilic addition)
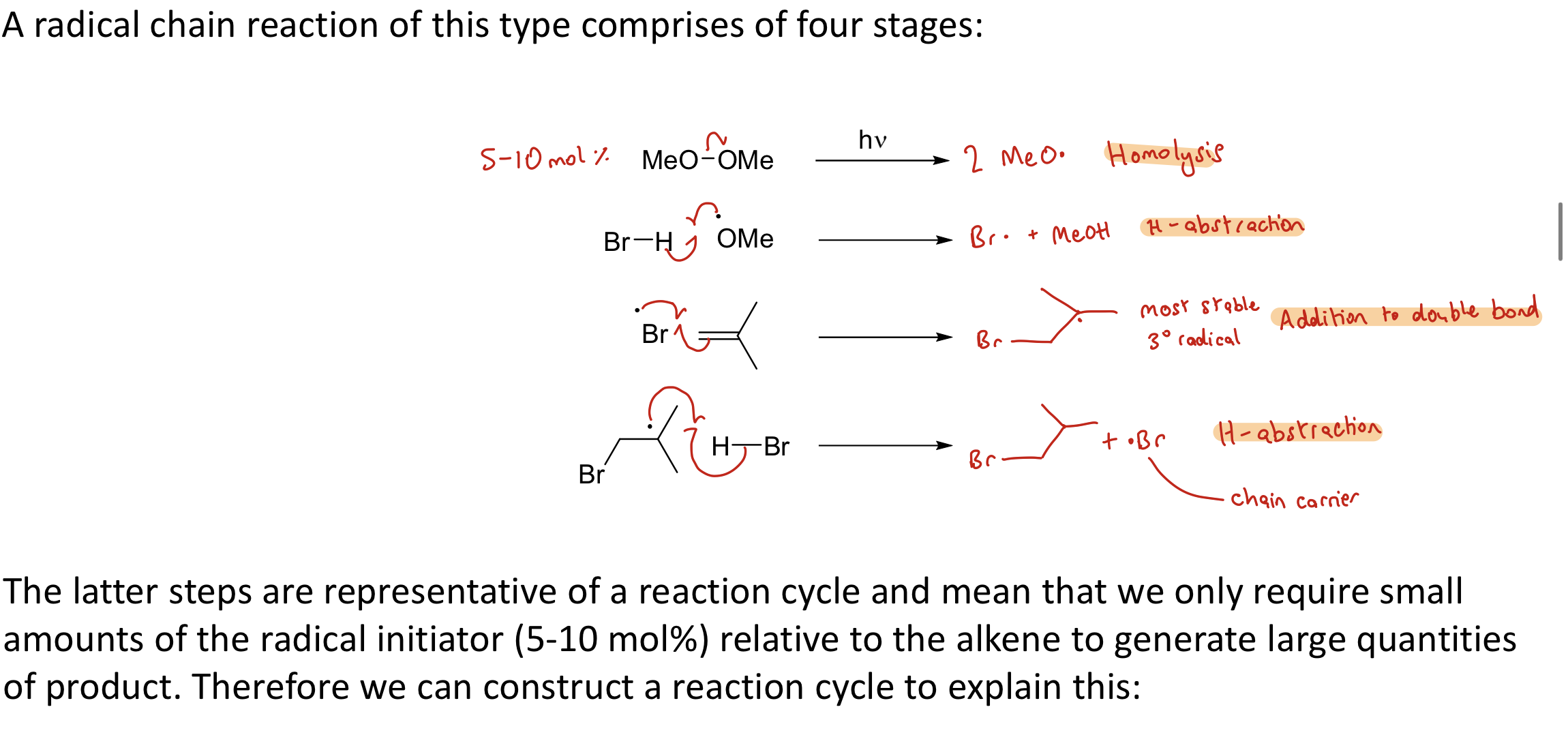
What are the 4 stages of a radical chain reaction?
Homolysis (formation of the radicals)
H-abstraction
Addition to double bond (of free radical to other structure to form most stable radical)
H-abstraction
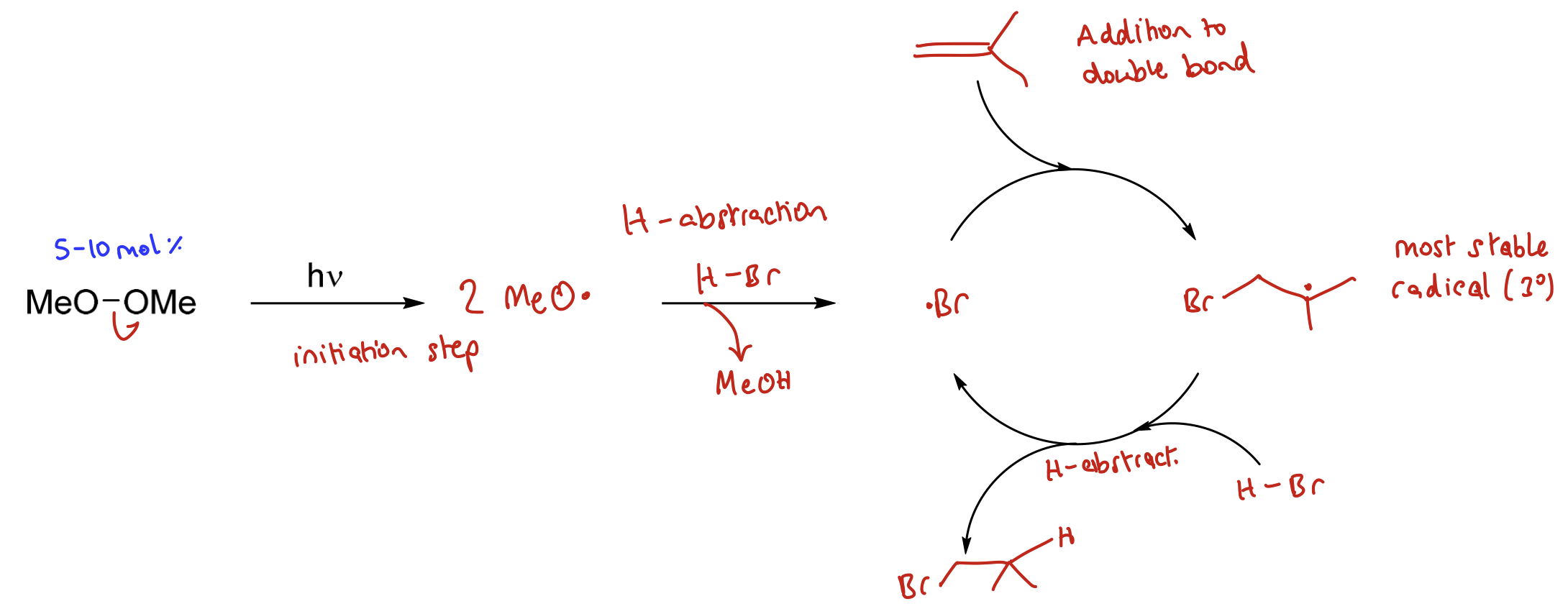
Latter steps representative of a reaction cycle so only require small amounts of the radical initiator to generate lots of product. Can present this reaction in a reaction cycle.
Show radical chain reaction cycle.
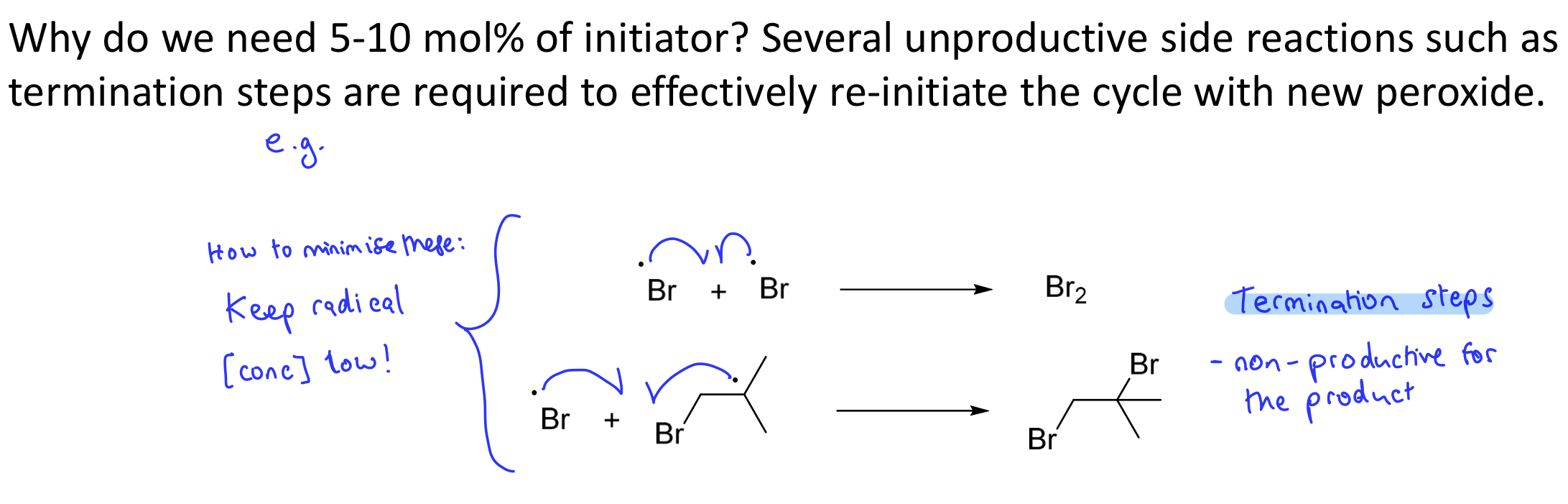
Why do you keep the concentration of the initiator low in a radical chain reaction?
Only 5-10%.
To minimise unproductive side reactions (some termination steps)
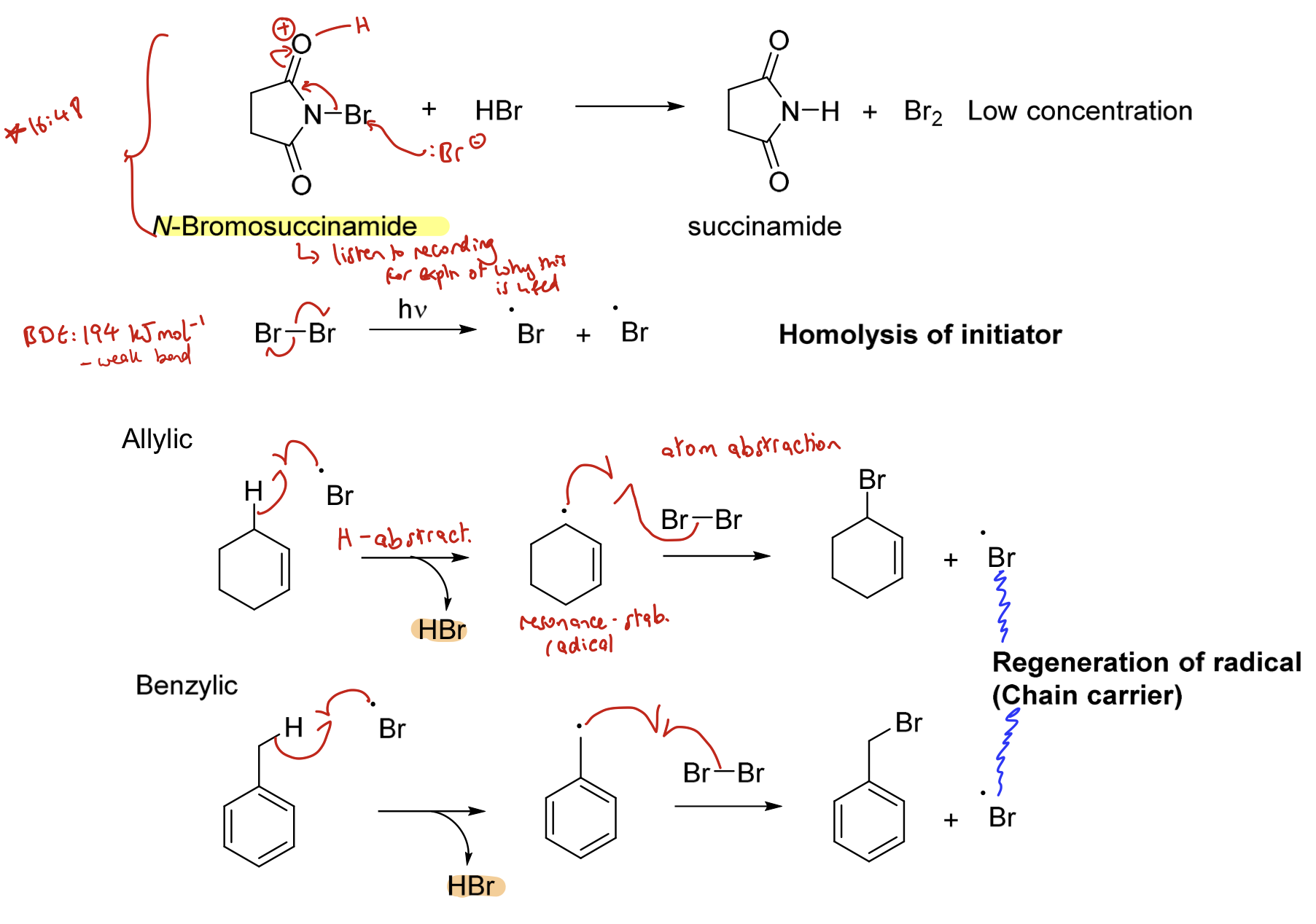
How can an allylic or benzylic hydrogen be substituted for a halogen (mainly Br, Cl and I)? Draw the mechanism.
Radical halogenation of an allylic or benzylic compound using N-halosuccinamides.
Homolysis of the initiator (halogen)
Halogen abstracts hydrogen (hydrogen abstraction) to form an allylic/ benzylic radical
This radical reacts with the halogen (e.g. Br₂) to create a halogen substituted allyl/ benzene and regenerates the radical (the chain carrier)
Can be represented in a cycle (not shown here).
What is the oxymercuration reaction and what are the initial steps?
A reaction that turns an alkene into an alcohol using HgCl₂ and NaBH₄.
1) Hg(II) salt coordinates to the alkene, making the alkene more electrophilic
2) OH then attacks the alkene, kicking out Cl.
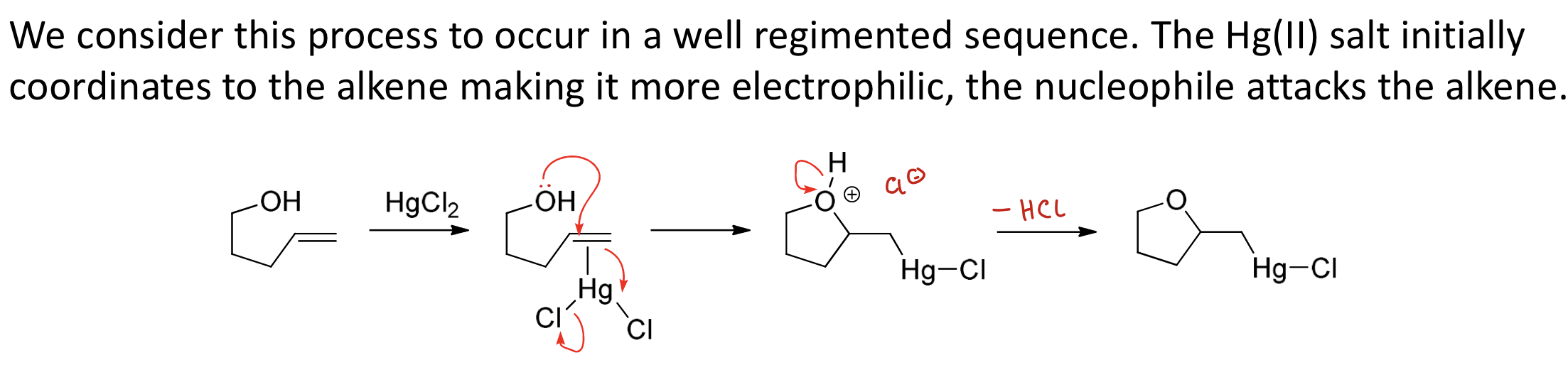
Draw mechanism of oxymercuration reaction.
Rxn turns an alkene into an alcohol using HgCl₂ and NaBH₄.
1) Hg(II) salt coordinates to the alkene, making the alkene more electrophilic
2) OH then attacks the alkene, kicking out Cl.
3) Nucleophilic ligand exchange (NaBH₄ then replaces Cl with H. )
4) R-Hg sigma bond homolysis (initiation) to give radicals
5) Propagation (H abstraction of the H attached to the Hg of another molecule)
6) Hg removed
7) Termination
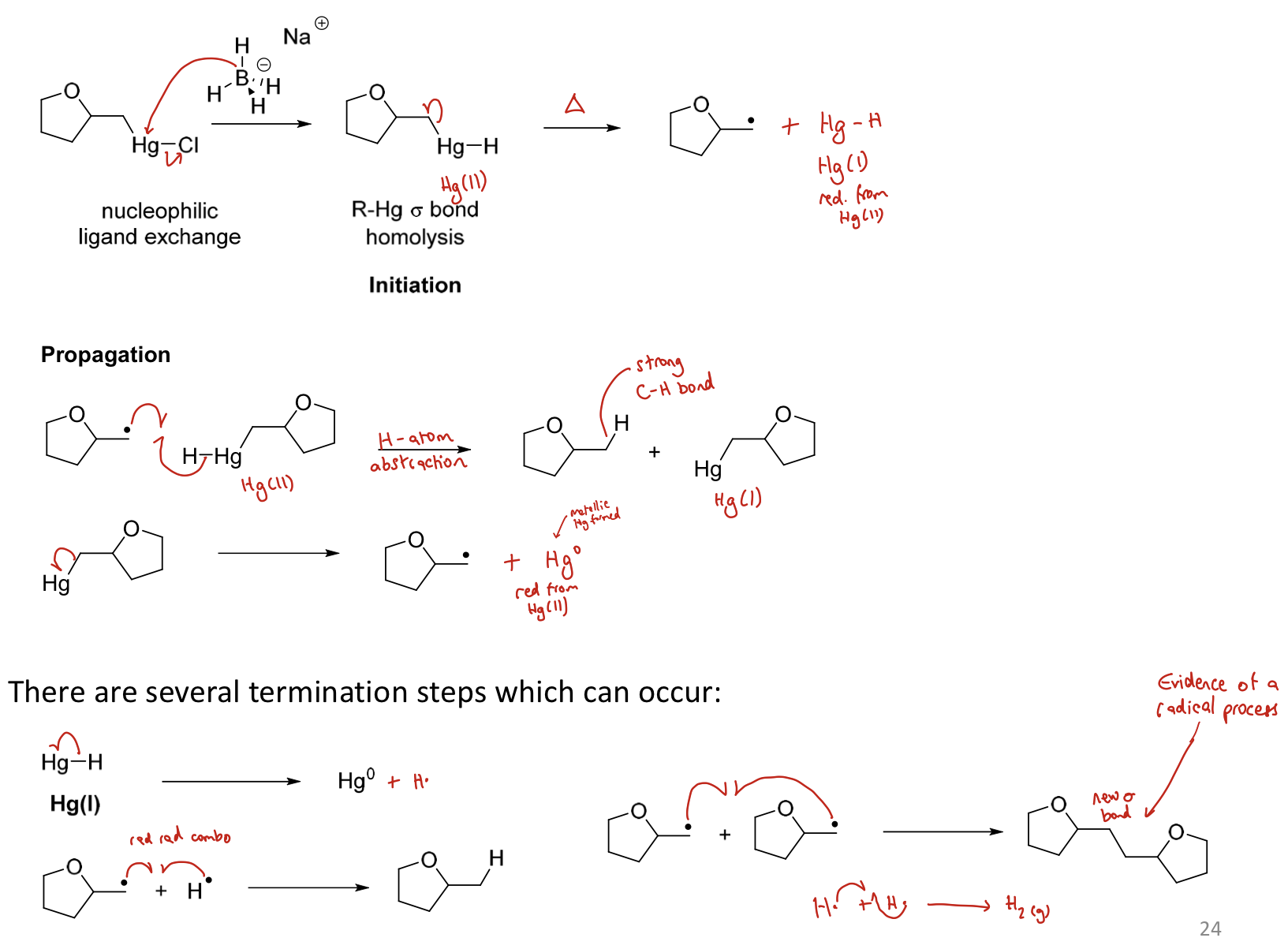
What is the benefit of knowing the radical involved?
Allows synthetic chemists to use the sequence to create more elaborate structures
How can a halogen be replaced by H?
What is used in a dehalogenation reaction?
Dehalogenation - radical substitution of C-X bonds
Bu₃SnH can be used to furnish a halogen-hydrogen exchange
Catalytic AIBN is used to initiate the radical-chain reaction (AIBN added slowly so does not quench R or react with the alkene instead)
What are Baldwin's Rules?
Used to name ring closures and used to determine if they are allowed
N-exo/endo-tet/trig/dig
N = number of atoms forming the ring (ring size)
exo = if the bond being broken is outside of the ring being formed
endo = if the bond being broken is part of the ring being formed
tet/ trig/ dig = hybridisation of the reaction centre
If sp3 hybridised = tet
If sp2 hybridised = trig
If sp hybridised = dig
Learn table to determine whether or not the cyclisation can actually occur
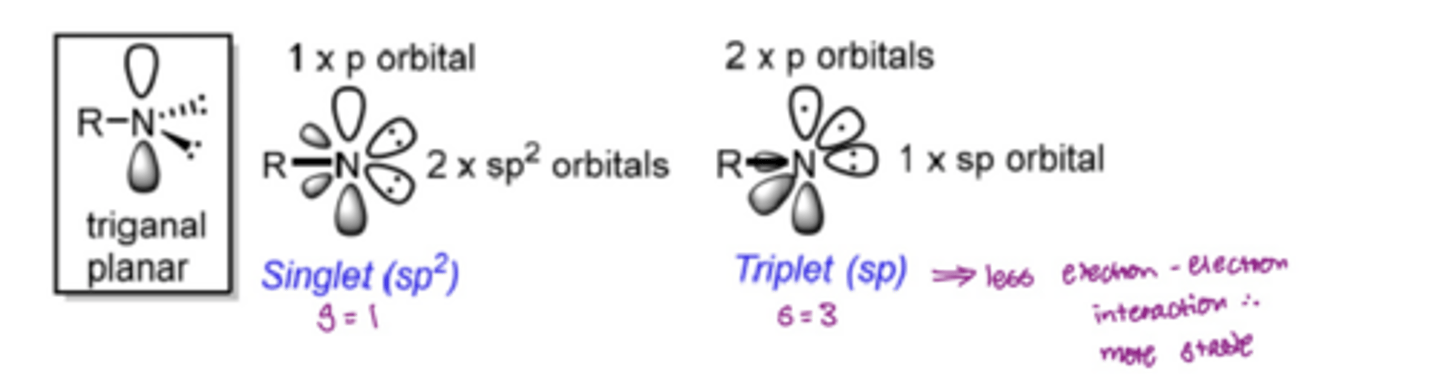
What are 4 features of a carbene
Neutral charge
Divalent (2 valence electrons)
Highly reactive carbon intermediates with short lifetimes
Exist in 2 different spin states: singlet and triplet
What is the difference between a singlet carbene and a triplet carbene
Singlet carbene:
Carbon is sp2 hybridised
The lone pair of electrons occupy the same p orbital
Can react in a concerted way
Triplet Carbene:
Carbon is sp hybridised
The lone pair of electrons occupy different orbitals
Most stable
Can be referred to as a diradical and react in a stepwise sequence
The different spin states react differently, forming different products
INSERT PICTURE
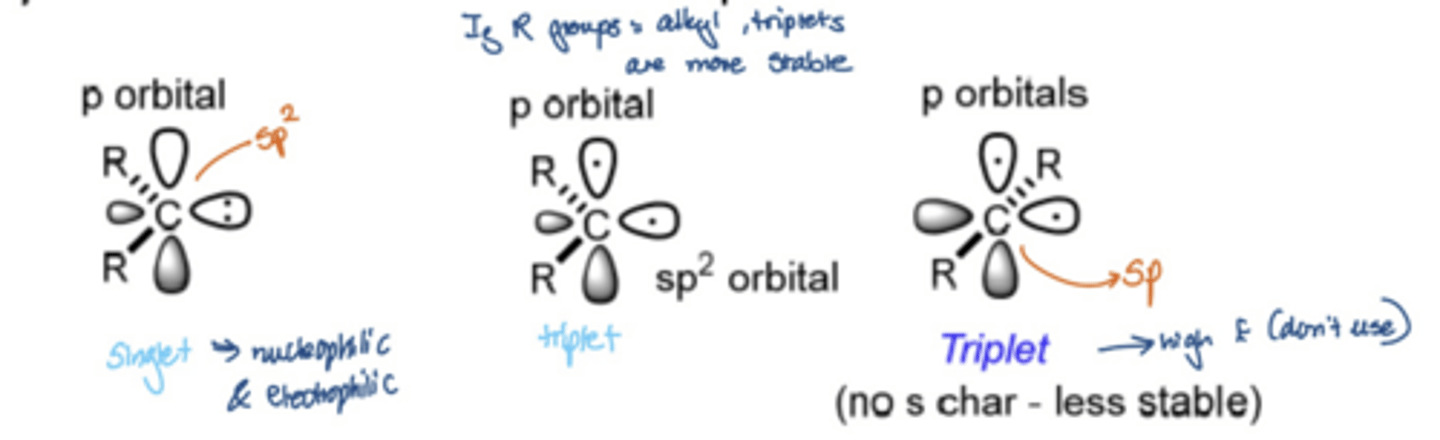
What happens when a triplet carbene reacts with an alkene
One electron from the carbene reacts with one electron from the pi bond to form a diradical intermediate
C-C bond rotation occurs to get the unpaired electrons close together
Electron spin inversion occurs so the electrons can react
ring closure
IF rotation occurs before spin inversion, the trans product is made (inversion slower than bond rotation)
IF spin inversion occurs before bond rotation, the cis product is made (very fast spin inversion)
STEP WISE PROCESS
What happens when a singlet carbene reacts with a cis-alkene
Similar to a chelotropic reaction
Lone pair of electrons attack the emtpy pi star orbital of the C=C
The pair of electrons in the pi bond are offloaded into the empty p orbital of the carbene
Ring closure
CONCERTED PROCESS- Make and break bonds simultaneously
Only Cis product is formed
How is a carbene generated from an alpha-elimination reaction
React a chloroform with NaOH (chloroform = CCl3H)
OH- removes H to generate a carbanion
negative charge puts an electron pair into the empty chlorine p orbital
Forms a singlet dichlorocarbene
lone pair of electrons on the Cl form a stabilising bond interaction with the empty C p orbital
What is the product when an electron deficient carbene reacts with an electron rich alkene
formed strained three membered rings
attack of alkene electrons into the empty p orbital if the dichlorocarbene
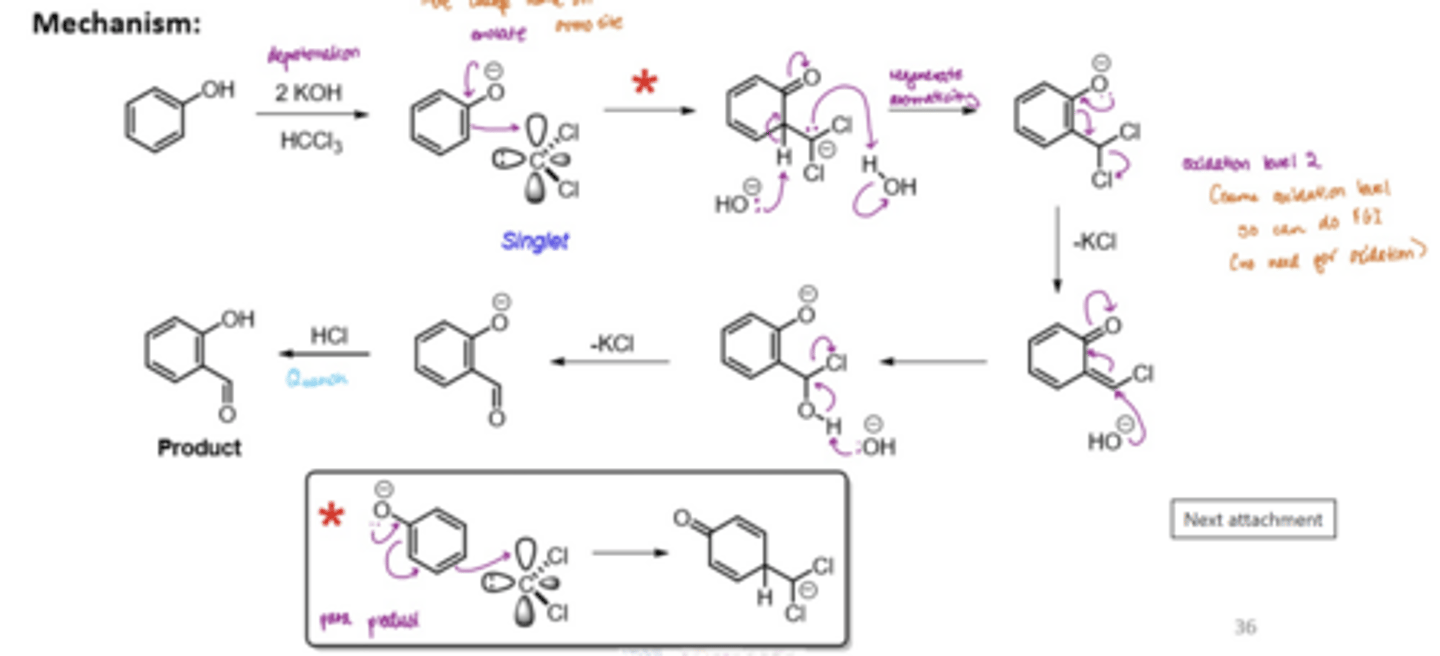
What is the Reimer-Tieman reaction
The alpha-formylation of a phenol
Deprotonate a phenol to form an enolate
React with a singlet dichlorocarbene
deprotonate this structure to regenerate aromaticity and quench the negative charge of the C with water
lone pair of electrons on the O come back into form C=O, resulting in Cl being kicked out, to form an alkene branch
React with OH to eliminate the alkene and kick out the remaining Cl
Quench the product with HCl
Can get an ortho or para product
How do carbenes undergo a 1,2-shift
step wise process
lone pair has to be 90 degreed to the p orbital so rotation has to occur
What occurs during the Wolff rearrangement
allows a diazoketone to be transformed into a ketene
trapped by water or alcohol to form the corresponding carboxylic acid or ester
diazoketone has a resonance form, which includes a Nitrogen triple bond and a C-O (-)
Under hv, C=O reforms and N2 is kicked out to form a carbene
1,2 shift occurs in a step wise process to form a ketene
If this ketene reacts with a nucleophile (water, amines, alcohols), the corresponding carboxylic acid will form
What happens in the Wolff rearrangement if the diazoketone is cyclic
Results in a ring contraction e.g. 6 membered ring to a 5 membered ring
Same as before but the 1,2-alkyl shift is part of a ring system, which results in ring contraction
What is the Arndt-Eister synthesis
Used to prepare beta-amino acids from the more readily available equivalent alpha- amino acids
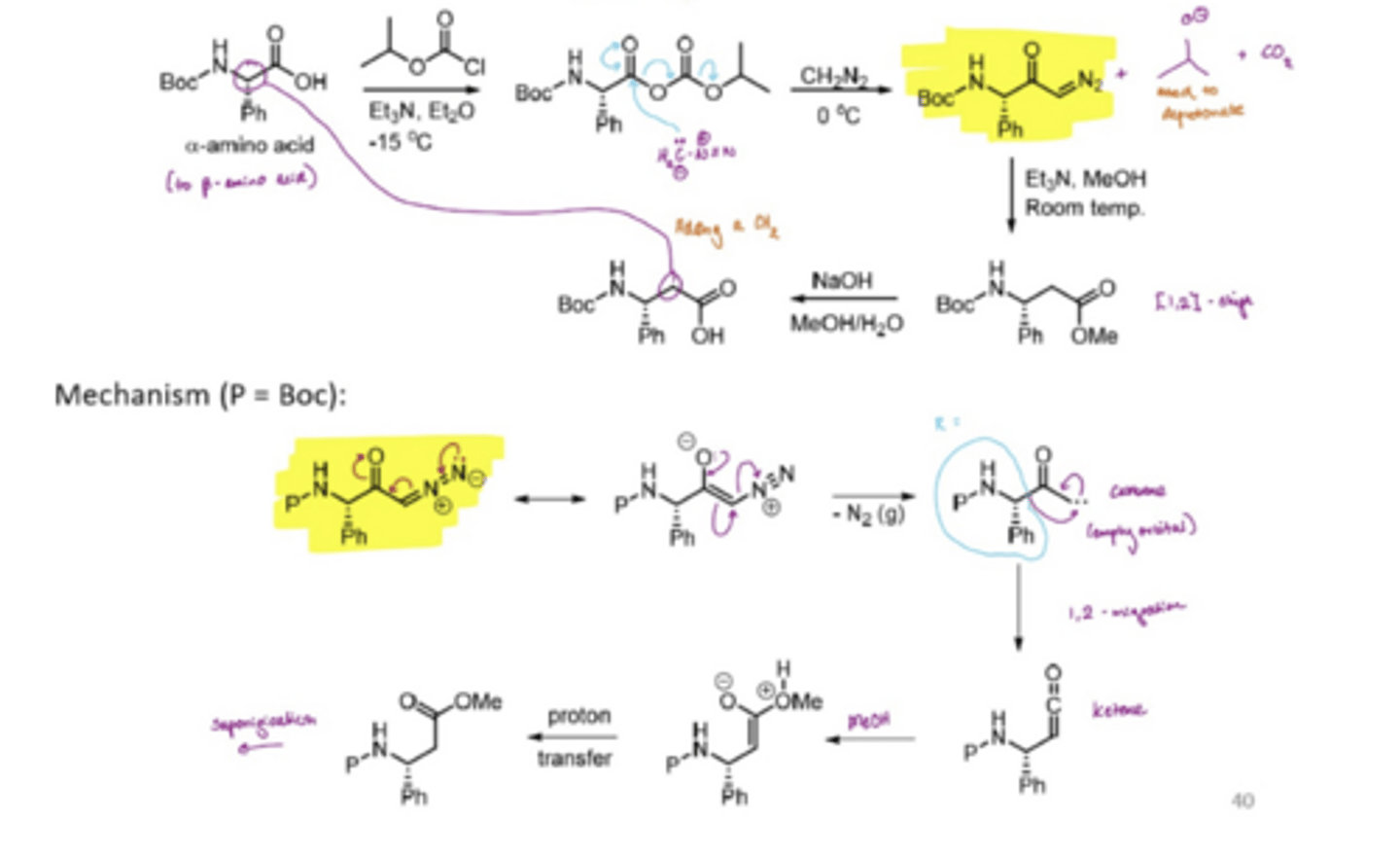
What is the Corey-Fuchs reaction
converts and aldehyde into an alkyne
1) formation of the ylid
React Ph3P with CBr4
React this ylid with an aldehyde
P(+) and O(-) react to form a cyclic structure with a P-O
Ring collapses due to the thermodynamic driving force of forming a P=O to form an alkene
react this alkene with Bu-Li,
Li substitutes a Br
Remaining Br is kicked off
Alpha-elimination produces a carbene
a 1,2-shift of the carbene results in the formation of an alkyne
What is the Seyferth-Gilbert homologation reaction
allows the direct transformation of an aldehyde into the corresponding alkyne
aldehydes form a mono-substituted alkyne
ketones form a disubstituted alkyne
MeOH attacks the C=O of the Ohira-Bestmann reagent, forming a good LG, so is then kicked out
The negative charge attacks an aldehyde at the C=O, forming a O(-)
O(-) attacks P to form a ring, which then collapses to form a P=O
the linear carbon nitrile undergoes alpha-elimination to form a carbene
carbene undergoes a 1,2 shift to form an alkyne
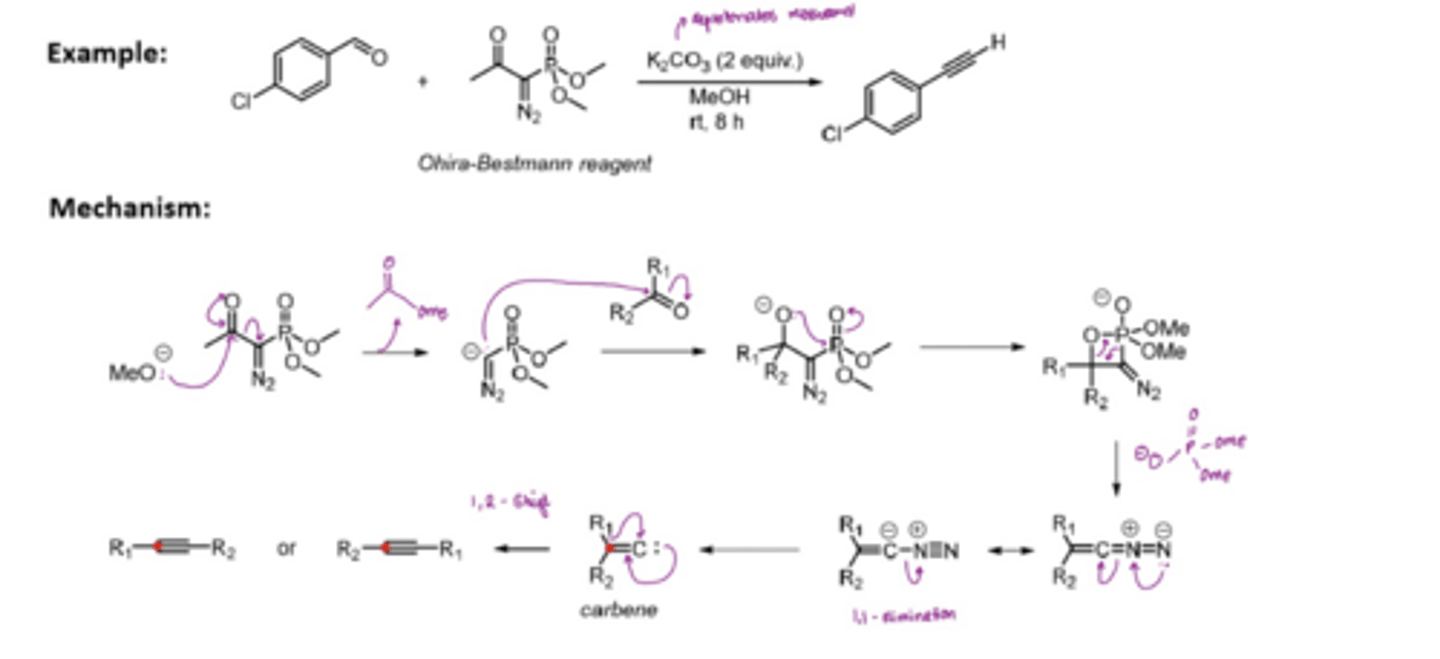
What are nitrenes
analogues if carbenes
nitrogen has 6 valence electrons
2 electrons involved in bonding
4 non-bonding electrons
can be a singlet (sp2 hybridised- electrons paired in 2 orbitals)
can be a triplet (sp hybridised- one electron in one orbital, one electron in another orbital, 2 electrons in another orbital)
Which state of nitrene is lower in energy
the triplet state is much lower in energy
what are the 4 methods of preparing nitrenes
1) elimination from azides (forms a triplet)
2) 1,1- elimination (forms a triplet nitrene)
3) via oxidation of a disubstituted hydrazine (forms a singlet since second N can stabilise the structure into an empty p orbital)
4) Reduction of nitro compounds (compounds with a N attached to an aromatic ring) (forms a singlet)
Which systems can nitrenes be added to
alkenes
aromatic systems (aromatic ring undergoes ring expansion in azepine synthesis)
what is the difference between triplet and singlet nitrene insertion into C-H bonds
singlet insertion involves a concerted process to form a product which retains the configuration (diagnostic of a singlet nitrene)
triplet insertion involves a radical reaction and forms a racemic product
what does the curtius rearrangement involve
converts an acyl chloride into a primary amine using an insertion into a C-C bond
N3 attacks the C=O of the acyl chloride, and kicks out the Cl to form an acyl azide
acyl azide can either undergo 1,1-elimination and proceed via nitrene (photochemical) or undergo a concerted process (thermal) to form an isocyanate
isocyanate undergoes decarboxylation to form a primary amine
What does the Hoffman rearrangement involve
abstracting CO2 from an amide to make an amine
remove an H from the N of the amide with OH-
Add a Br instead
Use OH- to take off the remaining H
next step can either be a concerted process or proceed via the nitrene intermediate
multi-step reaction to get an amine
What is Bredt's rule
Double bonds do not form to bridgehead carbons since too much strain is introduced into the system
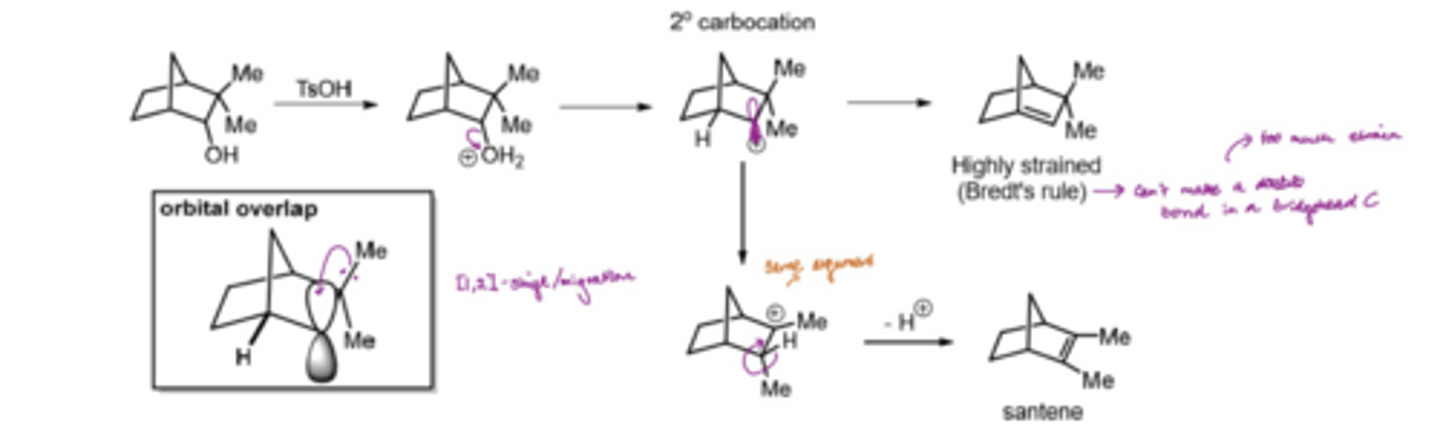
If there is inversion in stereochemistry in a substitution what can we assume
substitution by an SN2 process
If there is retention in stereochemistry in a substitution what can we assume
neighbouring group participation (retention via a double inversion process)
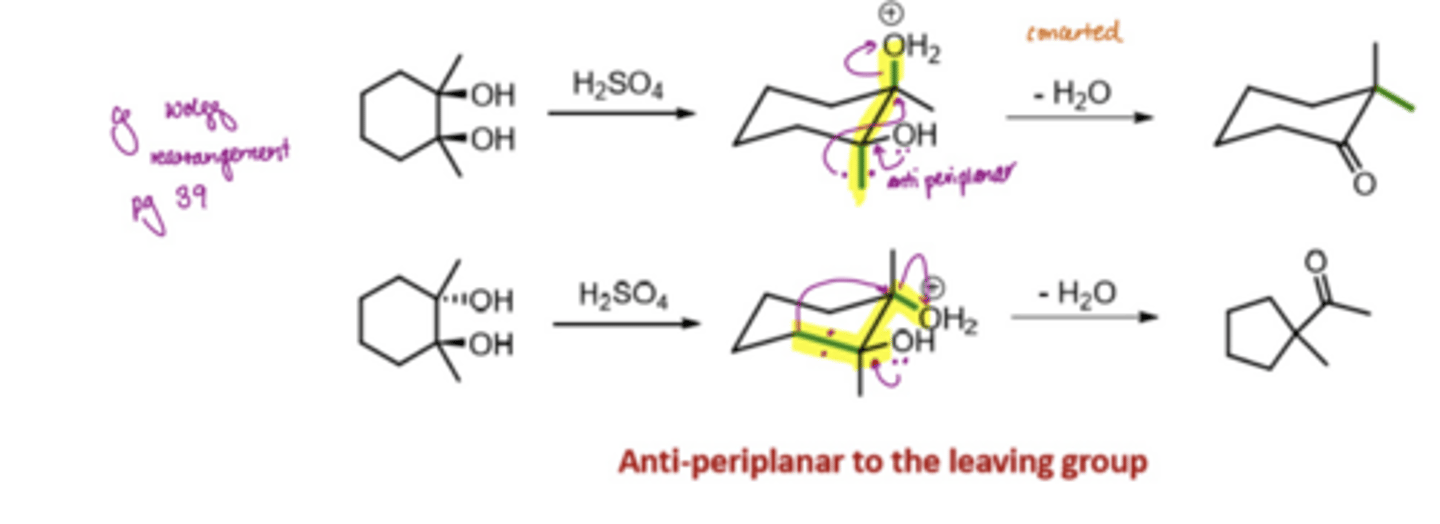
What does the pinacol rearrangement involve
forms 1,2-diols but can then undergo subsequent rearrangement of these species to form substituted ketone products
What is the orbital overlap evidence for the pinacol rearrangement
adopt the chair conformation in the intermediate state
has to be antiperiplanar to the LG
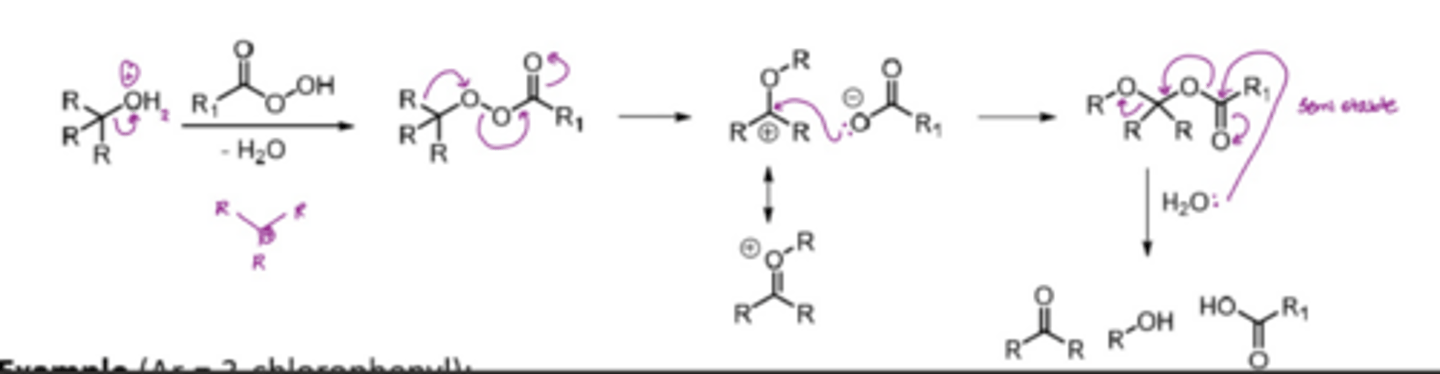
What does the semi-pinacol rearrangement involve
After selective activation of one of the hydroxyl groups, several LGs are possible (halogens, tosylates, mesylates and phosphonates)
e.g. react a dihydroxy with TsCl and pyridine to make a molecule with an OTS LG
Look for the anti-periplanar relationship between the bond and the LG
Lone pair on remaining OH comes down to form a C=O
Bonding pair from bond anti-periplanar to the OTS attacks the Carbon attached to the LG, and eliminates the LG
*anything that can be activated to generate a carbocation can be used
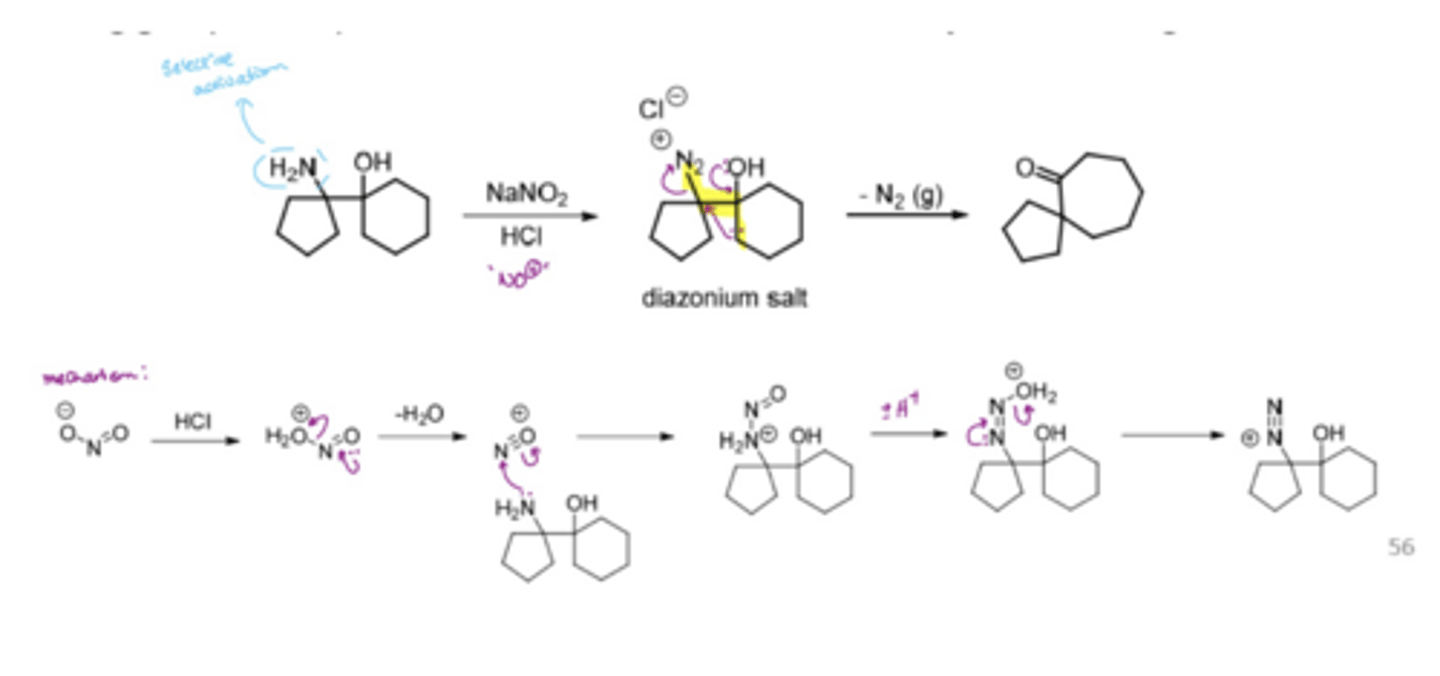
What does the Tiffeneau-Demjanoc rearrangement involve
Activates primary amines to make diazonium salt LG
selective activation of the amine group by NO2
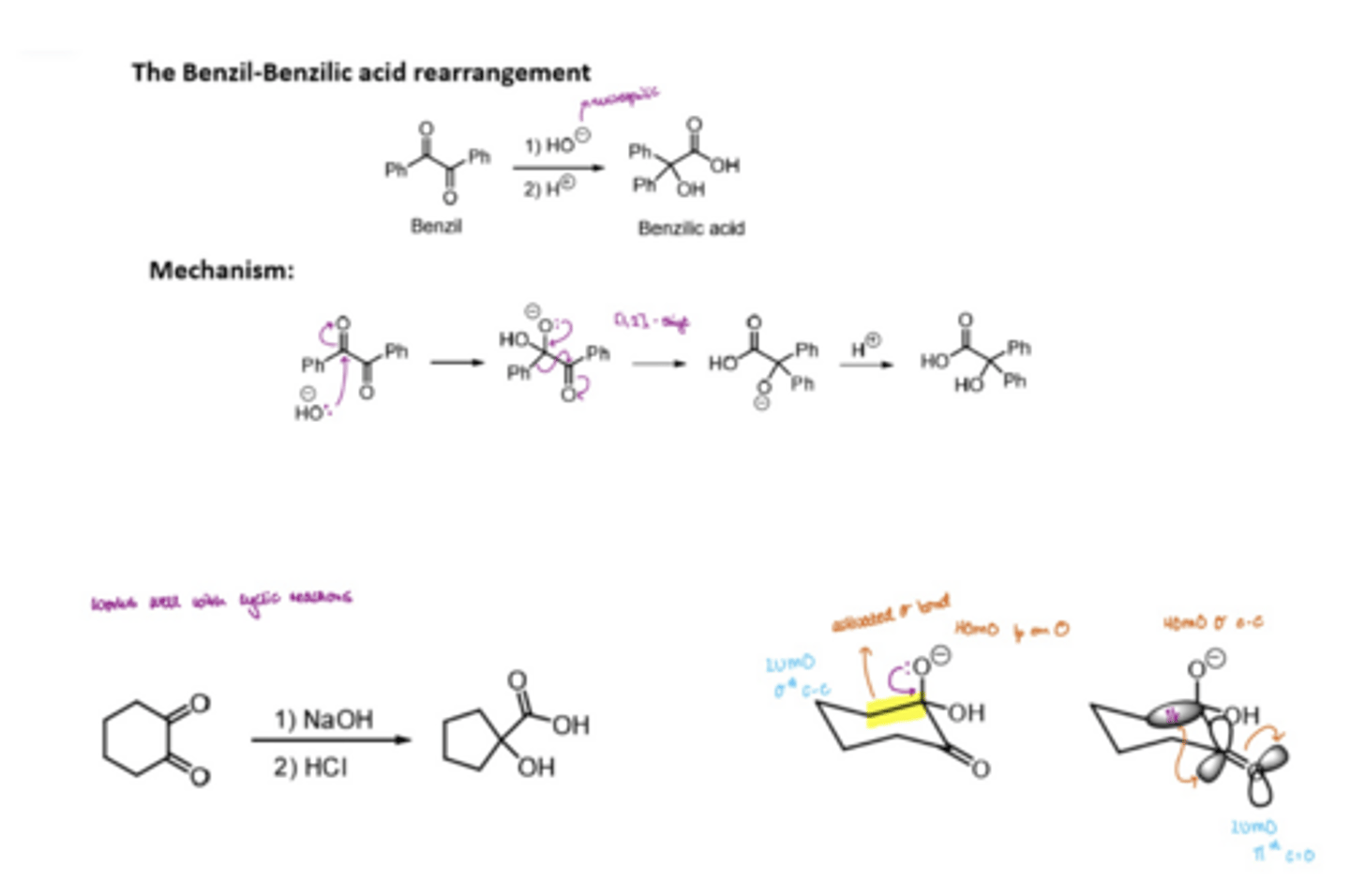
What does the benzil-benzilic rearrangement involve
activating a benzil molecule with OH-
1,2-shift of the phenyl group
Addition of H+ to the O- to form benzilic acid
What happens when a 1,2-diketone undergo a reaction in the presence of a strong base
forms alpha-hydroxycarboxylic acids
INSERT PIC
What does the Favorskii rearrangement involve
a base induced rearrangement
commenly a ketone reacts via a reactive syclic propanone intermediate to form an ester product
abstract beta hydrogen from the ketone molecule with EtO- to form a double bond and a C-O (-)
C=O reforms, double bond breaks, LG is kicked out
forms a strained carbonyl ring system
EtO- attacks the carbonyl to form C-O(-)
C=O reforms to and ring breaks to form the most stable anion (can be stabilised through resonance)
React with EtOH to form an ester
In done in a cyclic system, there is ring contraction (ring gets smaller)
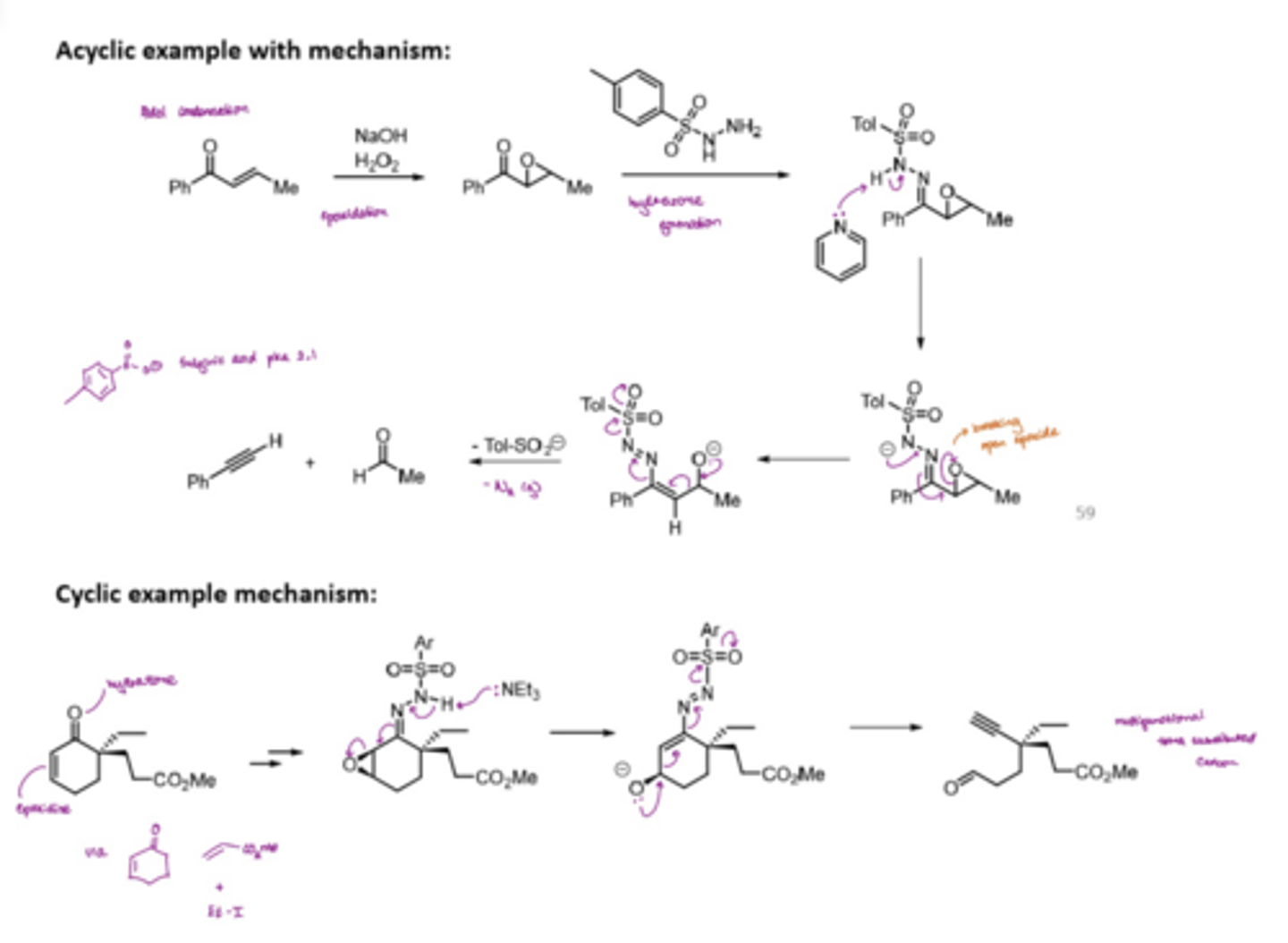
What does the Eschenmoser fragmentation involve
enables the transformation of an alpha,beta-epoxyketone to an alkyne and a carbonyl unit through the action of an aryl sulfonylhydrazines
involves the formation of an epoxide ring with NaOH and H2O2
epoxide ring forms a hydrazone
epoxide ring is broken open
end products = alkyne + carbonyl unit
What does the Prins reaction involve
nucleophilic attack of an alkene onto an activated carbonyl (normally an aldehyde)
if there is a hydroxy group on the alkene, the OH is a better nucleophile so attacks the carbonyl unit
activate the carbonyl through a proton source or a lewis acid
3 products can be generated:
Allylic system
1,3-diols
acetals
How is an allylic system formed in the prins reaction
through an intramolecular process
includes proton shuffling within the molecule
an SN1 type elimination
finally, the prins step which involves the alkene attack onto the activated carbonyl but they are part of the same structure
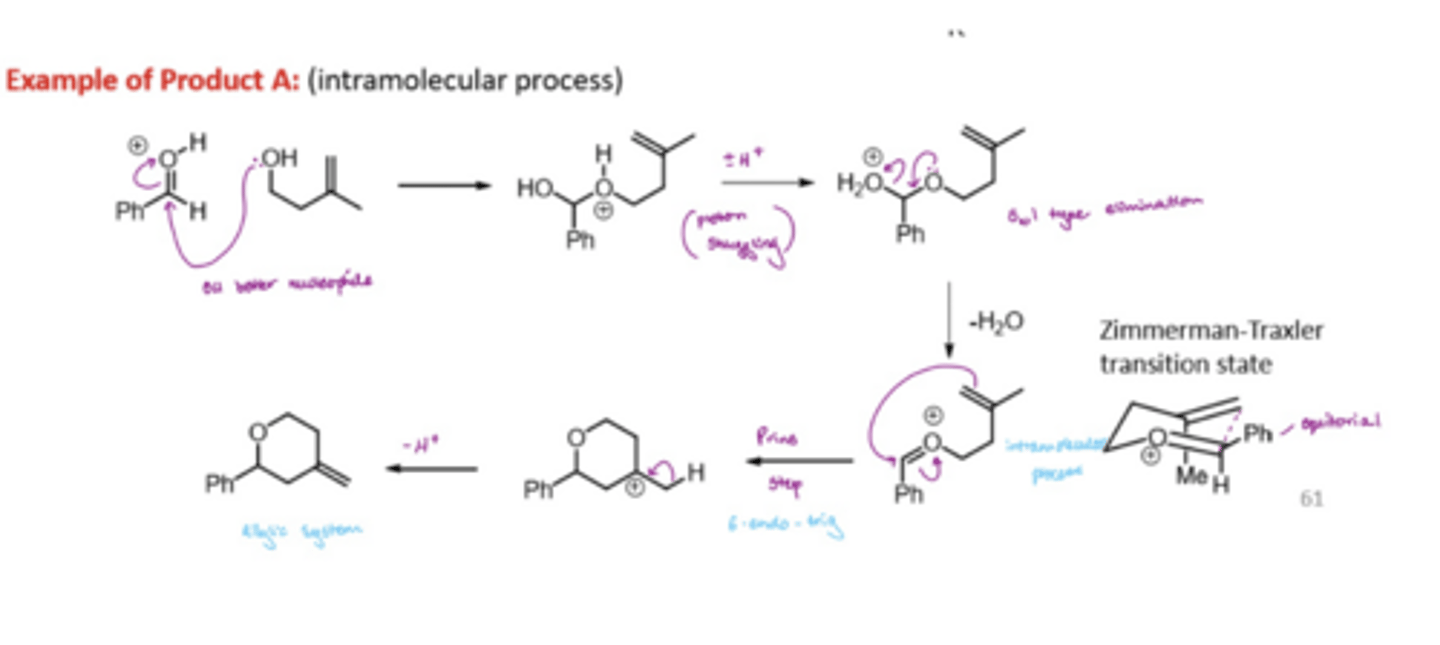
How is a 1,3-diol formed in the prins reaction
forms an oxonium species which undergoes the prins reaction
the attack of water from the least sterically hindered face
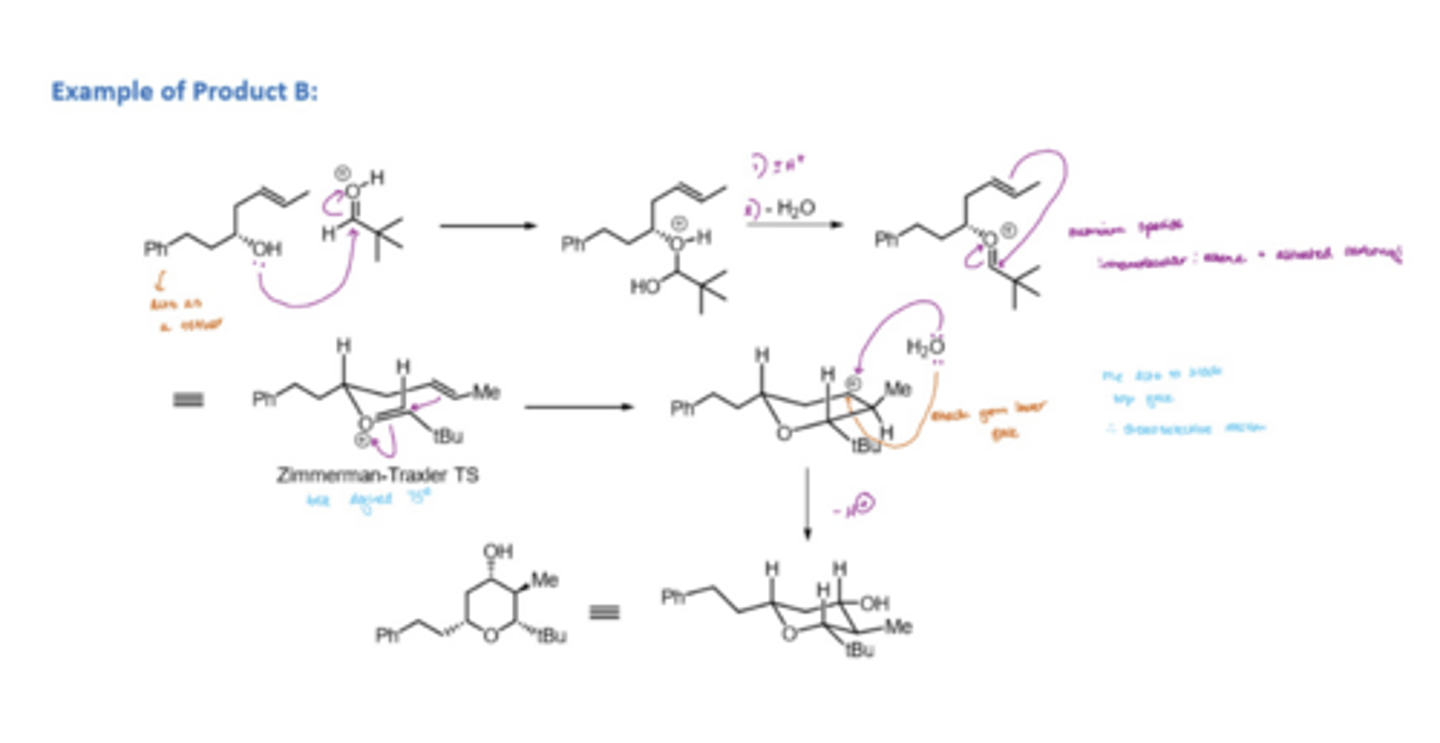
How is an acetal formed in the prins reaction
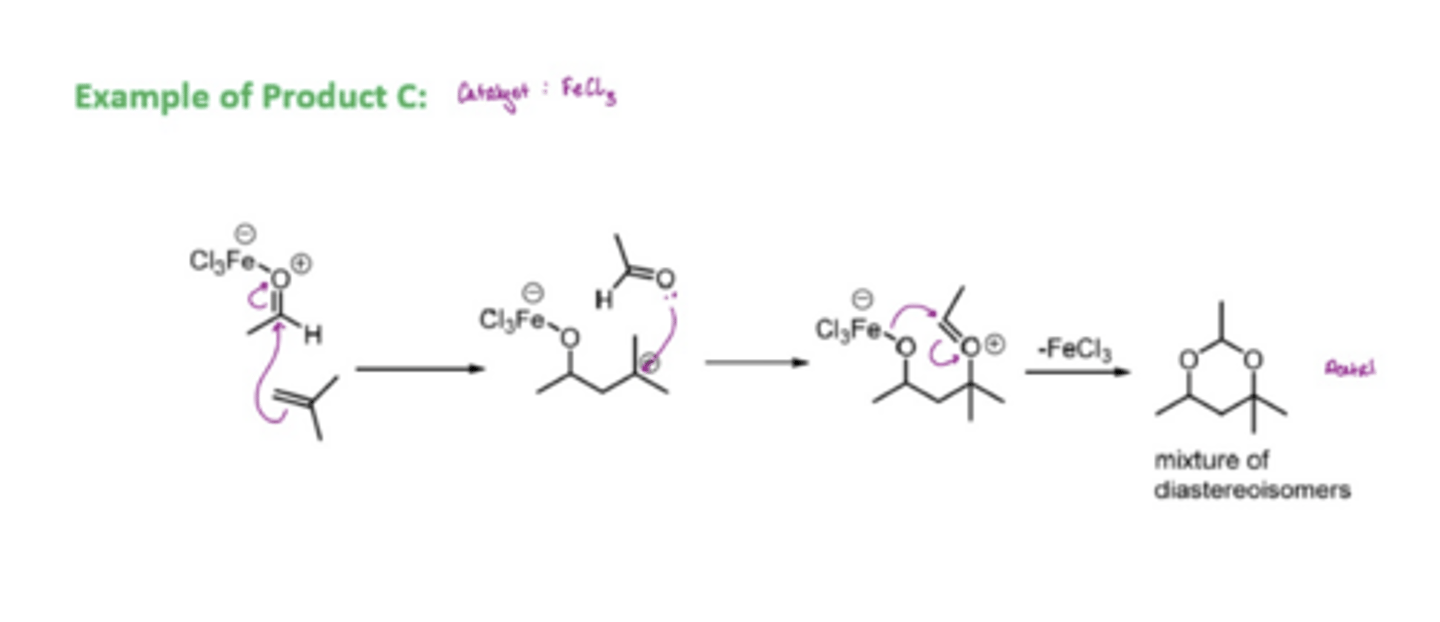
an FeCl3 catalyst is used
forms a mixture of diastereoisomers of acetal molecules
What does the Bayer-Villiger Rearrangement involve
uses a peroxide to convert a ketone to an ester (cyclic ketones are converted into lactones)
protonate the carboxyl group
react with a carboxylic acid
do some proton shuffling to set up an antiperiplanar relationship between the bond and LG
this migration step with the antiperiplanar relationship retains the stereochemistry
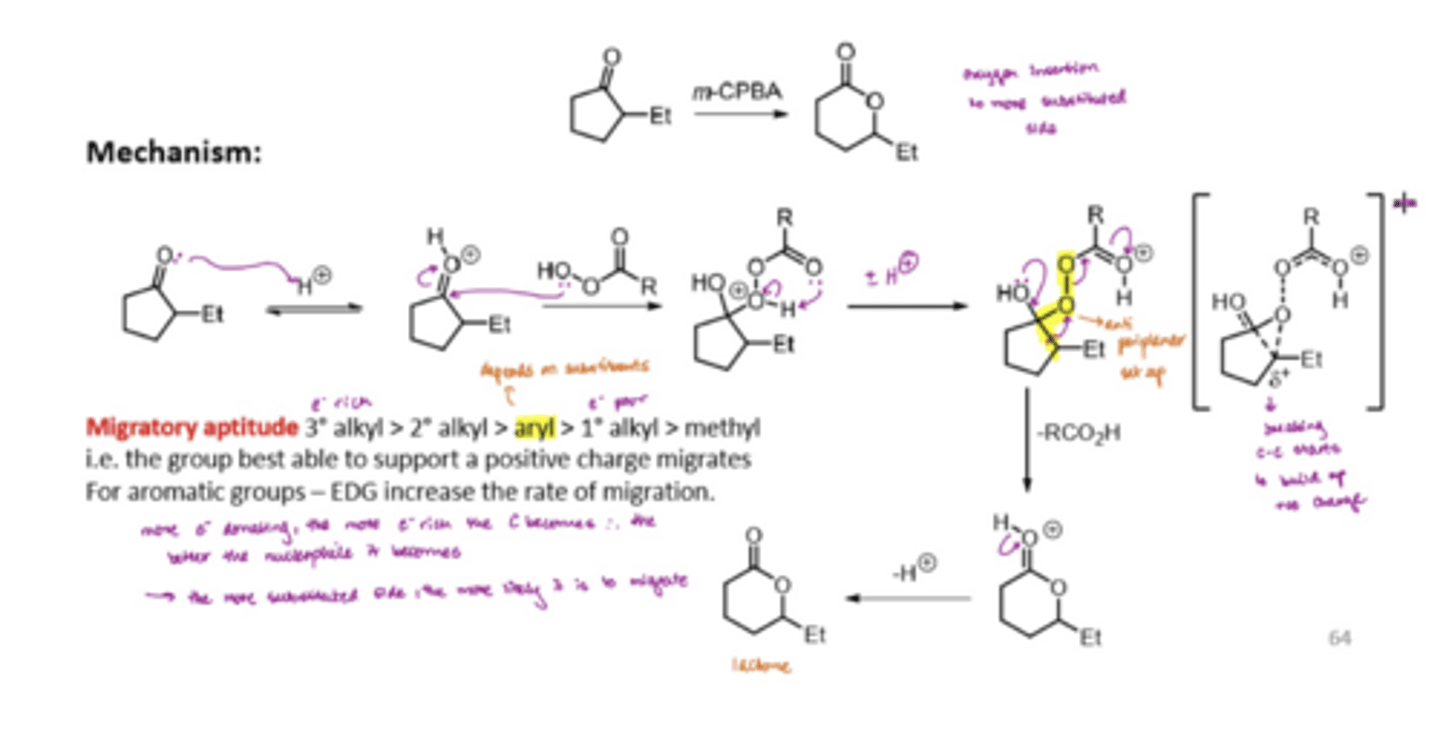
What is migratory aptitude
the group best able to support positive charge migrates for aromatic groups
The more electron donating, the more electron rich the carbon becomes, the better nucleophile it becomes
The more substituted side is more likely to migrate
tertiary alkyl > secondary > aryl > primary > methyl
What does the Criegee rearrangement involve
a tertiary alcohol is cleaved by oxidation of a peroxuacid to form a ketone
similar mechanism to Bayer-Villiger Rearrangement
What does the Beckmann rearrangement involve
the conversion of an oxime to a secondary amide
React an aldehyde with H2N-OH and H+ to get an oxime
react oxime with H2SO4 to establish bond migration-LG periplanar relationship
ring opening and add one more side to the ring e.g. 6 membered ring turns into a 7 membered ring
Add H2O to quench the positive charge to get a molecule which can undergo tautomerisation to produce a secondary amide
What does the Beckmann fragmentation involve
ring opening
react a hydroxy ketone with HONH2 to convert the C=O into C=N-OH
react with acyl chloride to set up antiperiplanar relationship
generate HCl, CO2, CO and the ring opened structure (entropy and enthalpy are favourable)
What does the Schmidt reaction involve
forms an amide
react ketone with NH4 to get a C=O=H bond
React with N3 to break C=O
Eliminate water through the attack of the N lp
activated complex with an antiperiplanar relationship
lose N2
Generate a ring with one more side than what you started with
Add water to quench positive charge
-H(+)
tautomerise to get an amide
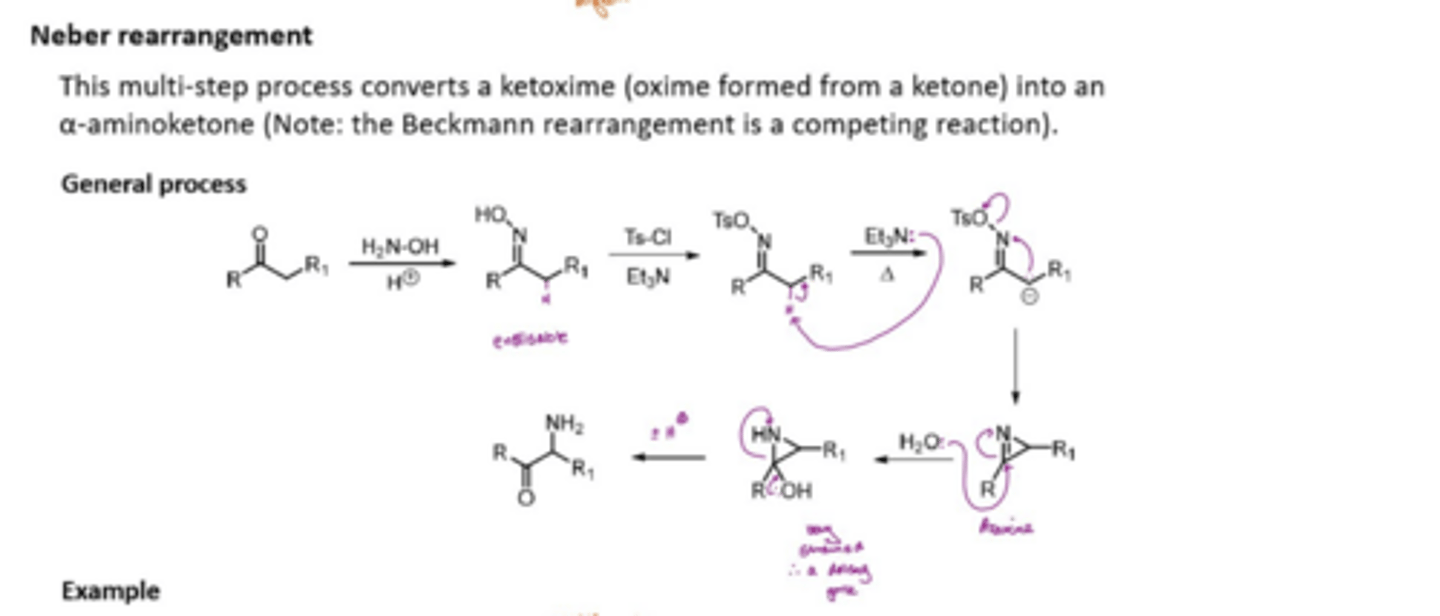
What does the Neber rearrangement involve
convertes a ketoxime (oxime formed from a ketone) into an alpha-aminoketone
Beckmann rearrangement is a competing reaction
React a ketone with H2N-OH to get an enolisable compound
react with TsCl and Et3N to knock off the OH with TsO ( a good LG)
deprotonate alpha carbon with Et3N to form an anion
Get the electron on the negative charge to attack the N and kick out TsO, forming an azarine (cyclic)
Add H2O to break C=N, this forms a very strained ring
lp on oxygen forms C=O and breaks the C-N bond which is holding the ring together
ring is broken to form an alpha-aminoketone