Exam 4 Biochem Book Problems
1/219
Earn XP
Description and Tags
UARK Biochemistry a Short Course: Chapter 20-28
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
220 Terms
Distinguish between an oxidizing agent and a reducing agent
An oxidizing agent, or oxidant, accepts electrons in oxidation-reduction reactions. A reducing reagent, or reductant, donates electrons in such reactions.
What is Respiration
An ATP-generating process in which an inorganic compound serves as the final electron acceptor
What is Redox potential
Measure of the tendency to accept or donate electrons
What does the Electron-transport chain decribe
Electron flow from NADH and FADH2 to O2
What does Flavin mononucleotide (FMN) do?
Accepts electrons from NADH in Complex I
What does Iron-sulfur protein do?
Facilitates electron flow from FMN to coenzyme Q
What is Coenzyme Q?
Lipid-soluble electron carrier
What does Cytochrome c do?
Donates electrons to Complex IV
What does Q cycle do?
Funnels electrons from a two-electron carrier to a one-electron carrier
What does Superoxide dismutase do?
Converts reactive oxygen species into hydrogen peroxide
What does Catalase do?
Converts hydrogen peroxide into oxygen
The standard oxidation–reduction potential for the reduction of O2 to H2O is given as 0.82 V. However, the value given in textbooks of chemistry is 1.23 V. Account for this difference.
The 1.23 V value is standard for electrochemical chemistry, at pH =0 (O2 + 4H + 4e- → 2H2O), while 0.82 V is the physiological standard in cells (standard reduction potential at pH = 7)
What is the evidence that modern mitochondria arose from a single endosymbiotic event?
-supported by the uniform, maternal inheritance of mitochondria across all eukaryotes
-universal reliance on a specific protein, the ATP/ADP translocase
Identify the oxidant and the reductant in the following reaction: Pyruvate + NADH + H+ → lactate + NAD+
Pyruvate accepts electrons and is thus the oxidant.
NADH gives up electrons and is the reductant.
How is the redox potential (ΔE′°) related to the free-energy change of a reaction (ΔG°′)?
the free-energy change is directly proportional to the change in reduction potential (ΔG°’ = -nFΔE’°)
Explain why coenzyme Q is an effective mobile electron carrier in the electron-transport chain
because it is hydrophobic, enabling free diffusion within the inner mitochondrial membrane, and it can reversibly transfer both electrons and protons.
-Its structure allows it to shuttle electrons from complexes I and II to complex III
Place the following components of the electron-transport chain in their proper order: (a) cytochrome c; (b) Q-cytochrome c oxidoreductase; (c) NADH-Q oxidoreductase; (d) cytochrome c oxidase; (e) ubiquinone.
c, e, b, a, d
Match: Complex I
NADH-Q oxidoreductase
Match: Complex II
Succinate-Q reductase
Match: Complex III
Q-cytochrome c oxidoreductase
Match: Complex IV
Cytochrome c oxidase
Match: Ubiquinone (Q)
Coenzyme Q
What are the reactive oxygen species, and why are they especially dangerous to cells?
Hydroxyl radical (OH ·), hydrogen peroxide (H2O2), superoxide ion (O2∸), and peroxide (O22−). These small molecules react with a host of macromolecules (proteins, nucleotides, and membranes) to disrupt cell structure and function.
Rotenone inhibits electron flow through NADH-Q oxidoreductase. Antimycin A blocks electron flow between cytochromes b and c1. Cyanide blocks electron flow through cytochrome oxidase to O2.
Predict the relative oxidation–reduction state of each of the following respiratory-chain components in mitochondria that are treated with each of the inhibitors: NAD+; NADH-Q oxidoreductase; coenzyme Q; cytochrome c1; cytochrome c; cytochrome a.
Rotenone: NADH, NADH-Q oxidoreductase will be reduced. The remainder will be oxidized.
Antimycin A: NADH, NADH-Q oxidoreductase and coenzyme Q will be reduced. The remainder will be oxidized.
Cyanide: All will be reduced.
What is the advantage of having Complexes I, III, and IV associated with one another in the form of a respirasome?
The respirasome is another example of the use of supramolecular complexes in biochemistry. Having the three complexes that are proton pumps associated with one another will enhance the efficiency of electron flow from complex to complex, which in turn will cause more efficient proton pumping.
The cytochrome b component of Q‑cytochrome c oxidoreductase enables both electrons of QH2 to be effectively utilized in generating a proton-motive force. Cite another recycling device in metabolism that brings a potentially dead-end reaction product back into the mainstream.
Triose phosphate isomerase converts dihydroxyacetone phosphate (a potential dead end) into glyceraldehyde 3-phosphate (a mainstream glycolytic intermediate).
Exercise is known to increase insulin sensitivity and to ameliorate type 2 diabetes. Recent research suggests that taking antioxidant vitamins might mitigate the beneficial effects of exercise with respect to ROS protection. (a) What are antioxidant vitamins? (b) How does exercise protect against ROS? (c) Explain why vitamins might counteract the effects of exercise
(a) Vitamins C and E
(b) Exercise induces superoxide dismutase, which converts ROS into hydrogen peroxide and oxygen.
(c) 1) the suppression of ROS by vitamins prevents the expression of more superoxide dismutase. 2) some ROS may be signal molecules required to stimulate the biochemical benefits of exercise.
What citric acid cycle enzyme is also a component of the electron-transport chain?
Succinate dehydrogenase is a component of Complex II.
Compare fermentation and respiration with respect to electron donors and electron acceptors
In fermentations, organic compounds are both the donors and the acceptors of electrons.
In respiration, the electron donor is usually an organic compound, whereas the electron acceptor is an inorganic molecule, such as oxygen.
Human beings have only about 250 g of ATP, but even a couch potato needs about 83 kg of ATP to open that bag of chips and use the remote. How is this discrepancy between requirements and resources reconciled?
The ATP is recycled by ATP generating processes, most notably oxidative phosphorylation.
Match: ATP synthase
converts the proton motive force to ATP
Match: Proton Motive Force
composed of a chemical gradient and a charge gradient
Match: Electron Transport Chain
generates the proton gradient
Match: Glycerol 3- Phosphate shuttle
cytoplasmic NADH to mitochondrial FADH2
Match: malate aspartate shuttle
cytoplasmic NADH to mitochondrial NADH
Match: respiratory (acceptor) control
ADP controls the rate of respiration
Match: uncoupling protein
results in heat instead of ATP
Match: F1 subunit
catalytic subunit
Match: F0 subunit
proton channel
Match: c ring
a proton merry go round
What is the yield of ATP when each of the following substrates is completely oxidized to CO2 by a mammalian cell homogenate? Assume that glycolysis, the citric acid cycle, and oxidative phosphorylation are fully active.
(a) Pyruvate (b) Lactate (c) Fructose 1,6-bisphosphate (d) Phosphoenolpyruvate (e) Galactose (f) Dihydroxyacetone phosphate
a. 12.5
b. 14
c. 32
d. 13.5
e. 30
f. 16
What is the effect of each of the following inhibitors on electron transport and ATP formation by the respiratory chain? (a) Azide (b) Atractyloside (c) Rotenone (d) DNP (e) Carbon monoxide (f) Antimycin A
a. Azide blocks electron transport and proton pumping at Complex IV.
b. Atractyloside blocks electron transport and ATP synthesis by inhibiting the exchange of ATP and ADP across the inner mitochondrial membrane space.
c. Rotonene blocks electron transport and pumping at complex 1
d. DNP blocks up ATP synthesis without inhibiting ETC by dissipating the electron gradient
e. carbon monoxide blocks ET and proton pumping at complex IV
f. Antimycin A blocks ET and proton pumping at complex 3
What is the mechanistic basis for the observation that the inhibitors of ATP synthase also lead to an inhibition of the electron-transport chain?
If the proton gradient is not dissipated by the influx of protons into a mitochondrion with the generation of ATP, the outside of the mitochondrion eventually develops such a large positive charge that the ETC can no longer pump protons against the gradient.
Years ago, it was suggested that uncouplers would make wonderful diet drugs. Explain why this idea was proposed and why it was rejected. Why might the producers of antiperspirants be supportive of the idea?
If oxidative phosphorylation were coupled, no ATP could be produced. Too much uncoupling will lead to tissue damage.
If actively respiring mitochondria are exposed to an inhibitor of ATP synthase, the electron transport chain ceases to operate. Why?
because the two processes are tightly coupled through the proton gradient
What is the actual role of protons in the synthesis of ATP by F0F1 ATP synthase?
The proton gradient is necessary for ATP synthesis because proton flow through the enzyme causes conformational changes that convert a T subunit to an O subunit with the subsequent release of ATP. The role of the proton gradient is not to make ATP, but to release it from the synthase.
It has been noted that the mitochondria of muscle cells often have more cristae than the mitochondria of liver cells. Provide an explanation for this observation.
There is a greater need for ATP during exercise. This increase in requirement allows there to be more sites of phosphorylation and these sites can exist with the increase in cristae.
If actively respiring mitochondria are exposed to an inhibitor of ATP-ADP translocase, the electron-transport chain ceases to operate. Why?
it halts the supply of ADP required for ATP synthase
Why must ATP-ADP translocase use Mg2+-free forms of ATP and ADP?
because the transporter's binding site is designed to recognize and distinguish between the net negative charges of the uncomplexed molecules
What causes the c subunits of ATP synthase to rotate? What determines the direction of rotation?
-proton motive force
-a high concentration of protons (H+) in the intermembrane space flowing to a lower concentration in the mitochondrial matrix.
Give an example of the use of the protonmotive force in ways other than for the synthesis of ATP.
ATP export from the matrix. Phosphate import into the matrix.
How does the inhibition of ATP-ADP translocase affect the citric acid cycle? Glycolysis?
-inhibits the Citric Acid (TCA) cycle by halting the transport of ADP into the mitochondrial matrix and ATP out→ causes mitochondrial NADH buildup.
-glycolysis accelerates to compensate for reduced ATP production via anaerobic pathway
Under some conditions, mitochondrial ATP synthase has been observed to run in reverse. How would that situation affect the proton-motive force?
When mitochondrial ATP synthase runs in reverse (as an ATPase), it hydrolyzes ATP to pump protons (H+) from the matrix into the intermembrane space → allowing the enzyme to maintain/ reinforce the proton-motive force.
Describe the evidence supporting the chemiosmotic hypothesis.
supported by evidence that electron transport creates a proton gradient across membranes, which drives ATP synthesis via ATP synthase
Predict the effect on ATP synthesis if the b and d subunits of the ATPase were absent.
causes significant impairment or total cessation of ATP synthesis.
-These subunits constitute the stator stalk (peripheral stalk), which is essential to hold the α3β3 hexamer stable, preventing it from rotating along with the central stalk. Without this stabilization, the enzyme cannot couple the proton motive force to mechanical rotation for catalytic activity
Why will isolated F1 subunits display ATPase activity but not ATP synthase activity? How can the enzyme then function as ATP synthase in mitochondria?
isolated: hydrolysis of ATP is exergonic and therefore favorable (so it will do the reverse rxn)
mitochondria: the power of the proton gradient will overcome the previous favorability of ATP hydrolysis
What does the fact that rotenone increases the susceptibility to Parkinson disease indicate about the etiology of this disease?
suggest that environmental toxins, specifically mitochondrial complex I inhibitors, can trigger PD pathogenesis through oxidative stress
Some cytoplasmic kinases (enzymes that phosphorylate substrates at the expense of ATP) bind to voltage-dependent anion channels. What might the advantage of this binding be?
the advantage is securing preferential access to ATP exported directly from the mitochondria.
Mice that are completely lacking ATP-ADP translocase (ANT−/ANT−) can be made by the knockout technique. Remarkably, these mice are viable but have the following pathological conditions: (1) high serum levels of lactate, alanine, and succinate; (2) little electron transport; and (3) a 6- to 8-fold increase in the level of mitochondrial H2O2 compared with that in normal mice. Provide a possible biochemical explanation for each of these conditions.
-Organic acids --> mice are deriving energy needs through anaerobic glycolysis (Lactate, alanine are products of anaerobic glycolysis). Alanine formation plays a role in succinate formation.
-The e- transport chain is slowed b/c the inner mitochondrial membrane is hyperpolarized. Without ADP to accept the energy of the PMF the membrane becomes polarized to such an extent that protons can no longer be pumped.
-Excess H2O2 is probably due to the fact that the superoxide radical is present in higher concentration b/c oxygen can no longer be effectively reduced.
The most common metabolic sign of mitochondrial disorders is lactic acidosis. Why?
because defective mitochondria cannot efficiently use oxygen to generate ATP (energy), forcing cells to rely on anaerobic glycolysis
Human beings do not produce energy by photosynthesis, yet this process is critical to our survival. Explain.
because it produces the oxygen necessary for respiration and serves as the foundation of the food chain, supplying all dietary energy
What is the overall reaction of photosynthesis?
6CO2 + 12H2O → C6H12O6 + 6H2O + 6O2
Photosynthesis can be measured by measuring the rate of oxygen production. When plants are exposed to light of wavelength 680 nm, more oxygen is evolved than if the plants are exposed to light of 700 nm. Explain
680 nm light produces more oxygen because it directly drives Photosystem II (PSII). PSII’s reaction center (P680) absorbs light maximally at 680 nm.
Light at 700 nm primarily drives Photosystem I (PSI), which is less efficient, leading to lower oxygen output.
If plants are illuminated by a combination of light of 680 nm and 700 nm, the oxygen production exceeds that of either wavelength alone. Explain.
exhibit a synergistic increase in photosynthesis known as the Emerson enhancement effect. PSII and PSI operate in series to drive the electron transport chain
What is the advantage of having an extensive set of thylakoid membranes in the chloroplasts?
maximizes the surface area available for light-dependent reactions, increasing the number of sites for chlorophyll, photosystems, and electron transport chains
What is the significance of photoinduced separation of charge in photosynthesis?
foundational step in photosynthesis that converts light energy into chemical energy. By moving an electron from a chlorophyll pigment to an acceptor, it creates a long-lived charge-separated state.
Explain how light-harvesting complexes enhance the efficiency of photosynthesis.
by acting as antenna systems that maximize photon absorption and rapidly transfer energy to reaction centers with near-unity quantum efficiency
Identify the ultimate electron acceptor and the ultimate electron donor in photosynthesis. What powers the electron flow between the donor and the acceptor?
the ultimate electron donor is H2O, and the ultimate electron acceptor is NADP.
The flow of electrons is powered by light energy (photons) absorbed in the chloroplasts. This light-driven electron transport chain creates NADPH and ATP, which are used to convert CO2 into carbohydrates.
In chloroplasts, a greater pH gradient across the thylakoid membrane (3 pH units) is required to power the synthesis of ATP than is required across the mitochondrial inner membrane (1 pH unit). Why?
This is because chloroplasts lack a substantial membrane potential to drive the process, requiring the chemical proton gradient to bear the entire energetic load for phosphorylation.
Chlorophyll is a hydrophobic molecule. Why is this property crucial for the function of chlorophyll?
because it allows the molecule to anchor into the hydrophobic thylakoid membrane of chloroplasts, where photosynthesis occurs
Why is chlorophyll an effective light absorbing pigment?
b/c its structure features a conjugated porphyrin ring with a magnesium ion that efficiently absorbs blue and red light photons to initiate photosynthesis. Its structure allows it to transfer excited electrons to photosynthetic machinery.
What are the various sources of protons that contribute to the generation of a proton gradient in chloroplasts?
generated primarily by the accumulation of protons (H+) in the thylakoid lumen through the photolysis of water and the pumping of protons via the plastoquinone cytochrome bf complex
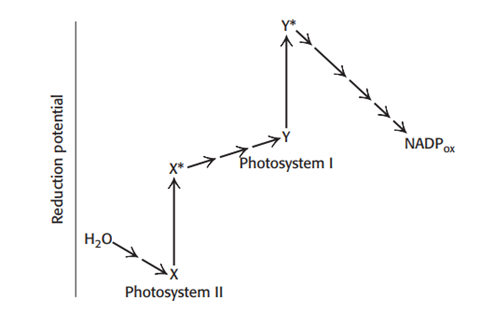
Explain the defect or defects in the hypothetical scheme for the light reactions of photosynthesis depicted here.
The electron flow from photosystem II to photosystem I is uphill/ exergonic. For this uphill flow, ATP would need to be consumed, defeating the purpose of photosynthesis.
(a) It can be argued that, if life were to exist elsewhere in the universe, it would require some process like photosynthesis. Why is this argument reasonable?
(b) If the starship Enterprise were to land on a distant planet and find no measurable oxygen in the atmosphere, could the crew conclude that photosynthesis is not taking place?
-yes, because photosynthesis is a highly efficient, fundamental process for converting abundant stellar light into chemical energy to power life
-No, Photosynthesis might use different reactants or products (non-oxygenic), or produced oxygen could be consumed by other processes
Predict the effect of the herbicide dichlorophenyldimethylurea (DCMU) on a plant’s ability to perform cyclic photophosphorylation.
has no effect on a plant's ability to perform cyclic photophosphorylation. DCMU functions by blocking the electron transfer from Photosystem II (PSII) by binding to the plastoquinone-binding site.
-Because cyclic photophosphorylation solely involves Photosystem I (PSI), the process continues unaffected
Suppose you had a suspension of chloroplasts that lacked ADP and Pi. You exposed these chloroplasts to light for a period of time, after which you plunged them into darkness while simultaneously adding ADP and Pi. To what extent, if any, would you expect ATP synthesis to take place?
can expect ATP synthesis to take place for a short period after the chloroplasts are moved to the darkness
What structural feature of mitochondria corresponds to the thylakoid membranes?
the inner mitochondrial membrane.
-thylakoid membrane is a distinct, third membrane system in chloroplasts
Compare and contrast oxidative phosphorylation and photosynthesis.
Oxidative phosphorylation, used in respiration, breaks down organic molecules (like glucose) to produce ATP and water in the mitochondria,
photosynthesis, used by plants, uses light to produce ATP and NADPH in the chloroplast, ultimately creating glucose
In what way is the electron transfer in ferridoxin-NADP+ reductase similar to that of the pyruvate dehydrogenase complex?
b/c both utilize flavin-based redox chemistry to transfer one-electron inputs into a two-electron reduction of NADP+/NAD+
Albert Szent-Györgyi, Nobel Prize–winning biochemist, once said something to this effect: “Life is nothing more than an electron looking for a place to rest.” Explain how this pithy statement applies to photosynthesis and cellular respiration.
highlights that life depends on energy produced by redox reactions, where electrons transfer from high-energy states to stable/resting lower-energy states.
In photosynthesis, light excites electrons, which "rest" in high-energy sugar bonds; in cellular respiration, these electrons move back to low-energy states (oxygen), releasing ATP
Why is the Calvin cycle crucial to the functioning of all life forms?
it’s the primary carbon-fixation process
because it converts inorganic atmospheric carbon dioxide into organic glucose, providing the essential energy source and carbon building blocks for nearly all organisms.
Differentiate between autotrophs and heterotrophs.
Autotrophs are producers that create their own food using sunlight or chemical energy
heterotrophs are consumers that obtain energy by consuming other organisms
Why are the reactions of the Calvin cycle sometimes referred to as the dark reactions? Do they take place only at night, or are they grim, secret reactions?
because they do not directly require light energy to proceed (light independent) they typically happen during the day, using ATP and NADPH generated by light-dependent reaction
The Calvin cycle can be thought of as taking place in three stages. Describe the stages.
1) Carbon Fixation (Rubisco attaches CO2 to RuBP)
2) Reduction (ATP/NADPH convert 3-PGA to G3P sugar)
3) Regeneration (G3P is used to remake RuBP)
match: carbohydrate with alpha-1,4 linkages only.
Amylose (Amylopectin has only some)
Match: 3-Phosphoglycerate is formed after carbon fixation.
C3 plants
Match: Carbon fixation results in oxaloacetate formation
C4 plants
Match: What compound is required for rubisco activity.
Carbamate
Suggest a reason why rubisco might be the most abundant enzyme in the world.
Rubisco is ubiquitous in photosynthetic organisms, crucial for carbon fixation, contributing to its massive abundance on Earth.
-Low catalytic efficiency, high demand, key enzyme
Explain why the maintenance of a high concentration of CO2 in the bundle-sheath cells of C4 plants is an example of active transport. How much ATP is required per molecule of CO2 to maintain a high CO2 concentration?
because C4 plants use ATP to move CO2 against its concentration gradient from mesophyll to bundle-sheath cells to minimize photorespiration. This concentrated, uphill transport requires energy to regenerate phosphoenolpyruvate (PEP), consuming 2 ATP per molecule of CO2 fixed.
Glyceraldehyde 3-phosphate dehydrogenase in chloroplasts uses NADPH to participate in the synthesis of glucose. In gluconeogenesis in the cytoplasm, the isozyme of the dehydrogenase uses NADH. Why is the use of NADPH by the chloroplast enzyme advantageous?
because it directly utilizes the reducing power generated by the light-dependent reactions (PSI) within that same organelle to drive the Calvin cycle.
-This locally produced NADPH acts as an efficient, direct electron donor for biosynthesis, whereas using NADH would require costly energy to transport it into the chloroplast
Rubisco requires a molecule of CO2 covalently bound to lysine 201 for catalytic activity. The carboxylation of rubisco is favored by high pH and high Mg2+ concentration in the stroma. Why does it make good physiological sense for these conditions to favor rubisco carboxylation?
The stroma will accumulate Mg^2+ and become alkaline as a consequence of the movement of protons from the stroma to the thylakoid space. Thus, rubisco is primed for activity when the light reactions are providing ATP and NADPH required for carbon fixation and glucose synthesis.
What are the light-dependent changes in the stroma that regulate the Calvin cycle?
The light reactions lead to an increase in the stroll concentrations of NADPH, reduced ferredoxin, and Mg^2+, as well as an increase in pH.
In the C4 pathway, one ATP molecule is used in combining the CO2 with phosphoenolpyruvate to form oxaloacetate, but, in the computation of energetics bookkeeping, two ATP molecules are said to be consumed. Explain.
a second ATP equivalent is consumed to regenerate PEP from pyruvate. The ATP→ AMP conversion in the regeneration step effectively costs two ATP molecules for each CO2 transported to bundle sheath cells, ensuring efficient carbon fixation
Before the days of pampered lawns, most homeowners practiced horticultural Darwinism. A result was that the lush lawns of early summer would often convert into robust cultures of crabgrass in the dog days of August. Provide a possible biochemical explanation for this transition
occurs because crabgrass is a C4 plant, giving it a biochemical advantage over C3 grasses in hot, dry conditions.
~In August, high temperatures increase the oxygenase activity of the enzyme Rubisco, making C3 plants inefficient, while C4 plants utilize PEP carboxylase to efficiently concentrate for photosynthesis, thriving while other grasses struggle
What is photorespiration, what is its cause, and why is it believed to be wasteful?
Rubisco sometimes reacts with O2 instead of CO2, catalyzing a useless oxygenate reaction. A salvage pathway recovers part of its carbon skeleton, recycling 3 of the 4 carbon atoms of two molecules of glycolate. However, one carbon atom is lost as CO2.
Why do high concentrations of CO2 inhibit photorespiration?
When CO2 levels are high, Rubisco binds CO2 (carboxylation) instead of oxygen (oxygenation), decreasing the wasteful process of photorespiration
Why would any plant use C4 metabolism instead of C3 metabolism given that C3 metabolism is so much more efficient?
C4 plants use the more energy-intensive metabolism (30 ATP vs. 18 ATP) because it minimizes photorespiration. While less efficient in cooler climates, C4 metabolism is advantageous in hot, dry, or low-CO conditions because its enzymes (PEP carboxylase) fix carbon more effectively, reducing water loss and preventing the wasteful binding of oxygen to RuBisCO.
Identify the similarities and differences between the Krebs cycle and the Calvin cycle.
-both fundamental metabolic pathways that operate in cycles, regenerates their starting compounds, and involve electron carriers (NADPH/NADH).
-Differences are that the Krebs cycle is catabolic (breaks down fuel to release energy), occurs in the mitochondria, and produces CO2. The Calvin cycle is anabolic (uses energy to build sugars), occurs in the chloroplast stroma, and consumes CO2
What are the three steps in glycogen degradation, and what enzymes catalyze each step?
Step 1 is the release of glucose 1-phosphate from glycogen by glycogen phosphorylase.
Step 2 is the formation of glucose 6-phosphate from glucose 1-phosphate, a reaction catalyzed by phosphoglucomutase.
Step 3 is the remodeling of the glycogen by the transferase and the glucosidase.