CSU Chemistry 110 FINAL
1/98
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
99 Terms
What is Chemistry?
The study of matter and the changes it undergoes.
SUSTAINABLE CHEMISTRY is designed to meet the needs of the present generations without compromising the needs of future generations.
What is Science?
It is based on observations and experimental tests of our assumptions. It is REPRODUCIBLE, TESTABLE, TENTATIVE, PREDICTIVE, and EXPLANATORY. Hallmark is the ability to think CRITICALLY.
What is a Scientific Hypothesis?
Are TESTABLE, PREDICTIVE, and EXPLANATORY. Educated, testable guess.
What is the Scientific Law?
A brief statement summarizing many observations of a physical phenomenon. Scientific laws are UNIVERSAL.
What is the Scientific Theory?
The best representation for an explanation for a phenomenon, but is always TENTATIVE. Theories organize scientific knowledge and are also useful for their PREDICTIVE value. A theory developed in one area is often found to apply in others.
EX: After many observations, Proust stated that elements with oxygen, depending upon the experimental conditions.
Applied Research
Work-oriented toward the solution of a particular problem in an industry or the environment.
EX: A biochemist is hired by a pharmaceutical firm to synthesize a medical remedy for cancer.
EX: A chemist develops a better, longer lasting, pain reliever.
EX: Scientist creates rBST to improve milk production in cows.
Basic Research
The search for the knowledge for its own sake. The results of basic research are often used as the basis for technological advances. Is conducted primarily in universities and research institutes.
EX: A researcher investigates the responses a plant has to drought conditions.
EX: A scientist seeks to extract biologically active compounds from sea sponges.
Mass
Is a measure of the quantity of matter that an object contains. Greater the mass the more difficult it is to change velocity. Does not vary with gravity. Is a better measure of the amount of matter than weight because MASS IS INDEPENDENT OF LOCATION.
Weight
Is a measurement of force. Varies with gravity.
Chemical Changes
Involves a change in the chemical identity of matter other substances that are chemically different.
EX: A tree is watered and fertilized, and it grows larger.
Physical Changes
Involves an alteration in the physical appearance of matter without changing its chemical identity or composition.
EX: Ice cube melts to liquid but is still water.
EX:Sheep are sheared, and the wool is spun into yarn.
State of Matter
SOLID: Object maintains its shape and volume regardless of its location.
LIQUID: Occupies a definite volume but assumes the shape of the portion of a container that it occupies.
GAS: Maintains neither shape nor volume. It expands to fill completely whatever container it occupies. They flow and are easily compressed.
What are Elements, Compounds, and Mixtures?
ELEMENTS: One of the fundamental substances from which all material things are constructed. Cannot be broken down into simpler substances by any chemical processes. Ex: Fe, Al, Na
COMPOUNDS: A substance made up of two or more elements chemically ocmbined in a fixed ratio. Ex: Water (H2O)
MIXTURES: Composed of two or more substances. Retain identity. Do not change chemically they simpy mix. Can be separated by physical means. Can be homogenous or heterogenous.
~ Homogenous: same compositon. Ex: Salt water
~Heterogenous Ex: Sand and water, grains of sand is different than the water.
Base units of the metric system
Length=Meter (m)
Mass=Kilogram (kg)
Time=Second (s)
Temperature=Kelvin (K)
Amount of substance=mole (mol)
Electric current=ampere (A)
Useful Calculations
Density: d = m/V ; m = d x V ; V = m/d
Temperature: K = C+273.15
Know names of philosophers/scientists and what they did.
LEUCIPPUS: Must be tiny particles of water
DEMOCRITUS:
ARISTOTLE: Believed that matter was continuous and was not made up of atoms.
Law of Conservation of Mass
Established by Lavoisier. States that matter is neither created nor destroyed during a chemical change.
Lavoisier is also known as the "Father of Modern chemistry."
Law of definite proportions
Established by Proust. States that a compound always contains the same elements in certain definite proportions and in no others.
EX: The observation that 10g of hydrogen combines with 80g of oxygen to form 90g of water, even when there is more than 80g of oxygen present in the reaction container.
Law of multiple proportions
Established by Dalton. Staes that elements might combine in more than one set of proportions, with each set corresponding to a different compound.
EX: Nitrogen forms a number of different compounds with oxygen, depending upon the experimental conditions.
Dalton's atomic theory
All matter is composed of extremely small and indestructible particles called atoms.
Elements are made of just one type of atom that is unique to that element.
Compounds are formed when atoms of different elements combine in fixed proportions.
A chemical reaction involves a rearrangement fo atoms, but there is no breaking, destroying, or creating of atoms.
What is an isotope?
Atoms that do not have the same number of protons but different numbers of neutrons.
Atoms of the same element that have different masses.
How are atomic masses determined?
Add the number of protons to the neutrons. (p+ + n)
Know how Mendeleev's periodic table was arranged.
He arranged the periodic table primarily in order of their ATOMIC MASSES.
What is ELECTROLYSIS, ELECTROLYTES, CATHODE, and ANODE?
ELECTROLYSIS: The chemical reactions cause by electricity.
ELECTROLYTES: A compound that conducts electricity when melted or put into solution.
CATHODE: A negative electrode.
Cathode rays are beams of electrons.
ANODE: A positive electrode.
Know scientists/philosophers and their experiments relating to the development of theories of atomic structure, and discovery involved in each experiment.
THOMSON: By measuring the deflection of cathode rays in electrical and magnetic fields, he was able to determine RATIO OF MASS TO CHARGE FOR THE ELECTRON.
GOLDSTEIN: The existence of POSITIVELY charged particles in gas discharge tubes was first experimentally verified.
MILLIKAN: "Oil Drop" apparatus allowed for the determination of the CHARGE of electrons.
RUTHERFORD: Proposed that atoms are mostly empty space, most of the mass of atoms is located in a dense, small volume nucleus, the nucleus is positively charged.
ROENTGEN: Discovered the x-ray.
What is radioactivity? What are the 3 types and properties?
Radioactivity is the spontaneous emission of radiation from certain unstable elements.
~Alpha particle: Has a mass of 4 and charge of 2+
~Beta particle: Is an electron with more energy than an electron in an atom. It has a charge of 1-.
~Gamma rays: A form of electromagnetic radiation. Are not deflected by a magnetic field. Exposed through x-rays. Has a mass of 0 and a charge of 0.
Know 3 types of subatomic particles, locations, and properties.
PROTON: p+; has a mass of 1 and a charge of 1+; is located in the nucleus.
NEUTRON: n; has a mass of 1 and a charge of 0; is located in the nucleus.
ELECTRON: e-; has a mass of 1/1837; has a charge of 1- and is located outside the nucleus.
Be able to calculate the number of PROTONS, NEUTRONS, or ELECTRONS.
PROTONS: The atomic number
NEUTRONS: Subtract the atomic number from the mass number.
ELECTRONS: Same as protons to even out.
Electron Configuration
Table on page 83.
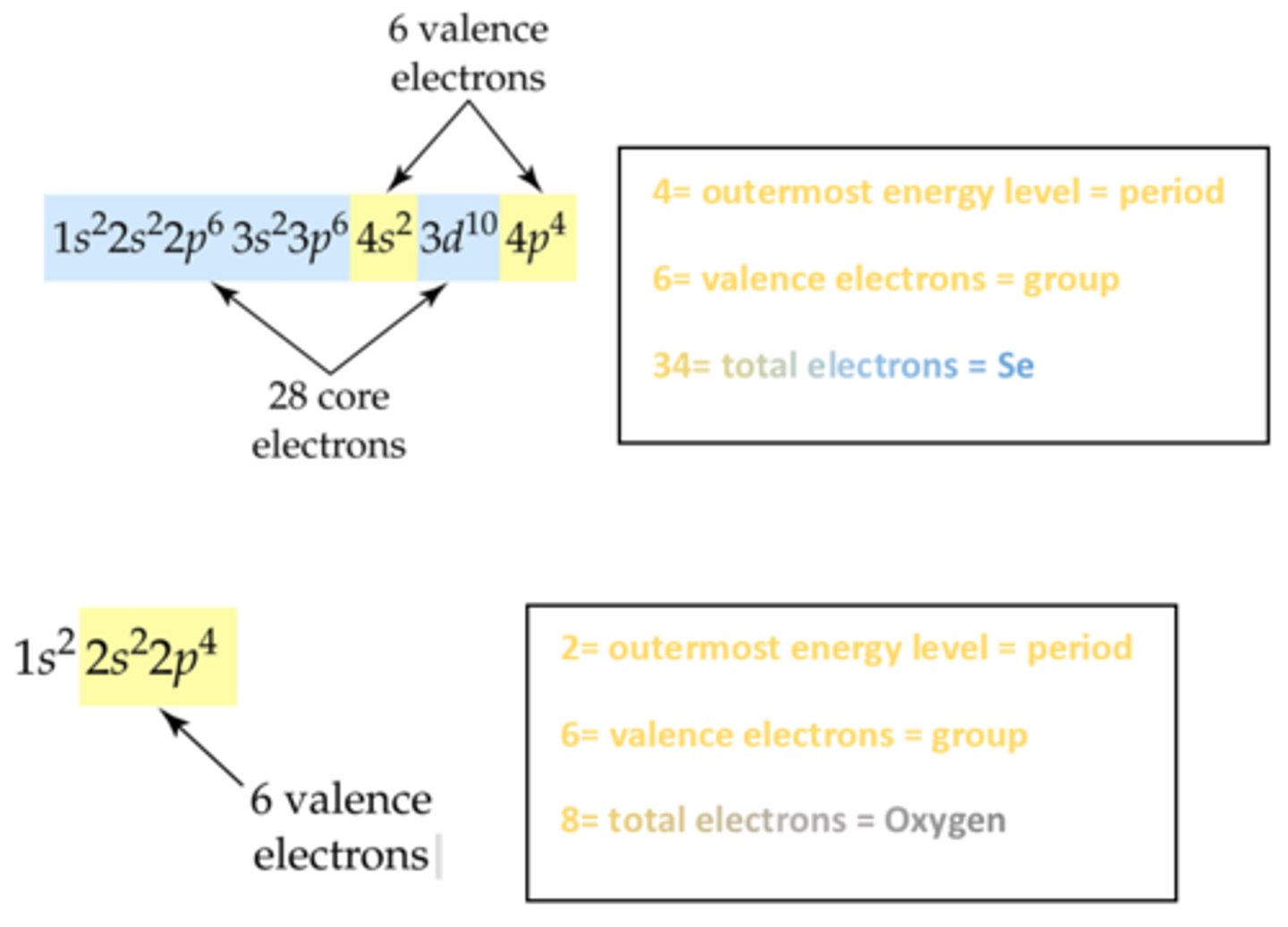
Names of Rows and Columns in modern periodic table.
ROWS: Periods
COLUMNS: Groups (Family)
~1A: Alkali metals
~2A: Alkaline earth metals
~7A: Halogens
~8A: Noble gases
Know Valence Electrons for each group.
The electrons in the outermost main shell.
The group number in front of (A) is the valence electrons.
Covalent Bond
Formed between two nonmetals.
Ionic Bond
Formed between a metal and a nonmetal.
VSEPR theory
VALENCE SHELL ELECTRON PAIR REPULSION theory; Used to predict the arrangement of atoms about a central atom in a molecule.
The basis of this theory is that electron sets arrange themselves as far away from each other as possible so as to minimize repulsions between these like-charge particles.
Avogradro's number
6.02 X 10^23
What is a dipole?
A molecule in which a concentration of positive electric charge is separated from a concentration of negative charge.
What are the properties of liquid, solid, and gas?
GAS: Has no definite shape and no definite volume.
LIQUID: Has a definite volume but no definite shape.
SOLID: Has a definite volume and shape.
Condensation
Gas changes into liquid.
Vaporization
Liquid is converted into a gas.
Freezing
Liquid is converted into a solid.
Melting
Solid is converted into liquid.
Sublimation
Solid is converted into a gas.
Ionic Compounds
Can dissolve in water
Those that dissolve can conduct electricity
Almost all ionic compounds are solids at room temperature
Have much higher melting points and boiling points than molecular compounds.
Takes more energy to melt a mole of an ionic compound than a molecular compound.
In solid, form is hard and brittle.
Molecular Compounds
Softer than ionic compounds.
Do not form conductive solutions.
Some are gases and liquids but are mostly solids at room temperature
Less energy to melt and boil molecular compounds due to weak bonds that connect them
Hydrogen Bonds
Are exhibited by dotted lines to emphasize their strength is much greater than that of other intermolecular forces.
Order of strength of intermolecular forces
Strong to weak
Hydrogen bond - Dipole-Dipole - Dispersion Forces
Kinetic-Molecular Theory
1.) Particles of a gas are in rapid, constant motion and move in straight lines.
2.) The particles of a gas are tiny compared with the distances between them.
3.) Because the particles of a gas are far apart, there is very little interaction between them.
4.) Particles of a gas collide with one another. Energy is conserved in these collisions; energy lost by one particle is gained by the other.
5.) Temperature is a measure of the average kinetic energy of the gas particles.
Gas laws
BOYLE'S LAW: For a given amount of gas at a constant temperature, the volume of the gas varies inversely with its pressure. P1V1=a=P2V2
CHARLE'S LAW: Volume of a fixed amount of a gas at a constant pressure is directly proportional to its absolute temperature. V1/T1=V2/T2
GAY-LUSSAC'S LAW: The pressure of a fixed amount of a gas at a constant volume is directly proportional to its absolute temperature. P1/T1=P2/T2
P2=P1T2/T1
IDEAL GAS LAW: PV=nRT n=mol
COMBINED GAS LAW:Incorporation of the simple gas laws. P1V1/T1=P2V2/T2
An arrhenius acid
H+
A molecular substance that breaks up in aqueous solution into hydrogen ions and an anion.
An arrhenius base
OH-
A substance that releases hydroxide ions in an aqueous solution.
An Bronsted-Lowery acid
A proton donor.
An Bronsted-Lowery base
A proton acceptor.
Acid
Tastes sour
Causes litmus indicator dye to turn red
Dissolves active metals such as zinc and iron, producing hydrogen gas
Reacts with bases to form water and ionic compounds called salts.
Base
Tastes bitter
Causes litmus indicator dye to turn blue
Feels slippery on the skin
Reacts with acids to form water and salts.
Acid-Base indicator
Test that identifies a substance as an acid or a base.
Strong Acids
Sulfuric Acid
Hydrochloric Acid
Nitric Acid
Neutralization Reaction
The products are salt and water.
Acidic and Basic Anhydrides
ACIDIC ANHYDRIDE: Nonmetal oxide reacts with water.
BASIC ANHYDRIDE: Metal oxide reacts with water to form bases.
Strong and weak acids and bases
STRONG ACID: Ionizes completely with water.(HCl-Hydrochloric acid)
WEAK ACID: Ionizes only slightly with water. (HCN) Most acids are weak.
STRONG BASE: Completely ionized in water. (NaOH-Sodium Hydroxide)
WEAK BASE:Slightly ionized in water. (NH3-Ammonia)
pH calculation
pH= -log[H+]
What is Oxidation?
During combustion, oxygen is added.
GAIN OXYGEN
LOSE HYDROGEN
LOSE ELECTRONS
INCREASE OXIDATION NUMBER
What is Reduction?
During the combustion, oxygen is removed.
LOSE OXYGEN
GAIN HYDROGEN
GAIN ELECTRONS
DECREASE OXIDATION NUMBER
Oxidizing agents
When a reactant causes another reactant to become oxidized.
IS WHAT IS REDUCED.
Reducing agents
When a reactant causes another reactant to become reduced.
IS WHAT IS OXIDIZED.
Function of a battery
A battery is a collection of electrochemical cells.
Lead batteries are rechargeable. Are also durable. Contain corrosive sulfuric acid and lead is toxic to the environment.
Current batteries involve lithium. Lithium has low density but high voltage. Lithium-ion batteries are also used in tablets and smartphones.
Properties of Ozone
Powerful oxidizing agent. Used in water treatment. Can completely break down urea. Can be a harmful air pollutant. Destructive to rubber. Has a shield that protects life on Earth from ultraviolet radiation from the sun.
Catalyst
A substance that increases the rate of a chemical reaction without itself being used up. Lower activation energy. Allows for the use of oxygen or hydrogen peroxide for cleaning instead of chlorine. Purifies water.
What is organic chemistry?
Chemistry of carbon-containing compounds.
Simple Alkanes
Single bond Equation: C(n)H2(n)+2
Methane: 1 carbon atom
Ethane: 2 carbons
Propane: 3 carbons
Butane: 4 carbons
Pentane: 5 carbons
Hexane: 6 carbons
Heptane: 7 carbons
Octane: 8 carbons
Decane: 10 Carbons
Toxicity of Ethanol
Generally acts as a mild depressant. Ingestion over a period of time can alter brain cell functions, cause nerve damage, increase disease of liver, cardiovascular system, and can give baby fetal alcohol syndrome.
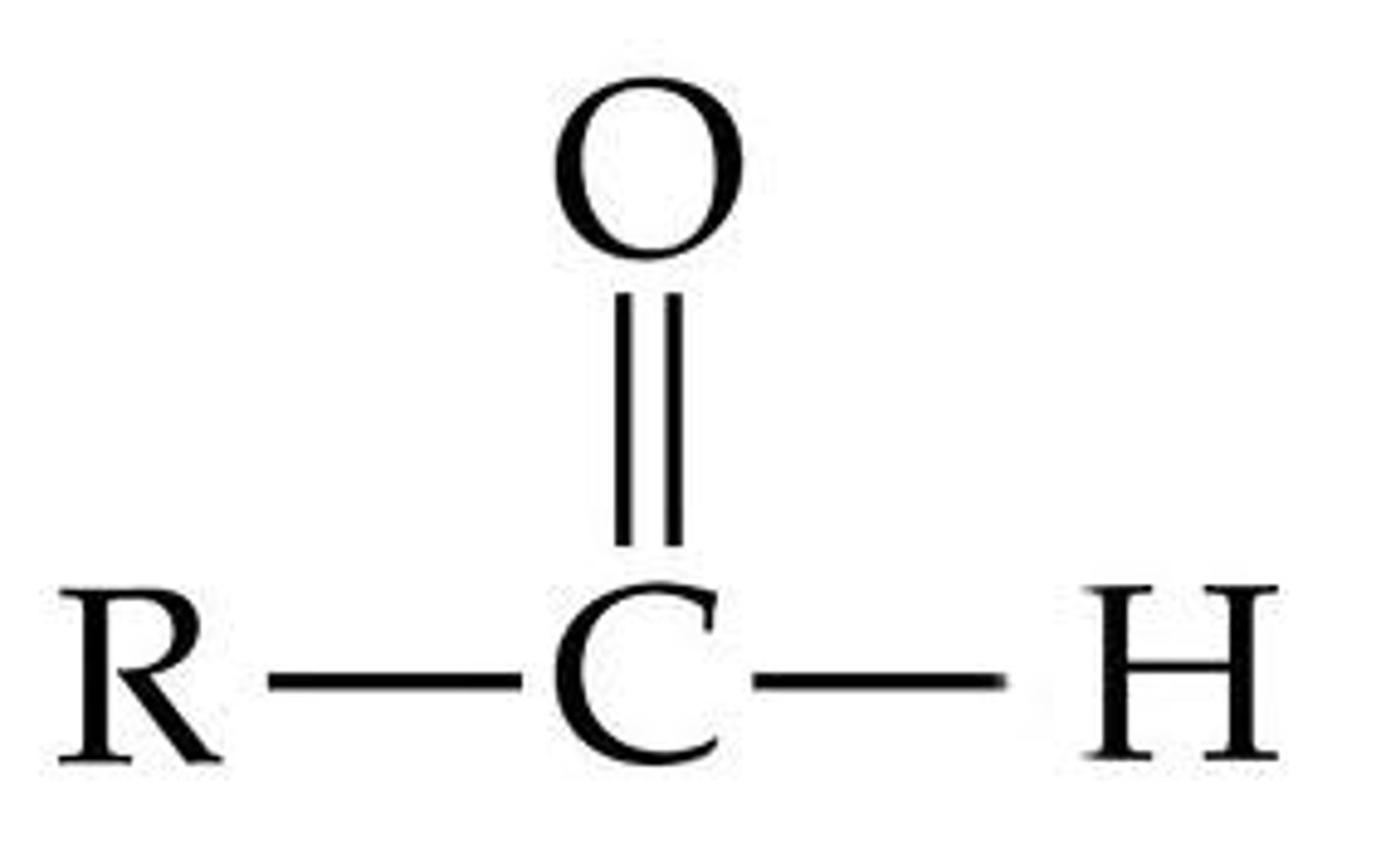
Aldehydes
Contains a carbonyl group which is carbon double bonded to oxygen. Have at least one H atom attached to the carbonyl carbon.
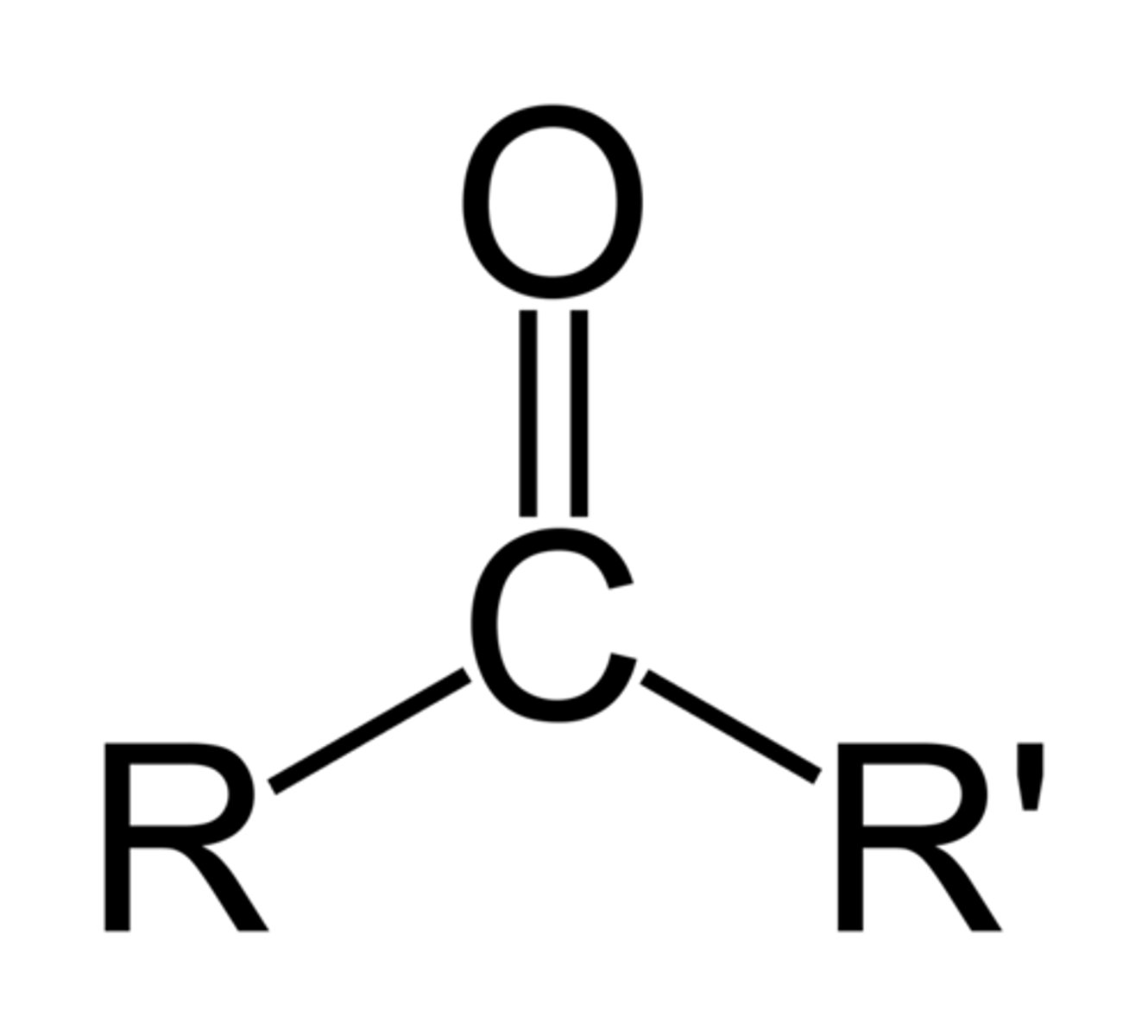
Ketones
Contains a carbonyl group which is a carbon double bonded to oxygen. Have two carbons joined to the carbonyl carbon.
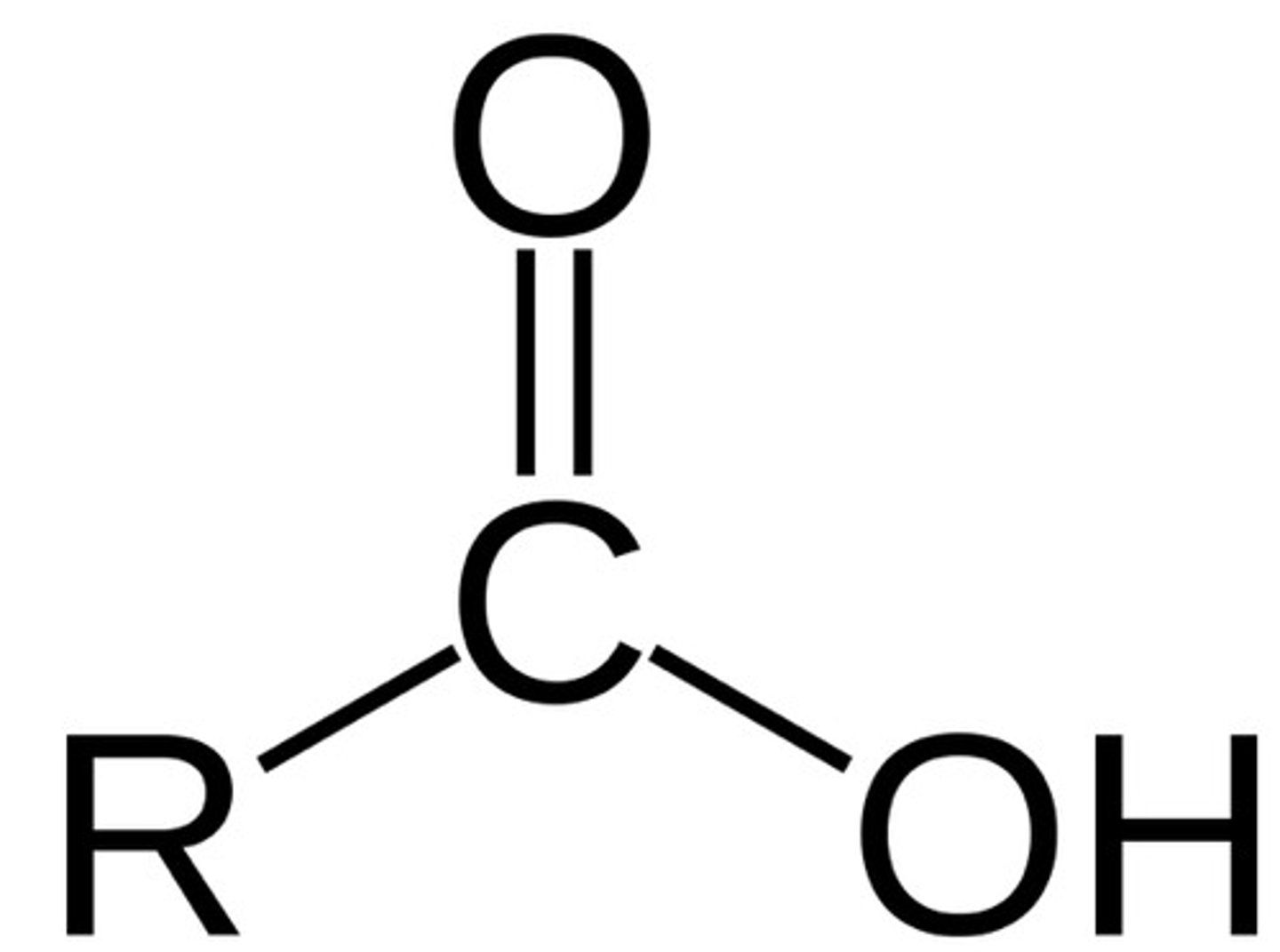
Carboxylic Acid
Monomers and Polymers and their relationship
MONOMER: Small molecule building block from which a polymer is made.
POLYMER:"many parts" is made from monomers.
Polymerization converts monomers to polymers.
HDPE
HIGH-DENSITY POLYETHYLENE
Has mostly linear molecules that pack closely together and can assume a fairly well-ordered, crystalline structure. Are rigid and have good tensile strength.
ARE USED FOR THREADED BOTTLE CAPS, TOYS, BOTTLES, AND MILK JUGS.
LDPE
LOW-DENSITY POLYETHYLENE
Has many side chains branching off the polymer molecules. The branches prevent the molecules from packing closely together and assuming a crystalline structure. Are waxy, bendable plastics that have low densities and melting points.
ARE USED TO MAKE PLASTIC BAGS, PLASTIC FILM, SQUEEZE BOTTLES, ELECTRIC WIRE INSULATION, AND HOUSEHOLD PRODUCTS.
LLDPE
LINEAR LOW-DENSITY POLYETHYLENE
Is a copolymer, a polymer formed from two or more different monomers. Are made by polymerizing ethylene with a branched-chain alkene.
ARE USED TO MAKE PLASTIC FILMS FOR LANDFILL LINERS, TRASH CANS, TUBING, AND AUTOMOTIVE PARTS.
Thermoplastic and Thermosetting polymers
THERMOPLASTIC: Can be softened and reshaped. Can be melted and remolded.
THERMOSETTING: Harden permanently when formed. Cannot be softened from heat and remolded. Heat cause discoloration and decomposition. Permanent hardness is due to cross-linking of polymer chains.
Use of Teflon
Is used for making non-sticking cookware, electric insulation, bearings, and gaskets.
Vulcanization of rubber
Vulcanization is when sulfur atoms cross-link the hydrocarbon chains side to side.
Rubber is naturally soft and tacky when hot. It is hardened with sulfur by vulcanization.
Rubber is then harder, stronger and suitable for automobile tires. It also has improved elasticity.
Most commonly split out molecule between monomers to make a condensed polymer
Water
Addition and Condensation Polymers
ADDITION POLYMERIZATION: Aka chain reaction polymerization. The monomer molecules add to one another in such a way that the polymeric product contains all the atoms of the starting monomers.
CONDENSATION POLYMERIZATION: Aka step-reaction polymerization. Some part of each monomer is NOT incorporated in the final polymer.
Methods used to minimize disposal of plastics
Recycling
Incineration (Burning)
landfills
Ionizing radiation
Radiation with enough energy to knock electrons off atoms or molecules.
5 Radioactive decay and nuclear change
ALPHA DECAY: Decrease nucleus by 4; Decreases atomic number by 2.
BETA DECAY: No change in nucleus number; Increases atomic number by 1.
GAMMA DECAY: No change in nucleus or atomic number.
POSITRON EMMISION: No change in nucleus number; decreases atomic number by 1.
ELECTRON CAPTURE: No change in nucleus number; decreases atomic number by 1.
Half-life
1/2^n n= NUMBER OF HALF LIVES
Common radioactive isotopes used in dating
CARBON-14: Charcoal and organic materials
HYDROGEN-3: Aged wine and brandies
LEAD-210: Skeletal remains
POTASSIUM-40: Rocks, Earth's crust, moon's crust
RHENIUM-187: Meteorites
URANIUM-238: Rocks and Earth's crust
Use of radioisotopes in treating foods
Used in preservation of food.
Use of iodine-131 in medical diagnostics
Thyroid diagnosis and therapy
Penetration powers of radiation
ALPHA: Least penetrating (Can be stopped by a sheet of paper)
BETA: More penetrating (thin sheet of aluminum, block of wood can stop)
GAMMA: Most penetrating (meters thick of lead to stop)
Nuclear fusion and fission
FUSION: Combination with another nucleus to form a bigger nucleus.
FISSION: Induced to split into two or more fragments by some external source.
What are ribosomes, mitochondria, and chloroplasts?
RIBOSOMES: Where protein synthesis takes place.
MITOCHONDRIA: Cell's "battery" where energy is produced.
CHLOROPLASTS: Found in plant cells only. Make photosynthesis possible.
Give examples of monosaccharides, disaccharides, and polysaccharides.
MONOSACCHARIDES: Carbohydrates that cannot be further hydrolyzed. Ex: FRUCTOSE is ketose, GLUCOSE, GALACTOSE are aldoses.
DISACCHARIDES: Carbohydrates whose molecules can be hydrolyzed to yield two monosaccharide units.
Ex: Sucrose= Glucose+ Fructose and Lactose= Glucose+ Galactose.
POLYSACCHARIDES: Composed of large molecules that yield many monosaccharide units on hydrolysis.
Ex: Starch, Cellulose
Functional groups contained in amino acids
Amino Group: NH2
Carboxyl Group : COOH
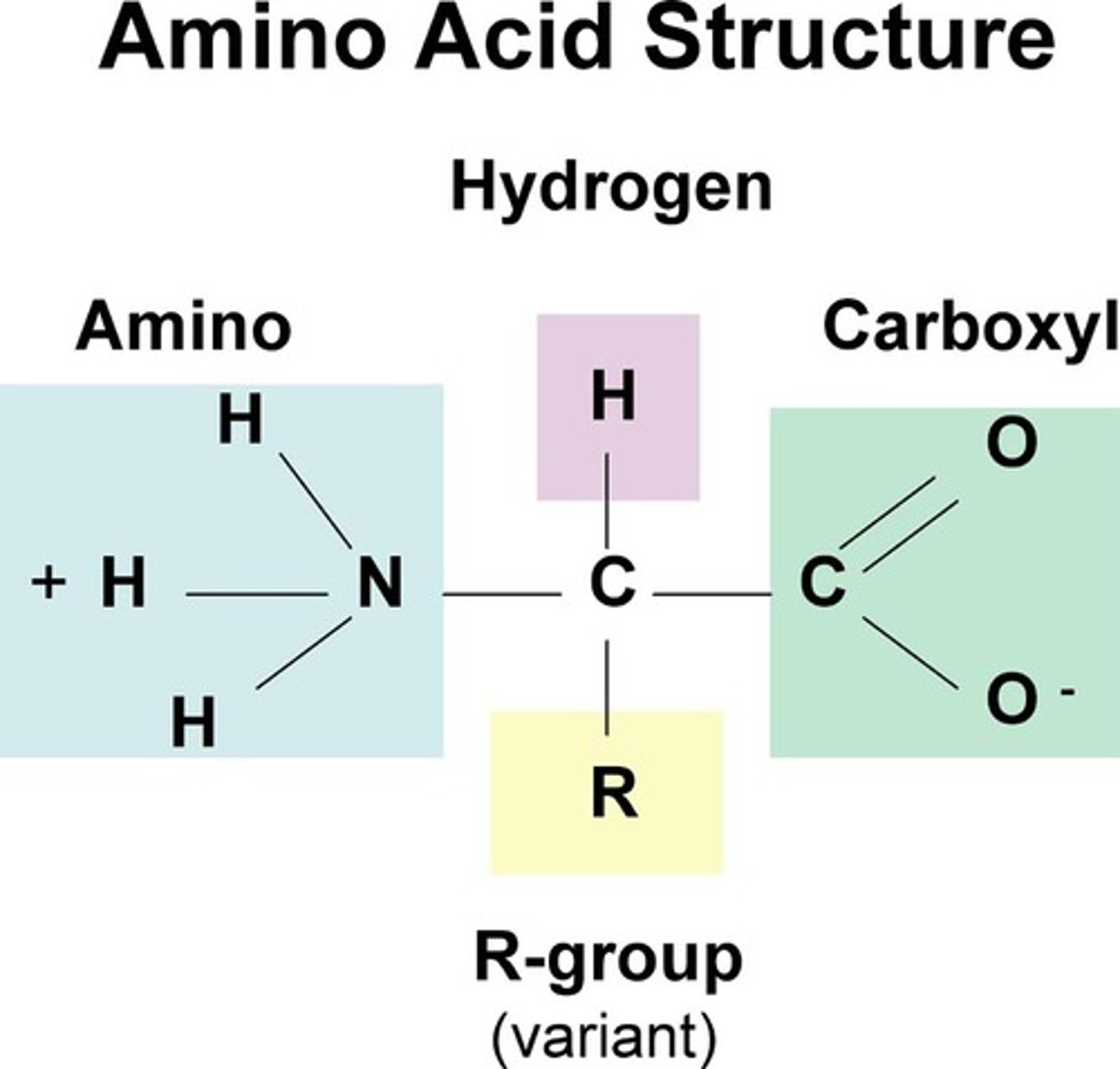
What is a peptide bond?
A molecule with more than 10 amino-acid units.
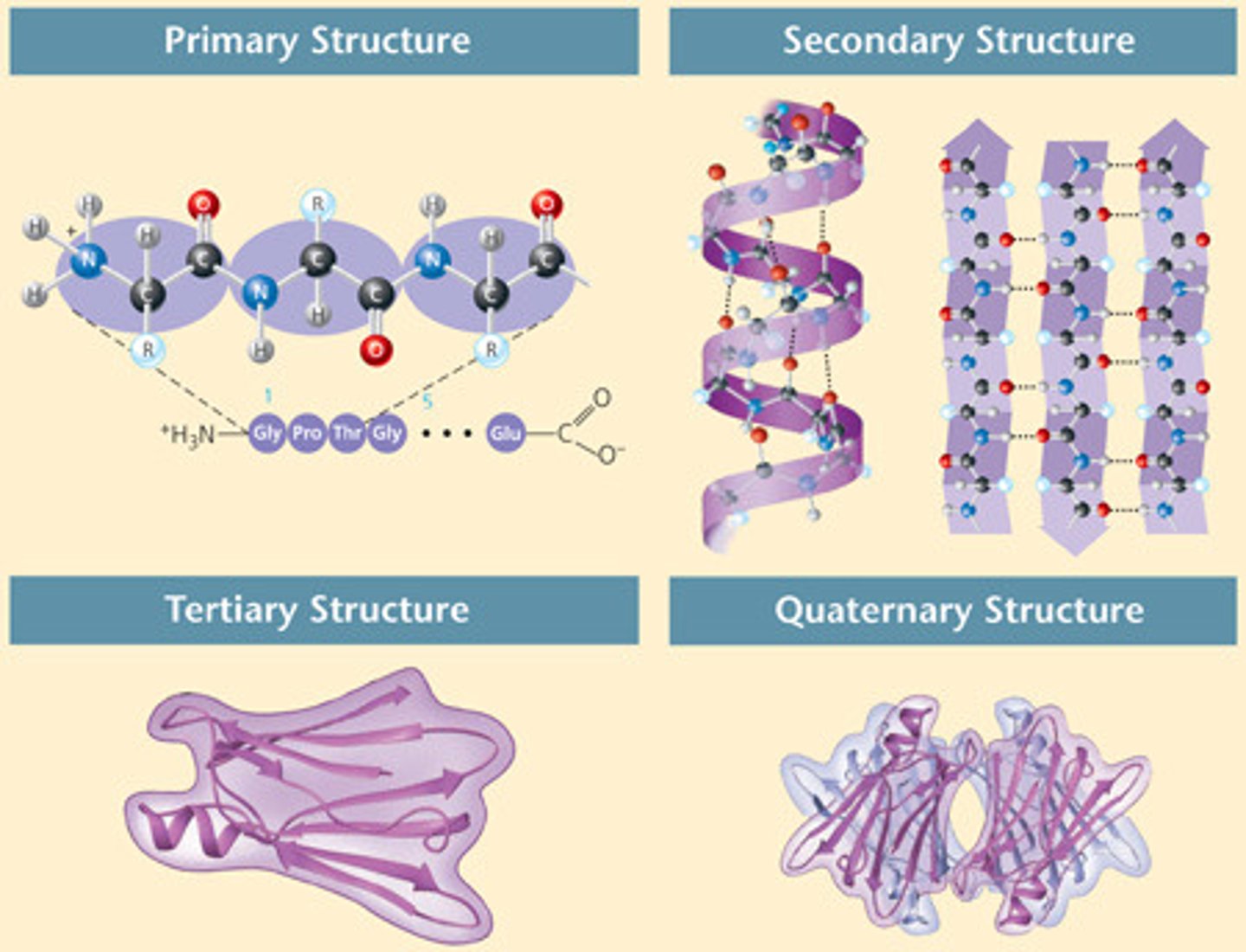
Structures of proteins
PRIMARY: Order of amino acids. (Sequence)
SECONDARY:Alpha helix, Beta pleated sheet when folded
TERTIARY: Protein folding creates spatial relationships between amino acid units that re relatively far apart in the protein chain.
QUATERNARY: Two or more polypeptide chains can assemble into multiunit structures.
Difference between DNA and RNA.
DNA: Double stranded
RNA: Single stranded
How many chromosomes are in human body cells?
46 or 23 PAIRS
What is replication, transcription, and translation?
REPLICATION: Copying or duplication of DNA molecules.
TRANSCRIPTION: Segment of DNA transfers it's information to mRNA .
TRANSLATION: mRNA is translated into protein.