Nucleophilic Substitution and elimination reactions (RJP lectures 1-6)
1/42
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
43 Terms
What is the mechanism for the general SN2 reaction?
Nucleophile attacks carbon and creates bond while leaving group leaves simultaneously (making and breaking bond at the same time)
SN2 is bimolecular (relies on 2 molecular species) rate depends on concentration of substrate and nucleophile
What is the mechanism for the general SN1 reaction?
Leaving group leaves on its own forming a carbocation intermediate. Nucleophile then attacks in at carbocation to form product
SN1 is unimolecular (relies on one molecular species) — rate is independent of concentration of nucleophile
Characteristics of a nucleophile
Generally have a full or partial negative charge and a lone pair of electrons
Characteristics of a substrate
Molecule that contains the leaving group
Nucleophile tends to be attracted to an atom that bears a full or partial negative charge
Where does the nucleophile attack in from in Sn2 and what does this do to the stereochemistry of the product?
The nucleophile does a backside attack (180º from leaving group) leading to Walden Inversion (inversion of stereochemistry)
SN2 is stereospecific
Where does the nucleophile attack from in SN1 and what does this mean for the stereochemistry of the product?
The nucleophile can attack from either the front or the back because the carbocation is planar — this leads to racemic mixture of products
Rate law for SN2
rate = k[nuc][subs]
Rate law for SN1
rate = k[subs]
first step is rate determining step
What is the mechanism for the general E2 reaction
Base picks up a proton from the substrate — base take a beta hydrogen (hydrogen connected to carbon adjacent to carbon with leaving group) — electrons from that bond move to form a double bond between the two carbons, and the leaving group leaves simultaneously. Bimolecular - rate depends on concentration of substrate AND base
What is the mechanism for the general E1 reaction?
leaving group leave first on its own forming carbocation intermediate. Then base takes beta hydrogen, electrons move to form double bond between carbons. Unimolecular - rate depends ONLY on concentration of substrate
Stereochemistry of the E2 reaction
E2 reactions are favored by the substrate conformation where the leaving group and the hydrogen atom that are eliminated are anti to each other - antiperiplanar/anticoplanar
If an E2 reaction produces a mixture of E and Z diastereomers then the major product will be the one produced from the more stable anticoplanar formation
E2 will be stereospecific if there is only 1 beta hydrogen to be eliminated
Stereochemistry of the E1 reaction
E1 reactions produce a mix of E and Z products as a result of the carbocation intermediate — even if there is only 1 beta hydrogen because rotation is possible
2 reasons that SN2, SN1, E2, and E1 compete
1) They all involve a substrate containing a leaving group
2) Any species that acts as a nucleophile also has the potential to act as a base and vice versa — so we refer to it as an attacking species
when acting as a nucleophile, the attacking species uses its lone pair to form a bond to an electron poor non-hydrogen atom
when acting as a base, the attacking species forms a bond to a hydrogen atom
Factors that influence competition between SN2, SN1, E2, and E1
1) strength of the attacking species
2) concentration of the attacking species
3) leaving group ability
4) type of carbon bonded to the leaving group
5) solvent effects
6) heat
Do strong nucleophiles favor SN1 or SN2? How does rate of reaction change with nucleophile strength? Generalization for determining nucleophile strength
Strong nucleophiles tend to favor SN2 while weak nucleophiles tend to favor SN1
Rate of SN2 reaction increases as nucleophile strength increases - SN1 rate is essentially independent of nucleophile strength
Strong nucleophiles typically have a total -1 charge; weak nucleophiles tend to be uncharged
How to identify the best nucleophile in an SN2 reaction
The less stable anion is the better nucleophile — more stable anions can delocalize the -1 charge better and are worse nucleophiles
Base strength in E2 and E1 reactions
Rate of E2 reaction increases as base strength increases - E1 rate is essentially independent of base strength
Strong bases tend to favor E2 while weak bases tend to favor E1
Steric hindrance in E2 vs SN2 reactions
strong, bulky bases tend to favor E2 over SN2 reactions — no significant steric hindrance in a proton transfer in E2, a lot of steric hindrance in SN2 trying to approach carbon
Examples of strong, bulky bases
DBN, DBU, LDA, tert-butoxide anion, neopentoxide anion
Concentration effects
High concentration of a strong attacking species favors SN2 and E2; low concentration favors SN1 and E1 — a weak attacking species favors SN1 and E1 regardless of concentration
Leaving group ability effects
SN1 and E1 are very sensitive to leaving group ability because it must leave on its own
SN2 and E2 are not as sensitive to leaving group ability because it is assisted by the attacking species
All reactions feasible with a good leaving group — with a poor leaving group only E2 and SN2 tend to be feasible
General characteristic of a good leaving group
Good leaving groups are the conjugate bases of strong acids (weak bases) — can stabilize -charge
Examples of unsuitable vs poor vs good leaving groups
Unsuitable: OH-, RO-, NH2-
Poor: F-, RCO2-
Good: Cl-, Br-, H2O, MsO-, TsO-, TfO-
Converting a poor leaving group to a good one
Acid conditions allow a reaction to take place with an otherwise unsuitable leaving group (ex OH-)
Proton transfer step converts OH to H2O, which is a good leaving group
R—OH + HBr —→ ?
conc. HBr facilitates a reaction to form alkyl bromide
1: proton transfer to make H2O LG
2: Br- attacks back in in SN2 reaction
RCOH + SOCl2—→ ?
Two sequential SN2 reactions, one at sulfur, the other at carbon — alcohol first acts as nucleophile on thionyl chloride — forms alkyl chloride R—-X
Alcohols as leaving groups
Require acidic conditions for reaction to proceed
Hybridization of carbon bonded to leaving group
Substitution and elimination reactions generally do not occur unless the carbon bonded to the leaving group is sp3 hybridized
Effect of alkyl groups on substitution and elimination reactions
SN2 reactions do not take place for tertiary substrates — steric hindrance is too great
SN1 and E1 do not take place for methyl substrates and most primary substrates — carbocation intermediates are excessively unstable
As the number of alkyl groups on the carbon atom increases, the rate of SN2 reaction sharply decreases while the rate of SN1 reaction sharply increases
E2 reactions are relatively insensitive to the number of alkyl groups on the carbon bonded to the leaving group - can occur with primary, secondary, or tertiary carbons, just not methyl substrates
** but if the LG is bonded to a primary allyl or benzyl carbon, SN1 and SN2 can take place because the primary carbocation is resonance stabilized
Temperature effects on competition
Higher temperatures tend to favor elimination
Tiebreaker for SN1 and E1 reactions
Determine whether heat is added — E1 is favored if heat is added, SN1 is favored in the absence of heat
Tiebreaker for SN2 and E2 reactions
Determine whether the attacking species acts better as a base or a nucleophile - if it is bulky, the attacking species acts better as a base and favors E2, otherwise, both are favored
Steps to predict the outcome of SN1/SN2/E1/E2 competition
1) Determine whether competition is feasible
2) Rule out reactions that are unfeasible
3) Consider the attacking species - which of the remaining reactions are favored by the attacking species
4) Apply the appropriate tiebreaker
Williamson ether synthesis reactants and mechanism
An ether is formed through the reaction of an alkoxide ion with an alkyl halide via an SN2 reaction
Zaitsev’s rule
The major elimination product is the more highly substituted alkene product — the one produced by deprotonating the carbon atom initially attached to the fewest hydrogen atoms
Hofmann product
The product formed in an E2 reaction exhibiting anti-Zaitsev regiochemistry as a result of a strong bulky base
The major product will be the less highly substituted alkene
Which is the kinetic product and which is the thermodynamic product in an E2 reaction?
Kinetic product is less highly substituted alkene (Anti- Zaitsev), thermodynamic product is more highly substituted alkene (Zaitsev)
Rules to reasonably incorporate proton transfer steps and carbocation rearrangements
Avoid the appearance of incompatible acids and bases: strong acids should generally NOT appear in mechanisms under basic conditions and vice versa
Proton transfer steps are fast and can be incorporated before or after other elementary steps to avoid incompatible species
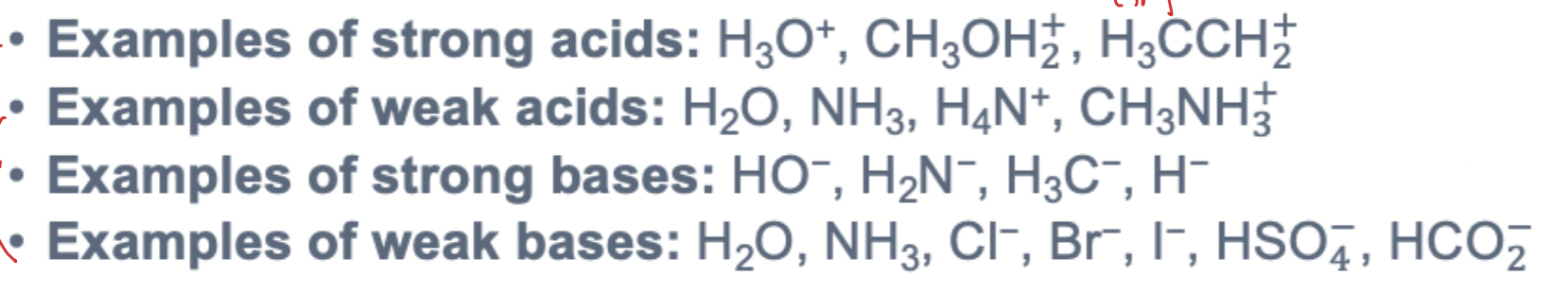
Which position should a substituent be in in a chair confirmation for the faster SN2 reaction? (axial or equatorial) and why?
Axial because it leave a less hindered approach for the nucleophile
Carbocation rearrangement
Carbocations will rearrange into more stable tertiary carbocations if possible via 1,2 hydride or 1,2 methyl shifts
What product forms when there is a resonance stabilized intermediate? (elimination)
When the carbocation intermediate has resonance structures, the more highly substituted alkene will form
What position must the leaving group sit in in the chair conformation to make E2 possible? why?
Axial because the leaving group and the beta hydrogen have to sit anti (180º) from each other for the reaction to occur
Solvent effects — which solvents favor which reactions
SN1 and E1 require polar protic solvent but SN2 and E2 can occur in protic solvent
Polar aprotic solvent favors SN2 and E2