33. NMR
1/38
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
39 Terms
What does NMR stand for?
Nuclear magnetic resonance
What are the basic principles of NMR?
● You can find the structures of complex molecules by placing them in a magnetic field and applying EM waves of radio frequency to them.
● If radio waves of the right frequency are absorbed, the nuclei flips from parallel to applied magnetic to field to anti-parallel.
● This energy change can be monitored and recorded. Uses the resonance of nuclei with spin.
How would you carry out NMR spectroscopy?
● Dissolve the liquid sample in suitable solvent, put in a tube along with a small amount of TMS and put the tube into an NMR machine.
● The sample is spun to even out any imperfections in the magnetic field and the spectrometer is zeroed against the TMS.
● Radiation with different radio frequencies but a constant magnetic field is applied to the sample and any absorptions (due to resonance) are detected
Give one use of NMR?
MRI scans
What kind of nuclei does NMR work with (and examples)?
Those with an uneven number of nucleons, meaning they will spin e.g. 1H, 13C
Energy level splits
When a carbon-13 nucleus is placed in a magnet, an energy gap arises between its 2 spin states.
The lower the electron density around the nucleus, the larger the energy gap will be.
Carbons in the same environment have identical energy level gaps between spin states (as they have equal electron densities around them)
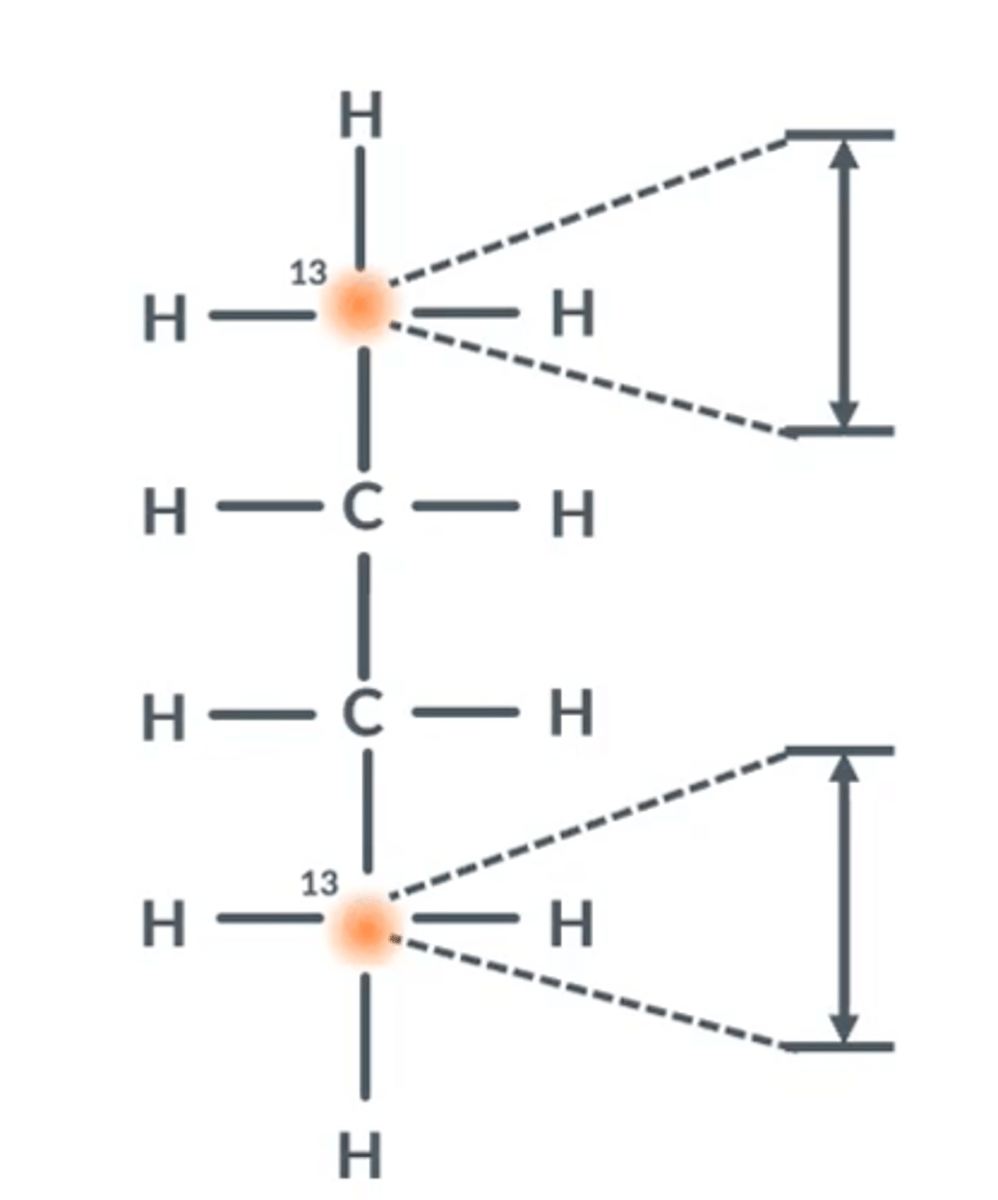
Interacting with energy gaps
Whenever molecules are placed in a magnet, each carbon-13 environment will have a certain frequency of
radio waves which causes its nucleus to switch spin states.
What percentage of carbon atoms are 13C?
1% - but modern instruments are sensitive enough to detect this
What defines the resonant frequency of a 13C atom?
The chemical environment that it is in; the amount of electron shielding it has.
2 carbons in the same environment must...
Have the same set of neighbouring atoms, the same number of bonds away.
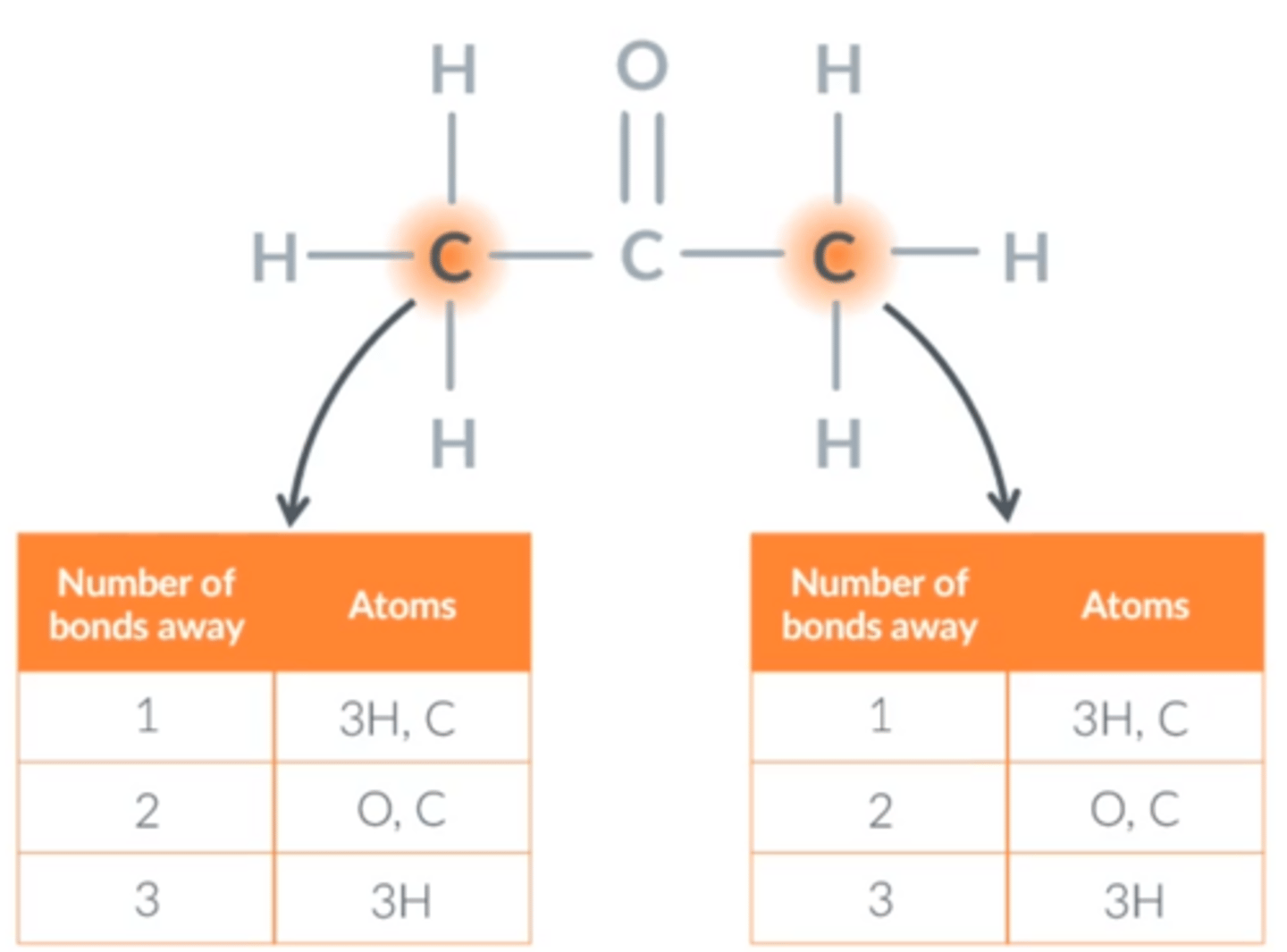
Symmetry shortcut
Whenever we can reflect a molecule so that it looks exactly the same as it did before we reflected it, any two carbons which swap places as a result of that reflection must be in the same environment.
Whenever we can rotate a molecule so that it looks exactly the same as it did before we rotated it, any two carbons which swap places as a result of that rotation must be in the same environment.
Whenever we can rotate a bond such that the molecule looks exactly the same as it did before the rotation, any two carbons which swap places as a result of that rotation must be in the same environment. SINGLE BONDS ONLY
What do the peaks on NMR spectra tell us?
The number of peaks is equal to the number of carbon environments in the molecule.
The position of peaks tells us what atoms are bonded to the carbons in each environment.
Chemical shift δ
The spectra are recorded on a scale known as the chemical shift (δ), which is how much the field has shifted away from the field for TMS
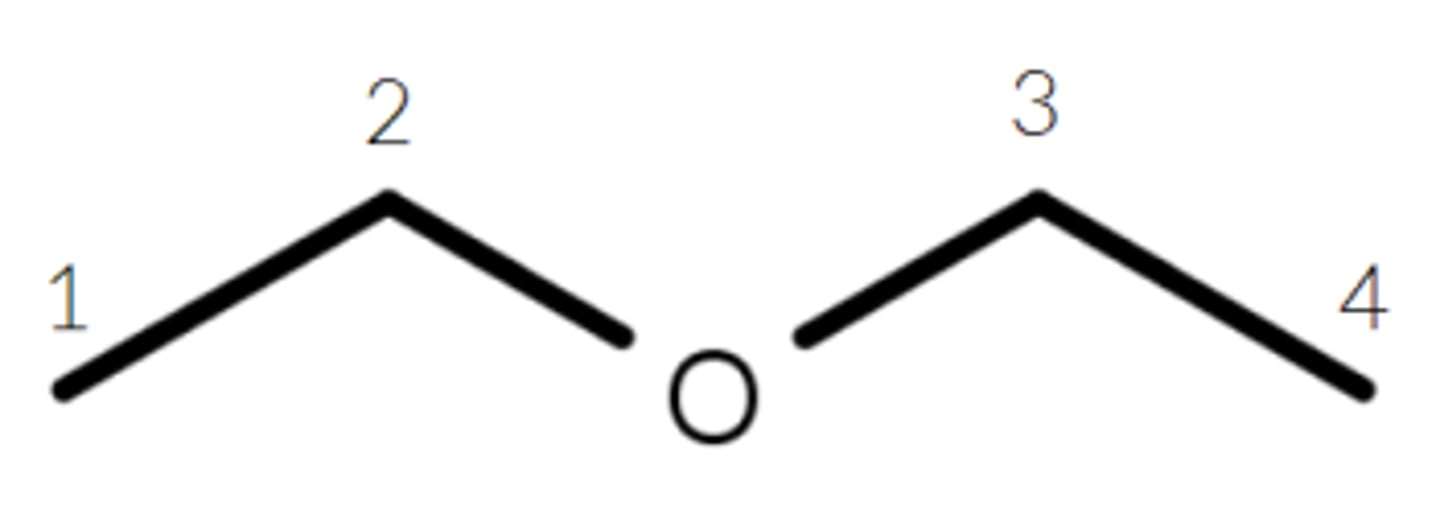
For this molecule, symmetry shortcut tells us...
What graph is produced by NMR spectroscopy?
Energy absorbed against chemical shift
What is chemical shift? What is its symbol? What are its units?
The resonant frequency of the nuclei, compared to that of a 1H atom in TMS.
Symbol _____
Parts per million (ppm)
What is the range of chemical shift for 13C NMR?
0-200ppm
What means 13C atoms show a different chemical shift value?
Having different chemical environments (but equivalent atoms show the same peak)
What kind of environment leads to a greater chemical shift?
A C atom next to more electronegative atom has a greater chemical shift.
Summarise what these mean for 13C NMR:
● Number of signals: One signal for each carbon environment (each set of inequivalent ¹³C atoms)
● Chemical shift: Greater δ from atoms closer to electronegative atoms or C=C
● Area under peak: no meaning
● Splitting: there is no splitting for ¹³C NMR
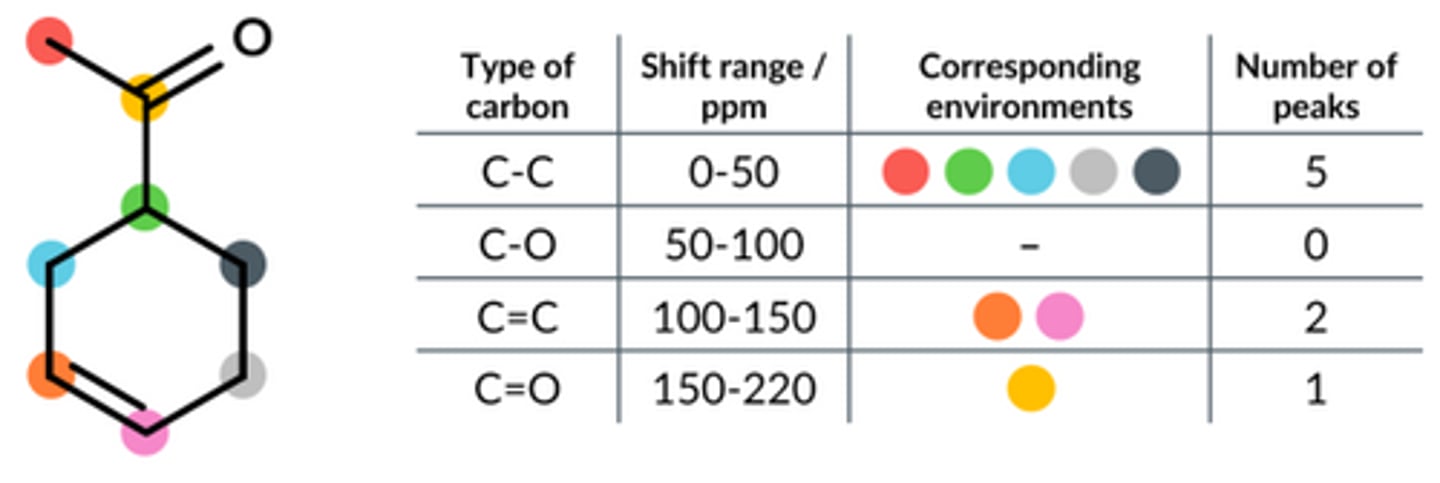
Carbon NMR is run on this molecule. State the number of peaks that occur in the following ranges.
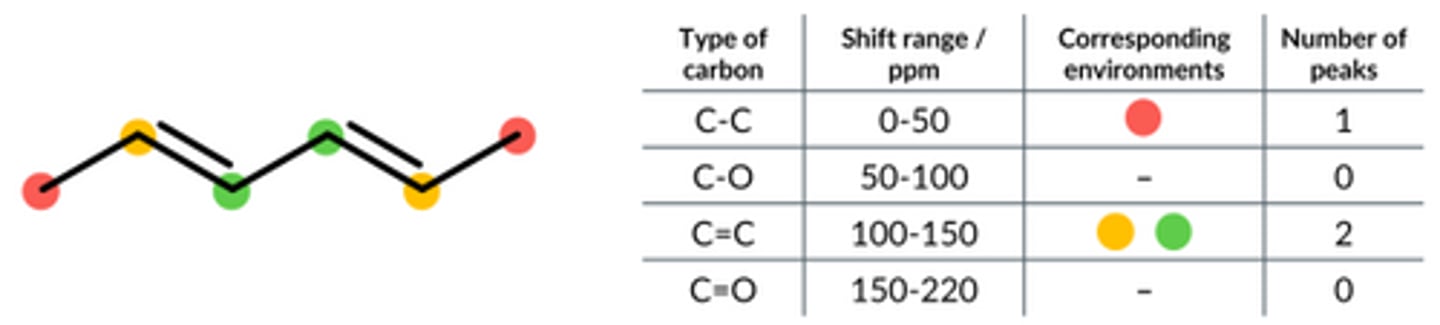
Carbon NMR is run on this molecule. State the number of peaks that occur in the following ranges.
Why is it easier to get a spectrum of 1H NMR than 13C NMR?
Most H atoms are 'H- it is much more abundant than 13C. This means almost all H atoms have spin so show up
What is the range of chemical shift for 1H NMR?
0-10ppm
What leads to a lower chemical shift value for H NMR?
1H with more electrons around them i.e. further from electronegative groups/atoms
On a low resolution spectrum, what peaks would you expect to see for H NMR?
One peak for each set of inequivalent H atoms (each chemical environment shows 1 peak)
What does the area under the peak represent (for H NMR)?
The area under the peak is proportional to the number of 1H atoms represented by the peak
What is the integration trace?
A stepped line that makes it easier to measure the area under the curve (height of line = area under that peak)
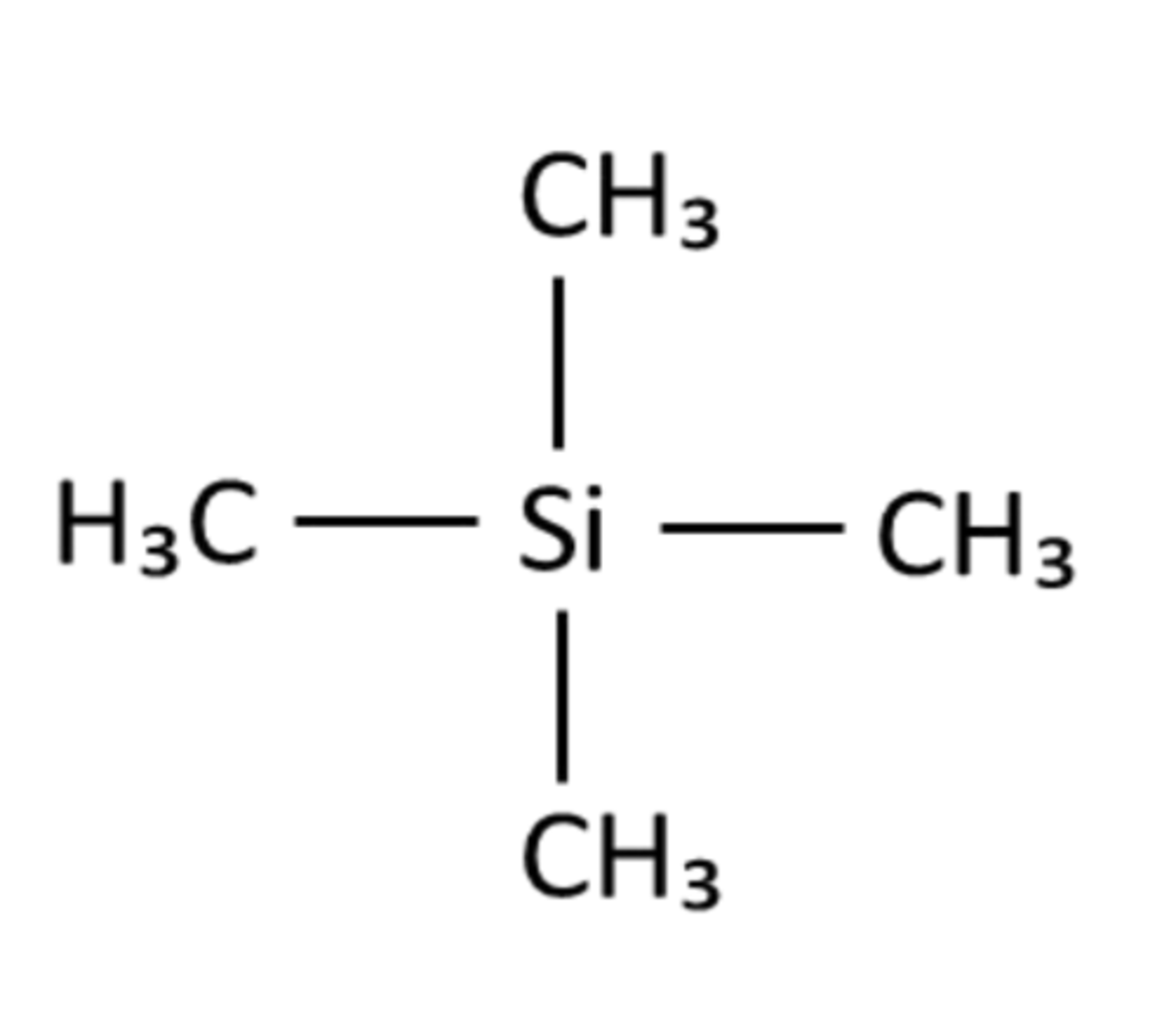
What is TMS (name and structure)?
Tetramethylsilane
PIC
What state is TMS at room temperature?
Liquid
Why is TMS used?
Can be added to sample to calibrate the NMR equipment.
It provides a peak at exactly 0 = Oppm. It is the reference point against which all are measured
Sane calibration compound used for both H and C NMR
What are other advantages of using TMS?
● Has only 1 carbon environment so only 1 peak produced → high intensity (has 4C) so easy to detect
● Inert so won't react with sample
● Low boiling point so easily removed from the sample
● Non-toxic
● It's signal is away from all the other H signals
When does splitting/spin-spin coupling occur?
Neighbouring hydrogen atoms (3 or fewer bonds away, or on the adjacent carbon) affect the magnetic field of 1H atoms and causes their peaks to split
What is the n+1 rule?
If there are n inequivalent 'H atoms on the neighbouring carbon then the peak will split into (n+1) smaller peaks
Draw the splitting patterns for 0, 1, 2 and 3 inequivalent H atoms 3 bonds or less away
PIC
Why must solvents used for 1H NMR not contain any hydrogen atoms?
Signals from the solvent would swamp signals from the sample, as there is much more solvent than sample.
Which solvents are used?
Deuterated solvents: CDCl₃, D₂O, C₆D₆
CCl₄ - tetrachloromethane
Summarise what these indicate for 1H NMR:
Number of signals: One main signal for each set of inequivalent 'H atoms (for each hydrogen environment)
Chemical shift: Larger • for 'H atoms closer to electronegative atoms or C=C
Splitting: Number of smaller peaks = 1 + number of inequivalent hydrogen atoms 3
bonds away
Area under peak: Proportional to the number of atoms represented by that peak
Why does the peak from
O-H bonds disappear if D2O is used as a solvent?
O-D bond is formed in preference to O-H due to labile protons that move/swap from one molecule to another.