CHEM207 Exam 3
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
Alkene Hydrohalegonation REAGENTS
HX
Alkene Hydrohalegonation
markovnikov X addition
enant
Radical Alkene HBr Addition
HBr, peroxides
anti-markovnikov
Initiation, propagation, termination
enant
Alkene Hydroboration-oxidation REAGENTS
1) BH3, THF 2) H2O2, -OH
Alkene Hydroboration-oxidation
syn addition of H and BH2
less sub OH
enant
Alkene Oxymercuration REAGENTS
1) Hg(OAc)2, H2O 2) NaBH4, -OH
Alkene Oxymercuration
more sub OH
backside attack of H2O breaks ring
no mechanism for step 2
enant
Alkene Halogenation REAGENTS
X2
Alkene Halogenation
anti addition of Xs
backside attack with X- breaks ring
enant
Halohydrin REAGENTS
X2, H2O/MeOH/RSH
Halohydrin
anti X, OH addition
backside attack of nuke
enant
Oxidation with peroxycarboxylic acids REAGENTS
1) mCPBA 2) trace acid
mCBPA
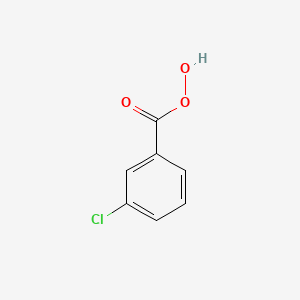
Oxidation with peroxycarboxylic acid
on more sub alkene
trans glycol
epoxied after step 1
nuke—>more sub C
enant
Syn-dihydroxylation REAGENTS
1) OsO4 2) H2O
Syn-dihydroxylation
cis glycol
no mechanism for hydrolysis
enant
Ozonolysis REAGENTS
1) O3 2) Me2S (red) / H2O2, H2O (ox)
Ozonolysis
red: aldehydes/ ketones
ox: carboxylic acids/ ketones
2X on alkane —> R=_H
1) NaNH2 (excess), NH3 (l)
2) H3O+
E2
R=_H —> R=_: (-)
NaNH2, NH3 (l) / NaH / THF
Sia2BH
