Introduction to Pharmaceutical Analysis: Extraction Techniques
1/18
Earn XP
Description and Tags
This set of flashcards covers extraction techniques in pharmaceutical analysis, including solid-liquid and liquid-liquid extraction, supercritical fluid extraction, and the principles and materials used in solid phase extraction (SPE).
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
Extraction Techniques
Methods used to separate analytes from materials in a formulation matrix, often using a solvent in which the analyte is highly soluble but matrix components have limited solubility.
Saxis alba L. (White willow)
A natural source used for the solid-liquid extraction of Salicillin, which is associated with Aspirin.
Papaver somniferum L. (Opium poppy)
A natural source from which Morphine is extracted through solid-liquid extraction.
Soxhlet extractor
An apparatus used for the continuous solid-liquid extraction of compounds.
Partition coefficient (P)
A measure of extraction efficiency
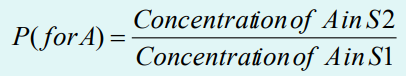
liquid-liquid extraction
place analyte solution solution (S1)
add S2, stopper the funnel, shake the phases and allow to settle
drain the lower layer into a beaker stopcock
remove the upper layer by pouring out of the top of the funnel
evaporate the solvent using vacuum and/or heat, or gas flow
Organic Acids and Bases Solubility
Free unionised acids and bases are generally soluble in organic solvents, whereas their salt forms are highly water soluble. Approximately 20% of drugs are weak organic acids and 75% are weak organic bases.
Liquid-liquid extraction for an organic base
direct removal of neutral and acidic excipients
acidic excipients left behind in aqueous layer
neutral excipients left behind in organic layer
Supercritical Fluid (SCF)
A substance above its critical temperature and pressure that exhibits gas-like viscosity and liquid-like density.
Carbon dioxide (CO2) in SFE
The most frequently used supercritical fluid because it is non-toxic and non-flammable, with a critical temperature of 31.1∘C and a critical pressure of 73.8bar.
Conditioning
The first step in Solid Phase Extraction (SPE) where the adsorbent is prepared.
Sample Addition
The second step in SPE where the analyte is "dead-stopped" on the medium using a solvent of low eluting power.
Washing
The third step in SPE where the medium is washed with solvents of low eluting power to remove contaminants/interferents.
Elution
The final step in SPE where the analyte is removed using a small volume of solvent with high eluting power.
SPE Cartridge Adsorbant
The material inside an SPE cartridge, typically consisting of 40-50μm particles and weighing between 50-500mg.
Styrene-divinylbenzene polymer gels
SPE adsorbents that are more lipophilic and have a higher loading capacity than coated silica gels.
Borate gels
Selective SPE adsorbents used for 1,2- and 1,3-diols, such as catechols and glycosides.
Immunoaffinity gels
SPE adsorbents where a ligand with high affinity for a target analyte, such as an antibody, is attached to the surface.
Molecularly Imprinted Polymers (MIPs)
Organic polymers designed with pre-determined selectivity for specific analytes, often referred to as "plastic antibodies."