Classification and Identification of Aldehydes and Ketones Lab 6
1/5
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
6 Terms
Chromic acid
Chromic acid will only oxidize aldehydes and not ketones. The solution will turn blue-green if it is an aldehyde.
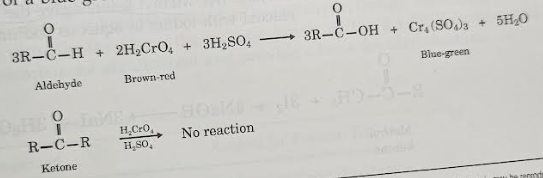
Tollens test
forms silver mirror when reacted with aldehydes. The aldehyde turns into a carboxylic acid. You have to make the tollens reagent with Tollens A, Tollens B, and ammonia bc it is explosive when dry. Also the reaction had to be done under warm conditions
iodoform test
Only reacts with methyl ketones. It will form a yellow iodoform/haloform precipitate. Turns methyl ketone into carboxylic acid. The NaOH will attch to the carbonyl carbon and then the CH3 will be kicked off, then the CH3 will attach to the Iodine to create the precipitate.
2-4 Dinitrophenylhydrazine test
Will react with both ketones and aldehydes. Will create a yello-red precipitate. the NH2 will replace the O from the carbonyl and then be double bonded to the C to make and imine?
identification by forming a derivative
sing a known reaction to convert it into another compound that is known. Then the melting point of that compound will be based on its starting material. This will tell us specifically what the starting material is.
what reactants did we test
isovaleraldehyde, benzaldehyde, 3-pentanone, acetone, and unknown