Phase 2 Reaction
1/49
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
50 Terms
Phase II Reactions
conjugation reactions:
– sulfate conjugation
– glutathione conjugation
– glucuronidation
– acetylation
– methylation
– amino acid conjugation
Sulfate Conjugation
makes sulfate ester
catalyzed by a dozen of sulfotransferases (SULTs)
uses PAPS as a high energy sulfonate donor
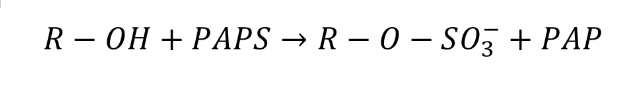
SULT
locates in cytosol of liver, GI mucosa and kidney
modulate the activities of steroid hormones, nuerotransmitters, toxicants and drus
different sulfotransferases from species as well as related forms of sulfation enzymes from different species, rats and humans, differ in thir substrate specificities and tissue distributions
PAPS
3’-phospho-5’-adenylyl sulfate
high energy sulfonate donor
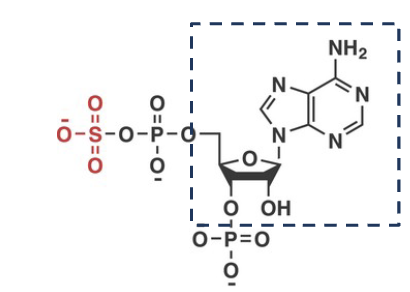
Products of Sulfate Conjugation
1. Sulfonated molecules are highly soluble in water and easily
excreted from the organism.
2. Electrophilic cation intermediates formation: Sulfate group is
electron-withdrawing and may be cleaved off heterolytically in
some molecules, leading to an electrophilic cation (+).
– an electrophile is a chemical species that forms bonds with
nucleophiles by accepting an electron pair.
DNA adduct
• Some sulfation reaction intermediates can spontaneously rearrange to release
sulfate and generate a reactive electrophilic cation molecule. Such electrophilic
molecules can react with DNA to form adducts.
• DNA adduct is a chemical covalently bond to a piece of DNA. This can interfere with
DNA replication, leading to toxicity and mutations.
– An electrophile is a chemical species that forms bonds with nucleophiles by
accepting an electron pair.
– An electrophilic cation is inherently reactive and can form covalent binding to
nucleophilic components, such as proteins, nucleic acids, and lipids.
– responsible for certain mechanisms of carcinogenicity and tumor
formation.

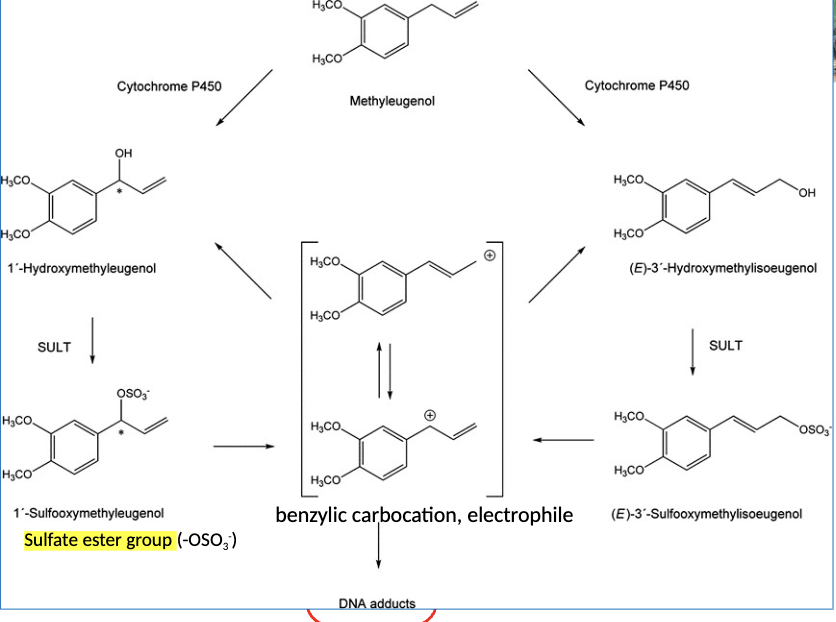
Sulfation of Methyleugenol
DNA adduct could be start of carcinogenesis
found as natural consituent in many plants
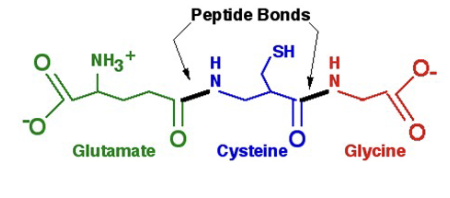
Glutathione
tripeptide that is one of the most important molecules in the
cellular defense against toxic compoundsIt is found in most cells but is especially abundant in the red blood cells
and in the liver (5mM)
Glutathione functions
antioxidant
forms glutathione conjugates to increase water solubility of the target substance
alpha peptide bond
covalent bond formed between two amino acids: They are
amide linkages formed between the carboxyl group (COOH) of one amino acid and the amino group (NH2) of another, releasing a molecule of water (H2O).
gamma peptide bind
first peptide bond that forms Glutathione is formed between the gamma-carboxyl group of the Glu side chain and the alpha-amino group of Cys
y-glitamyltranspetidase
only enzyme that can hydrolyze glutathione and are found on the surface of cells
Glutathione Conjugation
via glutathione s-transferases (GSTs) results in the formation of a glutathione-S conjugate
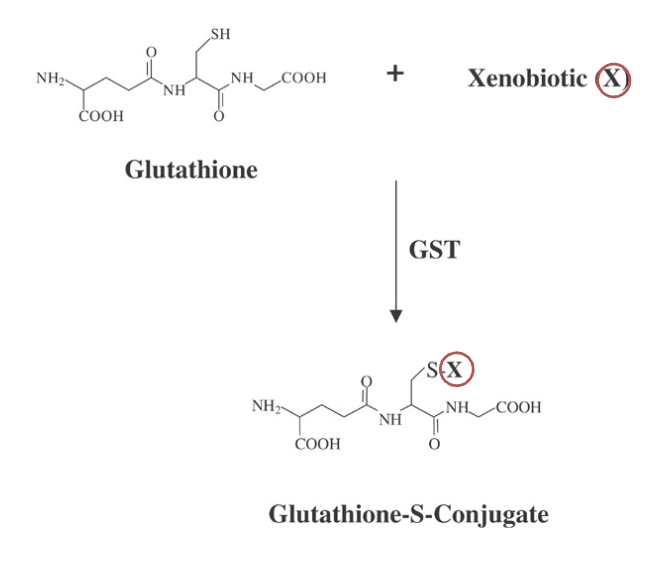
Glutathione Conjugation in Liver
1. Xenobiotics conjugated with glutathione include aromatic, aliphatic, heterocyclic and alicyclic epoxides, halogenated aliphatic and aromatic compounds, aromatic nitro compounds, unsaturated aliphatic compounds and alkyl halides
2. Reaction of the conjugation of the reduced form of glutathione (GSH) to xenobiotics is catalyzed by glutathione-S-transferases (GSTs)
3. GSH’s net negative charge and overall hydrophilicity greatly increases the aqueous solubility of the lipophilic moieties with which it becomes conjugated
Location of Glutathione
1. Mainly in cytosol
2. Some are found in the endoplasmic reticulum, i.e. microsomal
enzymes. Others are found in mitochondria
• Some are soluble proteins, while others are membrane bound
• Both types, the soluble GSTs and microsomal membrane bound
enzymes, exhibit a broad tissue distribution: they are found in liver,
kidney, brain, lung, heart, pancreas, small intestine, prostate, spleen,
and skeletal muscles
Glutathione and Excretion
1. Relatively more polar glutathione conjugates are eliminated into the bile.
2. Its molecular size (m.w. 307) ensures that its conjugated products are preferentially secreted via the biliary system which selects molecules of molecular weight greater than 300 to 500 according to the species, 325 ± 50 for rats and 500 ± 50 for humans.
3. In mammals, GSH conjugates are often further metabolized by hydrolysis and N-acetylation, either in the gut or in the kidney, to give N-acetylcysteinyl conjugates known as mercapturic acids, which are excreted in the urine.
increases bile and urinary excretion
Mercapturic Acid Formation
• For most compounds, mercapturic acid formation is a detoxication pathway, but the
precursor CysS-conjugates may undergo bioactivation to reactive intermediates
(Hashmi et al.1992; Anders and Dekant1998).
• The metabolites of endogenous substrates formed by the mercapturic acid pathway
may play a role in respiratory diseases, cancer, neurological disorders, and
cardiometabolic diseases that contribute to the pathophysiological development of
chronic inflammatory and metabolic disorders (
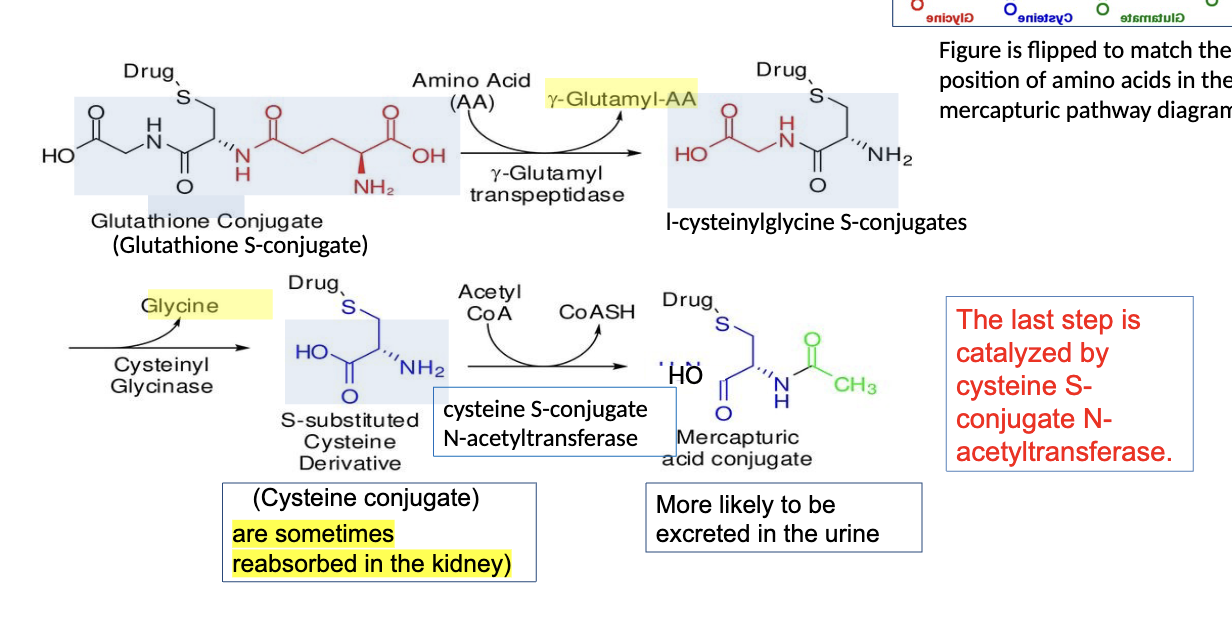
Cystein Conjugate and β-lyase
β-lyase metabolizes S-cysteine conjugates via both transamination and β-elimination.
The end products: pyruvate, ammonia, and a sulfur-containing fragment
The sulfur-containing intermediates are chemically reactive, and particularly toxic to kidney mitochondria.
The presence of β-lyase in the kidney makes some cysteine conjugated substances nephrotoxic.
• Example of the industrial chemical 4-bromobenzene Cysteine conjugate of 4-bromobenzenecan make aryl thiols that are very toxic
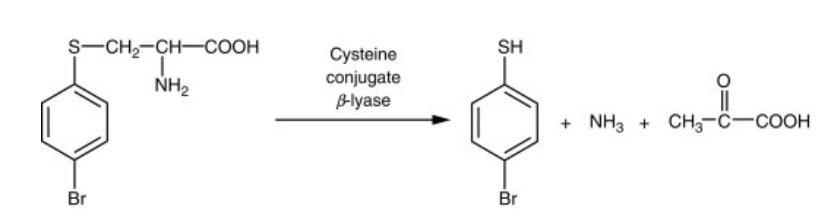
Toxifixation by β-Lyase
• β-Lyase is present in the cytosolic fraction of rat liver and kidney and also in the
microflora of the gut.
• Since thiols may be toxic and are more lipophilic than their cysteine conjugate
precursors, β-lyase reactions generally constitute a toxification pathway.
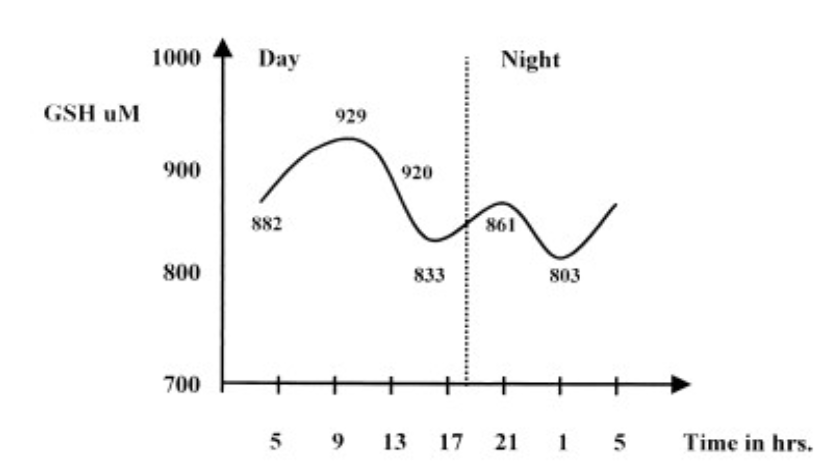
Glutathione circadian variation
A trend toward increasing GSH concentration at 9:00 AM and decreasing
concentration at 1:00 AM
Glucuronidation Reaction
• UDP-glucuronic acid (UDPGA): Activated glucuronic acid donor, as uridine
diphosphate glucuronic acid
• UGT: UDP-glucuronosyltransferase (enzyme)
• R–X–glucuronide: Conjugated (more water-soluble) product
• UDP: Uridine diphosphate (leaving group)
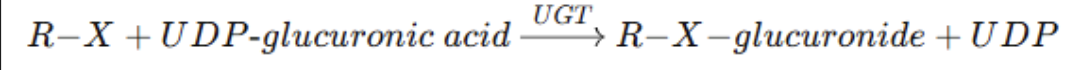
Glucuronidation
a major metabolic reaction, and mainly takes place in the
liver, for disposal of a variety of endogenous (such as Thyroid hormone) and
exogenous substrates (such as PCBs). It involves the transfer of glucuronic
acid from the cofactor UDP-glucuronic acid to functional groups, in
particular to nucleophilic functional groups, including hydroxyl groups (-
OH), amino groups (-NH2), or carboxyl groups (-COOH).
• PCBs/TCDDs are inducers of enzymes involved in glucuronidation.
• Unlike sulfotransferases (SULTs), UGTs have low specificity and high capacity.
• Glucuronic acid conjugation substantially increases the hydrophilicity of a
substrate enhancing its excretion.
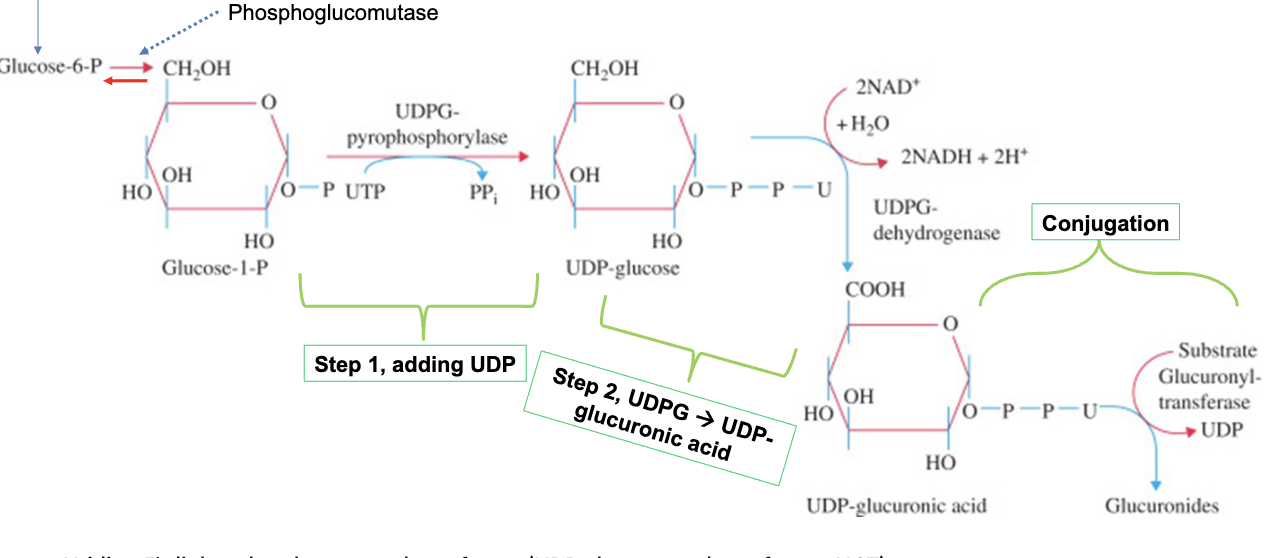
UDP-GA Formation
formed in cytosol from glucose-1-phosphate in 2-step reaction:
• 1st step is addition of uridine diphosphate (UDP) to glucose-1-phosphate
catalyzed by UDPG pyrophosphorylase
• 2nd step is conversion of UDP glucose to UDP glucuronic acid catalyzed by
UDPG dehydrogenase; oxidation of the substrate C6 alcohol into the
corresponding carboxylic acidx
Enzymes for Glucose to Glucuronide
1. Hexokinase:
– Glucose + ATP Glucose-6-P + ADP
2. Phosphoglucomutase:
– Glucose-6-P glucose-1-P⇌
3. UTP—glucose-1-phosphate uridylyl transferase, also known as glucose-1-phosphate
uridylyl transferase (or UDP–glucose pyrophosphorylase), is an enzyme involved in
carbohydrate metabolism. It synthesizes UDP-glucose from glucose-1-phosphate and
UTP:
– glucose-1-phosphate + UTP UDP-glucose + pyrophosphate⇌
4. UDP-glucose 6-dehydrogenase is a cytosolic enzyme, primarily occurs in the liver, also
found in kidneys and brain:
– UDP-glucose + 2 NAD+ UDP-glucuronic acid + 2 NADH + 2 H+
5. UDP-glucuronyltransferases (UGTs):
– UDP-glucuronic acid + substrate Glucuronide
Formation of UDP-GA and Conjugation
Billibrubin
lipid-soluble, orange-yellow, metabolite produced in the
liver by the breakdown of hemoglobin. It is practically insoluble in
water, mw 584.66. Glucuronidates billirubin is excreted in bile.
Billirubin toxicity
• Bilirubin (unconjugated and free bilirubin not bound by albumin) causes oxidative damage at the cellular level, causing oxidative stress through dysfunctions of endoplasmic reticulum, mitochondria, and plasma membrane.
• The most concerned biological effects of bilirubin are its harmful effects on the CNS of NBIs when it reaches high serum levels.
• In newborns the cell injuries could cause bilirubin encephalopathy and irreversible neurologic dysfunction, including hearing loss and even death. The threshold of bilirubin toxicity is lower in preterm or premature newborns.
• However, molecular biology studies in adults have evidenced that bilirubin is a cytoprotective antioxidant.
• Low to moderately high bilirubin levels account for a potent antioxidant.
• However, paradoxically, once certain threshold is exceeded, it is harmful for the developing brain.
• Different studies have demonstrated that oxidative stress is a key mechanism in bilirubin induced neurotoxicity.
• Bilirubin is lipid-soluble, passes through the membranes, and is incorporated to cells and organs. In the setting of intrauterine life, this characteristic allows bilirubin to cross the placental barrier from the fetus to the mother.
Billirubin threshold
• Bilirubin in blood is mostly bind to albumin. Only free indirect bilirubin (FIB) can pass through blood-brain barrier and cell membranes.
– Preterm newborns have lower albumin binding capacity.
– Conditions like hypothermia, hypoxia, acidosis, and asphyxia favor
neurotoxicity by breaking the binding and generating FIB.
– Certain drugs like ceftriaxone and ibuprofen compete with bilirubin for
albumin binding
– High lipid intake, > 1.5 g/kg, significantly reduces the bilirubin-albumin binding capacity and increases FIB levels in PNBIs born at less than 28 weeks of gestational age.
• Physiological hemolysis caused by oxidative stress due to the change from
placental to pulmonary oxygenation.
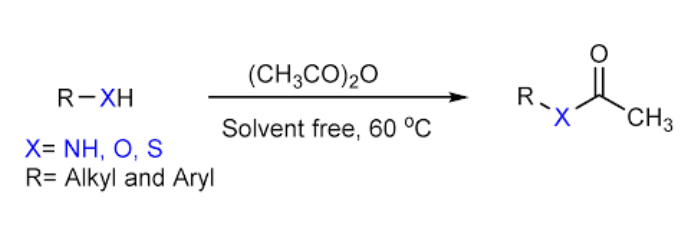
N-Acetylation
• Enzyme: N-acetyltransferases (NAT), two isozymes NAT1 and NAT2
• Cofactor: acetyl-coenzyme A (acetyl CoA)
• N-acetylation is a major route of biotransformation for xenobiotics
containing an aromatic amine (R-NH2)
• Unlike other Phase II reactions, acetylation masks an amine with
nonionizable group and are less water soluble than the parent
compound
present in mercapturic-acid conjugates
N-acetyltransferases
• Acetylation is catalyzed by cytosolic N-acetyltransferases with acetyl
coenzyme A as the required cofactor.
• This reaction is particularly important for compounds such as
aromatic amines
• The enzymes are found in most mammals with the exception of dogs
(and foxes), which are unable to acetylate xenobiotics.
Acetaminophen in cats and dogs
• Acetaminophen (APAP)-glucuronide was the principal metabolite excreted in the urine of dogs; its fraction of the total metabolites excreted in urine remained constant at the three dose levels
• Cat is at increased risk from APAP exposure because of impaired glucuronidation. In cats, APAP-sulfate was the major metabolite in urine at all three dosage levels, but the fraction of the total urinary metabolites represented by APAP-sulfate decreased as the dosage increased, suggesting saturation of its sulfate conjugation pathway.
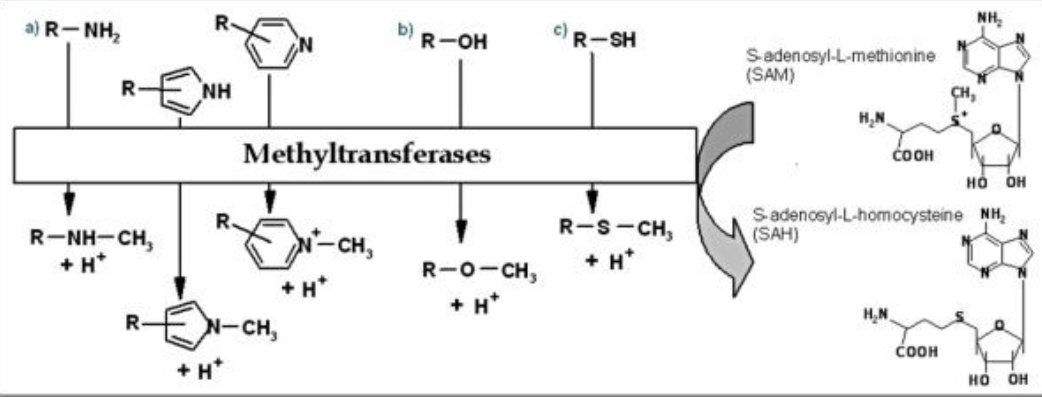
Methylation
adding a –CH3 group, does not dramatically alter the solubility of
substrates and results either in inactive or active compounds, requires the co-factor S-adenosylmethionine (SAM)
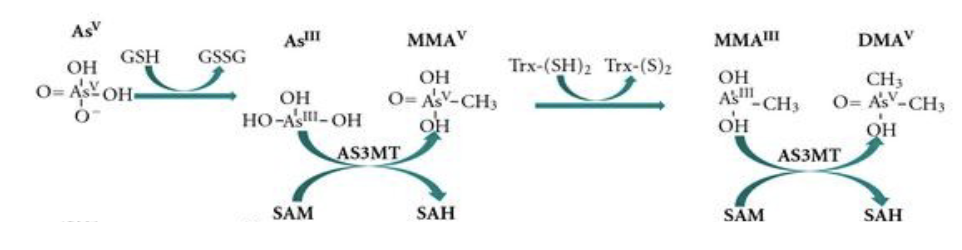
Methylation of Arsenics
• AsIII is most toxic, in terms of acute toxicity.
• As methyltransferase (AS3MT), cofactor SAM
• Monomethylarsonic acid (MMAv) to dimethylarsinic acid (DMAV)
– These two organic arsenic metabolites are not readily excreted.
• Thioredoxin (Trx) is a class of small redox proteins; AS3MT: arsenite
methyltransferase (human)
glycine
most common amino acid in amino conjugation
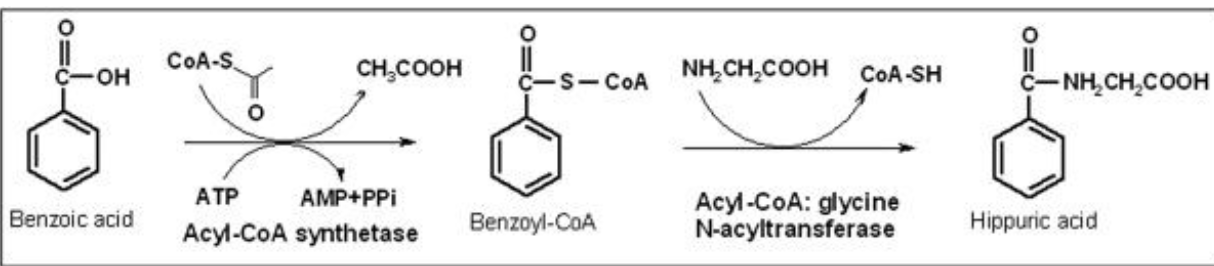
Hippuric Acid formation
the conjugation involves initial formation of a benzoyl acyl-CoA thioester that is then conjugated with glycine. this is done with benzoic acid.
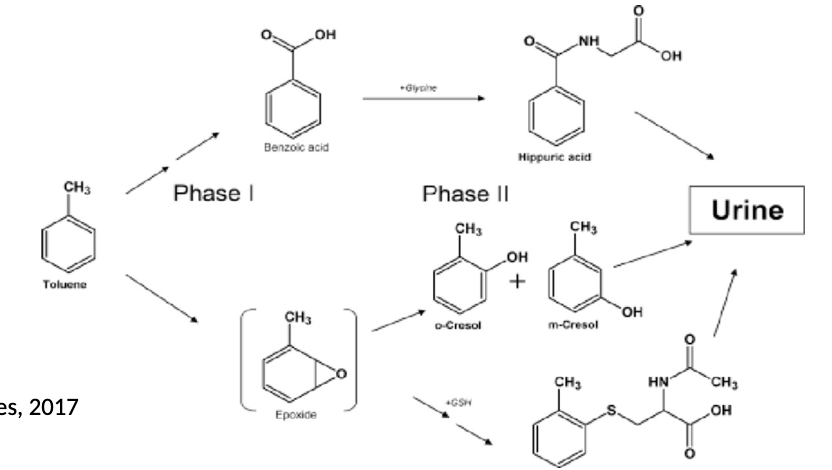
Toulene
Solvent in glues and household products.Biotransformation primarily involves liver enzymes, particularly cytochrome P450, oxidizing it to benzyl alcohol, then further to benzoic acid, which is then conjugated with glycine to form hippuric acid, the primary urinary metabolite.
cholic acid
major bile salt with conjugates of glycine and taurine that facilitates the absorption of lipids and lipid-soluble vitamins
Species variations in amino acid conjugates in bile
• In most species, including humans, primates, dogs, and rats, bile acids
can be conjugated with glycine and taurine.
• Rabbits and pigs form predominantly glycine-conjugated metabolites.
• Cats have limited ability to synthesize glycine-conjugated bile acids. In
cats, bile acids are almost exclusively (over 90%) taurine conjugates.
• The needs for taurine can be sufficiently synthesized by humans and
dogs, but cats have limited capacity to synthesize taurine.
• Taurine deficiency can lead to various disorders, such as blindness and
dilated cardiomyopathy
• While dogs utilize glycine conjugation, they are not inherently deficient in it; rather, they have a greater taurine synthetic ability and can adapt to using glycine for bile acid conjugation, unlike cats.
taurine deficiency
• Endogenously produced bile acids are mostly synthesized in the liver: they are taurine or glycine conjugates in humans.
• High capacity for taurine conjugates in cats and dogs.
• Both human and dogs can synthesize taurine, but not cats.
• Taurine deficiency:
– Mitochondria diseases
– Impairing the vision, retinal cells degeneration, such as feline taurine retinopathy.
– Central retinal degeneration (FCRD).
– Dilated cardiomyopathy (DCM), weakening of the muscle cells in the heart
– Taurine is a component of bile salts, and its deficiency may cause digestive
disturbances.
– Studies suggest that taurine deficiency might be a driver of the aging process.