Pharm 4b: Nucleic Acid/Protein Synthesis Inhibitors
1/85
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
86 Terms
Why is folic acid important in cells?
Needed for synthesis of DNA, RNA, and amino acids.
What molecule is part of folic acid synthesis and is targeted by sulfonamides?
Para-aminobenzoic acid (PABA).
What is sulfamethoxazole (SMX)?
A folate synthesis inhibitor and Dihydropteroate synthase - a PABA analog for folic acid
What is the mechanism of action of sulfamethoxazole?
Competitively inhibits dihydropteroate synthase and blocks conversion involving PABA toward folic acid synthesis.
Is sulfamethoxazole bactericidal or bacteriostatic?
Bacteriostatic.
What enzyme does trimethoprim inhibit?
Dihydrofolate reductase (DHFR).
What does DHFR normally do?
Reduces dihydrofolate to tetrahydrofolate.
What is the mechanism of action of trimethoprim?
Competitively binds and inhibits DHFR.
Is trimethoprim bactericidal or bacteriostatic?
Bacteriostatic.
Why are TMP and SMX used together?
They are synergistic, block sequential steps in folate synthesis, and lower the likelihood of resistance.
What is the brand name combination of TMP-SMX?
Bactrim.
What infections does TMP-SMX treat?
UTIs, respiratory tract infections, and GI infections.
What organisms are covered by TMP-SMX for UTIs?
E. coli, K. pneumoniae, and Proteus.
What organisms are covered by TMP-SMX for respiratory infections?
Streptococcus pneumoniae and Haemophilus influenzae.
What organisms are covered by TMP-SMX for GI infections?
E. coli, Shigella, and Salmonella.
What are the adverse effects of sulfamethoxazole?
Photosensitivity and nephrotoxicity.
What are the adverse effects of trimethoprim?
Bone marrow suppression, megaloblastic anemia, leukopenia, and black tarry or clay-colored stools.
How are quinolones commonly identified by name?
They end in -floxacin.
What do quinolones inhibit?
Nucleic acid synthesis, specifically one or both Type II topoisomerases.
What do quinolones inhibit in Gram-negative bacteria?
DNA gyrase.
What do quinolones inhibit in Gram-positive bacteria?
Topoisomerase IV.
Are quinolones bactericidal or bacteriostatic?
Bactericidal.
Why do fluoroquinolones have stronger activity than older quinolones?
Newer quinolones have fluoride added, enhancing bactericidal properties.
What are examples of fluoroquinolones?
Ciprofloxacin and Ofloxacin.
What are the main uses of fluoroquinolones?
UTIs and GI infections.
What organisms are covered by fluoroquinolones?
E. coli, Klebsiella, Campylobacter jejuni, Salmonella, and Shigella.
What morphology clue is attached to many fluoroquinolone targets?
Many are Gram-negative rods.
What is special about levofloxacin?
It covers usual UTI/GI organisms plus pneumonia organisms.
What additional organisms does levofloxacin cover?
Streptococcus pneumoniae, Mycoplasma, Chlamydophila pneumoniae, and Legionella.
What major adverse effects are associated with fluoroquinolones?
Tendinitis, tendon rupture, and peripheral neuropathy.
What age group should not receive fluoroquinolones?
Anyone under 18 years old.
What is rifampin derived from?
Rifamycin B; it is a semisynthetic derivative.
What is the mechanism of action of rifampin?
Inhibits bacterial DNA-dependent RNA polymerase and halts RNA synthesis.
What is rifampin mainly used for?
Tuberculosis in most patients.
What other infections can rifampin treat?
Mycobacterium avium complex and leprosy.
What body fluid color change is classically seen with rifampin?
Orange urine or sweat.
Who is rifabutin mainly used for?
HIV patients with TB.
How does rifabutin differ from rifampin in CYP effect?
Rifabutin is a less potent CYP450 inducer.
What is metronidazole?
A nucleic acid inhibitor and a prodrug.
How is metronidazole activated?
By pyruvate ferredoxin oxidoreductase (PFOR).
What organisms does metronidazole work against?
Anaerobes and protozoa.
Why is metronidazole selective for anaerobes and protozoa?
Eukaryotes lack PFOR, so the drug is not activated in them.
What is the mechanism of action of activated metronidazole?
Forms cytotoxic compounds that bind to DNA.
What protozoa are treated by metronidazole?
Giardia, Entamoeba, and Trichomonas vaginalis.
What anaerobic bacteria are treated by metronidazole?
Clostridioides difficile and Bacteroides.
What is the size of the bacterial ribosome?
70S
made up from 50s and 30s (math doesnt add up)
What makes up the bacterial 30S ribosomal subunit?
A single 16S rRNA molecule.
What makes up the bacterial 50S ribosomal subunit?
23S rRNA.
How are aminoglycosides commonly identified by name?
Often end in -mycin or -micin.
What aminoglycosides are specifically listed?
Streptomycin, Gentamicin, and Amikacin.
What infections are aminoglycosides used for?
Francisella tularensis and Yersinia pestis.
What is the mechanism of action of aminoglycosides?
Bind the 30S ribosomal subunit and inhibit protein synthesis.
Are aminoglycosides bactericidal or bacteriostatic?
Bactericidal.
What happens at low concentrations of aminoglycosides?
Ribosomes misread mRNA during elongation, inserting wrong amino acids into proteins.
What happens at high concentrations of aminoglycosides?
Complete inhibition of protein synthesis.
What is the aminoglycoside triad of adverse effects?
Ototoxicity, acute renal failure, and neuromuscular blockade.
What is important about aminoglycoside ototoxicity?
Can cause irreversible auditory or vestibular damage, damaging hair cells.
What serious outcome can aminoglycoside neuromuscular blockade cause?
Respiratory paralysis.
How are aminoglycoside adverse effects related to dosing?
Dose dependent and duration dependent.
How are tetracyclines commonly identified by name?
They end in -cycline.
What tetracyclines are most often used?
Doxycycline and Minocycline.
Why are doxycycline and minocycline commonly used?
Better patient compliance.
Are tetracyclines broad or narrow spectrum?
Broad spectrum.
What organisms are listed under tetracycline coverage?
Borrelia burgdorferi and Rickettsia rickettsii.
What is the mechanism of action of tetracyclines?
Bind reversibly to 16S rRNA.
Are tetracyclines bactericidal or bacteriostatic?
Bacteriostatic.
What is the age restriction for tetracyclines?
Avoid in children under 8 years old.
What are the classic tetracycline adverse effects?
Tooth staining, possible reduced skeletal growth, sun sensitivity, and increased sunburn risk.
What note was made about tetracycline tooth staining?
Current research suggests this may not be as absolute as once thought.
How are macrolides commonly identified by name?
They end in -thromycin.
What macrolides are listed?
Erythromycin, Azithromycin, and Clarithromycin.
What infections do macrolides treat?
- Pneumonia
- Mycoplasma
- Legionella
- Chlamydophila pneumoniae
- H. pylori with amoxicillin
- Chlamydia trachomatis
What is the mechanism of action of macrolides?
Bind the 50S ribosomal subunit and block the exit tunnel where peptides emerge.
What is the most common reason patients stop macrolides?
GI intolerance.
What is telithromycin?
A derivative of erythromycin, formerly classified as a ketolide.
How does telithromycin work?
Same mechanism as macrolides, with higher affinity for the 50S ribosomal subunit.
Who should not receive telithromycin?
Patients with myasthenia gravis, as it can worsen muscle weakness.
What is chloramphenicol?
A natural product from Streptomyces and a protein synthesis inhibitor.
What is the mechanism of action of chloramphenicol?
Binds 23S rRNA and acts on the 50S subunit.
Is chloramphenicol bactericidal or bacteriostatic?
Bacteriostatic.
What infections is chloramphenicol used for?
H. influenzae, N. meningitidis, Bacteroides, and Rickettsia species.
When is chloramphenicol usually used?
When no safer option is available.
Why is chloramphenicol rarely used?
High toxicity and can inhibit mitochondrial protein synthesis.
What are the major adverse effects of chloramphenicol?
Gray baby syndrome, aplastic anemia, and bone marrow damage.
Know the table of antibiotics and their MOA and types of infections
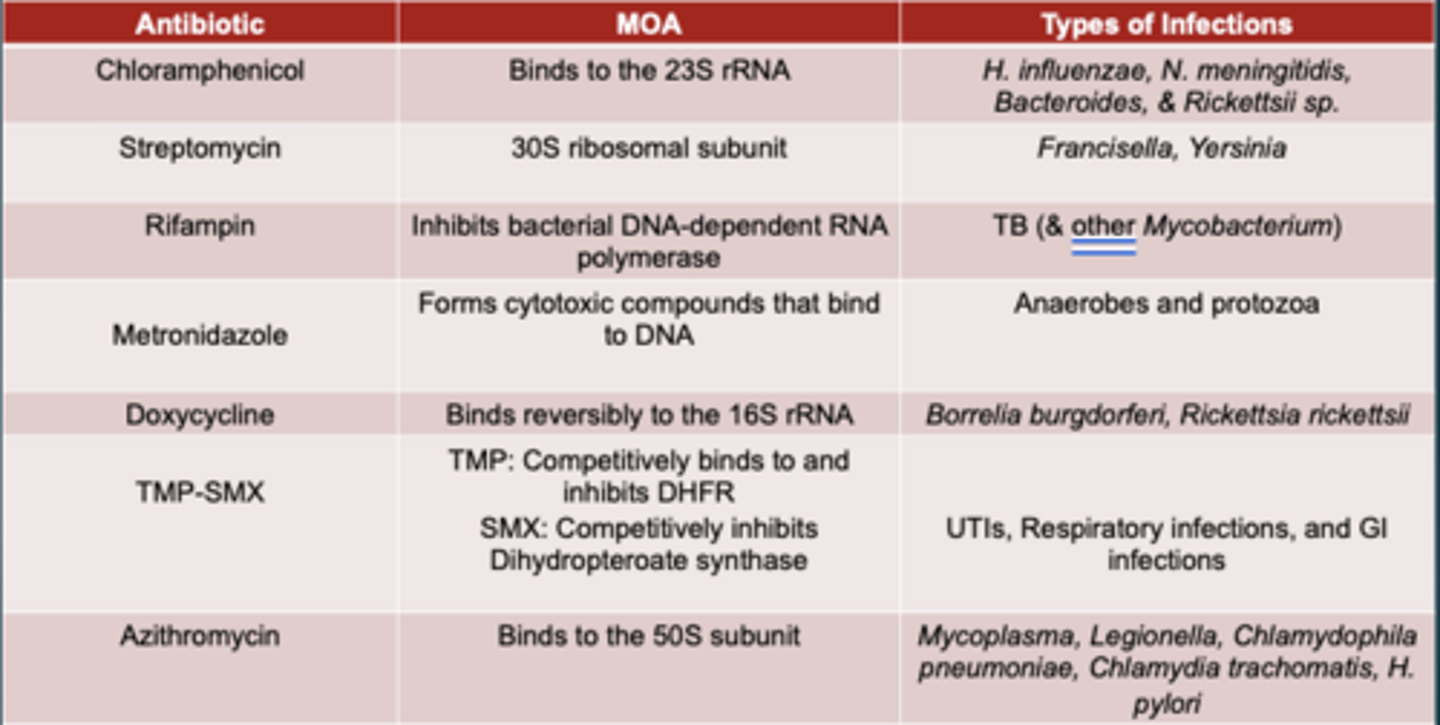
Match the adverse effect with the antimicrobial: chloramphenicol, tetracycline, aminoglycoside, and rifampin
