Everything deck
1/588
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
589 Terms
Ionic compounds
Formed when electrons are transferred from one atom to another
Forms ions with complete valence shells
The cations and anions are attracted to each other by strong electrostatic forces
Have high melting opints
Naming binary ionic compounds
Cation first, Anion second, add -ide suffix
Covalent bonds
Sharing one or more pairs of electrons so each atoms achieves a noble gas configuration
Coordinate bonds
Both shared electrons come from the same atom
2 Electron domains
Linear, 180°
3 electron doains
Trigonal planar, 120°'
bent or V-shaped (smaller than 120°)
4 electron domains
Tetrahedral, 109.5°
Trigonal pyramidal, 107°
Bent, or V-shaped 105°
Differences in single, double. triple bonds
The more bonds in one, the shorter and stronger
Boiling point
Liquid to gas
All attractive forces between particles are broken
Good indication of the strength of intermolecular forces
Melting point
Crystal structure is broken down, but attractive forces between particles exist
Impurities impact
Impact structure and result in lower melting points
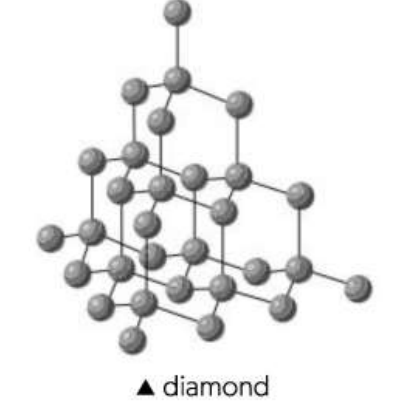
Diamond properties
Each carbon covalently bonded to four others
Giant tetrahedral diamond structure'
109°
1.54/1010 m long lengths.
All electrons are localized = Doesn’t conduct electricity
All bonds equally strong, so hard to break and high melting point
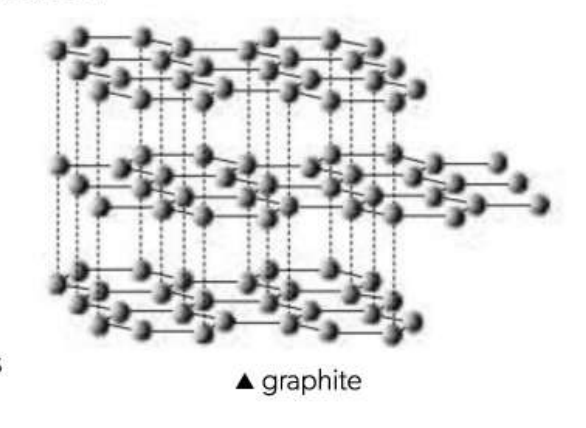
Graphite
Each Carbon is bonded to three other carbons
Layers of hexagonal rings
120°
1.42/1010 m
Weak attractive forces between layers = layers slide over each other = lubricant and waxy feeling
Between layers, electrons are delocalized = Good conducter of electricity
Graphene
One atom thick layer of Graphite
Extremely light
Semiconductor
200 times stronger than steel
Fullerenes
Large spheroidal molecules = hollow cage of sixty or more carbon atoms.
E.g. Buckminsterfullerene C60
Hexagons and pentagons → geodesic spherical structure similar to a football
Silicon
Giant tetrahedral covalent structure like diamond.
hard brittle solid
high melting point (4000°C)
metalloid
semiconductor.
Silicon dioxide (silica)
SiO2, quartz
Giant tetrahedral covalent structure similar to diamond and silicon.
Very hard and has a high melting point (1600°C)
No delocalized electrons = does not conduct electricity.
unipositive cation
positively charged ion with a charge +1
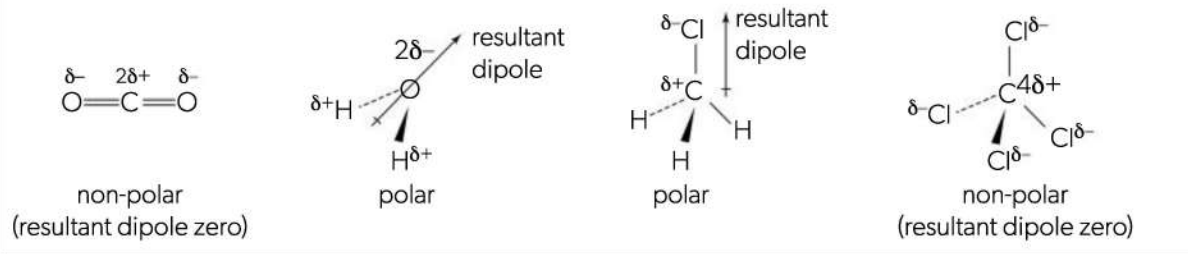
Bond polarity
More electronegative atoms exert greater attraction for electron pair(s)
One end of the bond will be more electron rich, resulting in bond dipole (polar bond)
Put number behind the atom with positive pole for how many bonds its pulling electrons with. E.g. NH3 would have Nitrogen with 3§+
Shape can also cause polarity.
Allotropes
Element can exist in different crystalline forms.
Different bonding and structural patterns, thus different chemical and physical properties
Intermolecular forces
Van Der Waals Forces
London Dispersion Forces
Dipole-Dipole Forces
Hydrogen Bonds
Van Der Waals Forces
More inclusive term for intermolecular forces where a temporary dipole occurs between both polar and non-polar molecules
London Dispersion Forces (LDP)
Between NONPOLAR molecules or atoms
Temporary dipoles
Can induce another dipole in a neighbouring particle
The strength of the dispersion forces increases with increased surface area/molar mass
→ When the carbon chain increases the boiling point increase
→ Branching decreases the surface area and thus the dispersion forces.
Dipole-Dipole Forces
Forces between POLAR molecules
Permanent dipoles
Attracted to each other by electrostatic forces.
Relatively weak, but the attraction is stronger than London dispersion forces.
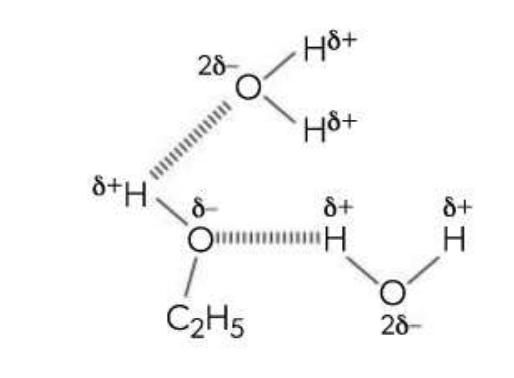
Hydrogen Bonds
Between POLAR molecules, that have hydrogen is bonded with a small, highly electronegative element
e.g. N-H, O-H or F-H
As the electron from hydrogen is pulled away, the proton attracts a non-bonding pair of electrons from the F, N, or O atom
Strong dipole-dipole attraction
The strength increases with increased number of hydrogen bonds formed.
Solubility
Maximum amount of a solute that can dissolve in a specific amount of solvent
"Like tends to dissolve like"
As the carbon chain lengthens in organic materials with polar ends, they become less soluble in water
Polar solvent example
Water
Non-Polar solvent example
Heptane or tetrachloromethane
Solvent for both non-polar and polar compounds
Ethanol
Requirements for conduction
Electrons or ions that are free to move
Covalent compounds (except some like graphite) = Not conductive
Metals = Contain delocalized electrons = Excellent conductors
Molten ionic salts = conduct electricity, but are chemically decomposed in the process
Metallic Bonding
The attraction that two neighbouring positive ions have for the delocalized electrons between them.
Valence electrons detach from atoms to create a closely packed lattice of cations in a "sea" of delocalized electrons.
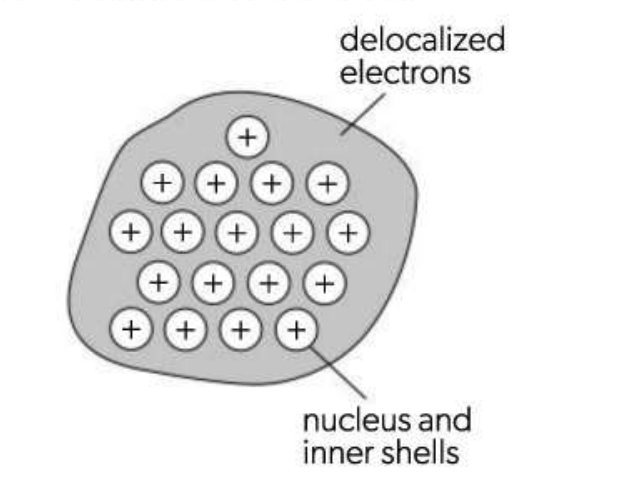
What does metallic bonding depend on?
The charge of the ions
Radius of the metal ion
Characteristics of metals
Malleable → bent and reshaped under pressure
Ductile → drawn out into a wire
= Because the closely packed layers of cations can slide over each other without breaking more bonds than are made.
Determining factors in metal melting point
Electron density (the size and charge of the ion formed) when the valence electrons are delocalized.
→ Smaller the metal ion formed = stronger the metallic bond and higher melting point.
The way in which the atoms are arranged in the solid metal
Most metals tend to have quite high melting points
Macroscopic properties
Observable, measurable characteristics of matter at a laboratory scale, visible to the naked eye or with low-magnification tools
Microscopic properties
Behavior, structure, and interactions of individual atoms, molecules, or ions, invisible to the naked eye. E.g., molecular geometry, bond energy, intermolecular forces
Three extreme cases for types of bonding
Caesium (metallic)
Fluorine (covalent)
Caesium fluoride (ionic)
Alloys
Homogeneous mixtures that are usually made up of two or more metals
Alters the properties of the metal → Distort the structure of the original metal as the bonding is less directional
Less ductile & less malleable than pure metals
Brass
Principle Metal: Copper
Added Metal: Zinc
Bronze
Principle Metal: Copper
Added Metal: Tin
Solder
Principle Metal: Lead
Added Metal: Tin
Pewter
Principle Metal: Lead
Added Metal: Copper, antimony, bismuth, or lead
Steel
Principal Metal: Iron
Added element: carbon
Extra: Adding chromium makes stainless steel, which has a much increased resistance to corrosion.
Polymers
Consist of monomers
Macromolecules with high molar masses
Subdivided into thermoplastics and thermosets
The longer the chain length of a polymer, the higher the strength and melting point
Cross-linking (or branching) and the orientation of the substituent groups can also affect particular properties
Thermoplastics
Many alkenes polermarize to thermoplastics
Soften when heated
Harden when cooled
Can be remoulded each time they are heated
Thermosetting polymers
E.g. Bakelite, polyurethanes, and vulcanized rubber
Form prepolymers in a soft solid or viscous state which, when cured, irreversibly turn into hardened thermosets
Elastomers
Flexible polymers
E.g. Rubber
Can be deformed under force but return to nearly their original shape once the stress is released.
Addition polymers
Long-chain molecules formed by joining many unsaturated monomer molecules (typically alkenes) together through a chain-reaction that breaks their double bonds
poly(2-methylpropene)
Butyl Rubber
Elastomer
Car tyre inner tubes and cling film
Polystyrene
Contains air trapped in the polymer
Light material
Good thermal insulator
Used in packaging as it has good shock absorbing properties
polymers
Long-chain, high-molecular-weight molecules
Large molecule
Covalent bonds
→ low electrical conductivity / insulators
→ low thermal conductivity
→ low density
→ inert — non-biodegradable
Properties of polymers
Light (made mostly of light elements C and H) & Durable (long so strong LDPs)
Longer chain → more LDF — high softening point & decomposition points
No delocalized electrons → thermal & electrical insulators
Strong intermolecular forces makes it insoluble in water & many non-polar solvents
No clear melting point since many contain chains of different lengths → Varying LDP
Environmental considerations
Reduce, Reuse, Recycle
Takes lots of energy to recycle
Plastics deteriorate when they are recycled
Natural polymers
Natural sources.
E.g. starch, lignin, silk, DNA, proteins…
Synthetic polymers
Made in laboratories.
E.g. plastics; PE, PET, kevlar, and synthetic textiles; nylon…
Polymerization
Chemical process that combines small molecules into long-chain, high-molecular-weight molecules.
Method of Polymerization
Polyaddition
Polyaddition
Double bonds will break and the monomer will form two covalent bonds with other monomers
Monomer

reactant
Repeating unit
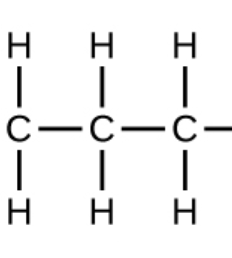
Naming of polymers
Prefix poly- and the name of the monomer (e.g. ethene = polyethene)
Amphoteric
Can be both an acid and a base
acyclic
chain
cyclic
ring
heterocyclic
ring with other atoms
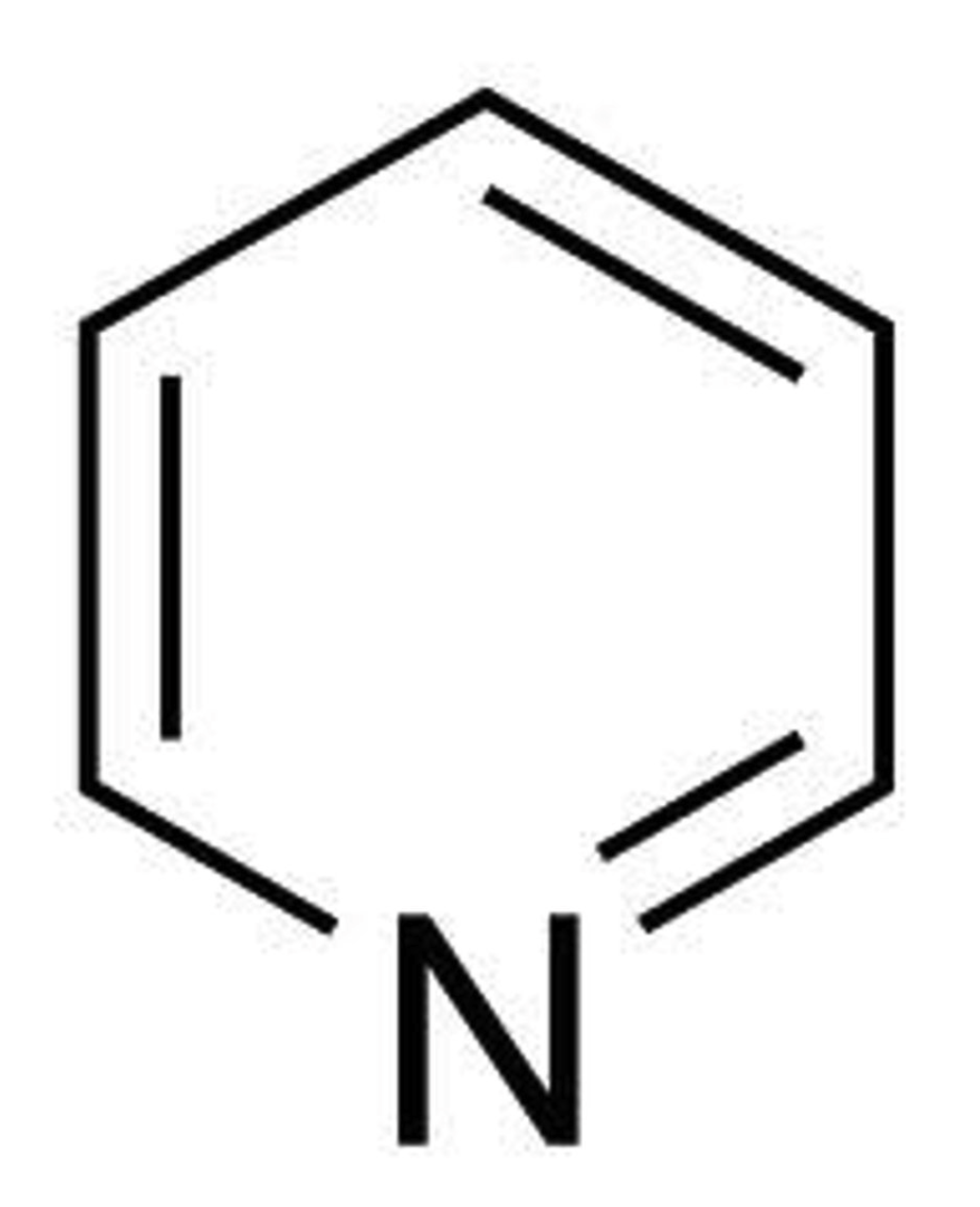
Saturated cmpound
Only single C-C bonds
Unsaturated compound
One C=C or C=-C bond
Alipahatic
No benzene ring
Polyunstaurated compound
Multiple C=C or C=-C bond
homologous series
series of organic compounds with the same general formula, but differ by one unit
- similar chemical properties but different physical
BP / MP Factors
Length (longer -> more LDF)
Branching (straight -> more LDF)
Functional group (-OH forms hydrogen bonds, others from dipole-dipole)
Stereochemical formula
3D formula of molecules bonds
Bond INTO plane
Dashed wedge
Bond OUT OF plane
Solid wedge
Structural isomer
Compunds with same molecular but different structural formula
Chain isomers
Different branching
Functional group isomer
Different functional group
Position isomer
Functional group is at different position
primary alcohol
OH bonded to C bonded to one chain
Secondary alcohol
OH bonded to C bonded to two chains
Tertiary alcohol
OH bonded to C bonded to three chains
Primary, secondary, tertiary amide
N is bonded to x amout of carbon chains
Homolytic Fission
Each of the two atoms forming the bond retains one of the electrons, resulting in the formation of two free radicals
Heterolytic fission
Both of the electrons in the shared pair go to one of the atoms, resulting in a negative ion and a positive ion
Covalent bonds break
homyltically or heterolytically
Free radicals
Signified with a dot
Contain an unpaired electron
Are very reactive
Alkanes reactivity
Relatively unreactive due to strong C-C and C-H bonds
Combust with oxygen
Substitution with Halogens in UV light
Mechanism of chlorination of methane
Initation
UV breaks the bond between two halogens as they are weaker than C-C or C-H bonds
Creates two free radicals
Propagation
Highly reactive Cl radical comes in contact with Methane, it produces H-Cl and a methyl radical
Highly reactive methyl radical reacts with Cl2 to produce chloromethane and sanother Cl radical
Innitiates chain reaction
Termination
Two radicals react together to form a non-radical
Cl radicals can still react with chloromethane to produce dichloromethane… etc.
Nucleophile
Electron rich species with min. 1 lone pair (attracted by +) which can create a coordination covalent bond
e.g. OH, water, ammonia
Nucleophilic substitution reactions
Heterolytic fission
(R-X) → R + X(-)
R + X(-) + Nu -> (R-Nu) + X(-)
Electrophiles
"Electron poor" species which can accept a coordinate bond
- Neutral but molecules or positive charge
Examples of electrophiles
H+, Cl+, NO2+, and CH3+
Hydrogen halides
Electrophilic addition reactions of alkenes
Unsaturated → Saturated
Leb test for presence of alkene
Add bromine, solution goes from yellow-ish to clear due to the electrohilic addition reaction
Hydration
Alkene + Water to form Alcohol
electrophilic addition
Double bond breaks to form 2 new bonds
1. halogens and alkenes
2. hydrogenhalades and alkenes
3. hydration (addition of water)
Corrosive