Lecture 31: Meat quality
1/18
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
Breaking down Z line
Done by proteases
Naturally present in muscle
Larger effect during post mortem
Specific proteases involved in meat tenderization
Calpain/Calpastatin system
Proteasome and caspase system
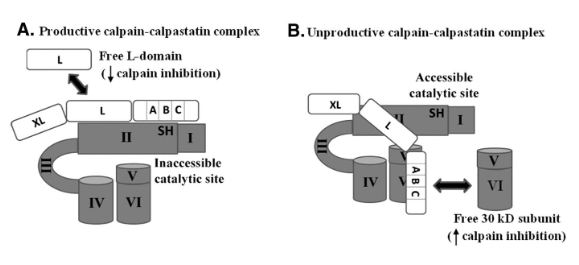
Protease: Calpains
Endogenous protease
Need Ca2+ for activity
Mew Calpain binds to 5-8 Ca ions
M calpain binds to 11-20 Ca ions
Optimal temp for best activity is 25 to 30 degrees celcius
pH for activity is 7
Inhibited by Calpastatin
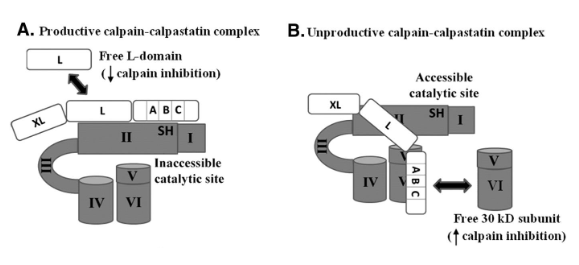
Protease: Calpastatin
Endogenous proteases
Also need Calcium ions for activity
Inhibits calpain by preventing calpain proteolytic activation
Causes conformational change in calpains and blocks active sites

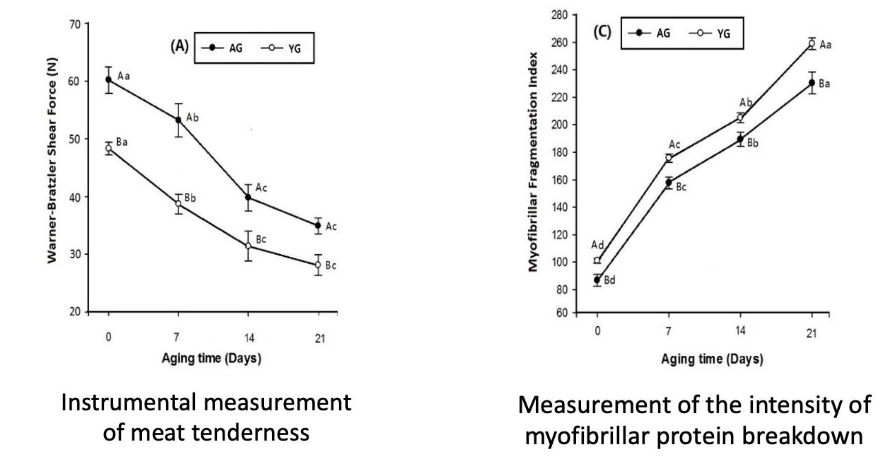
Tenderness evaluation: Warner Bratzler Shear Force
Assessing tenderness differences within given round muscle
Little loss of accuracy
Tenderness evaluation: Sensory panel testing
Methods for assessing tenderness differences based on individual perceptions
Aging and proteolysis
Instrumental measures of meat tenderness decreases
Measures of intensity of myofibrillar protein breakdown increases
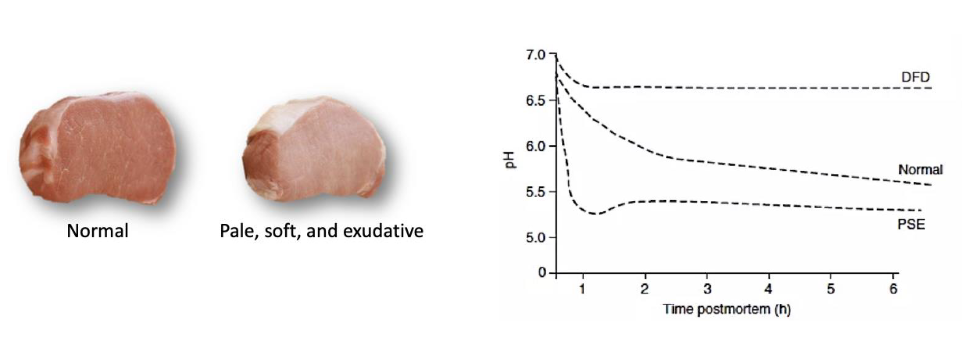
Meat colour
Primary quality trait evaluated by final consumer when purchasing

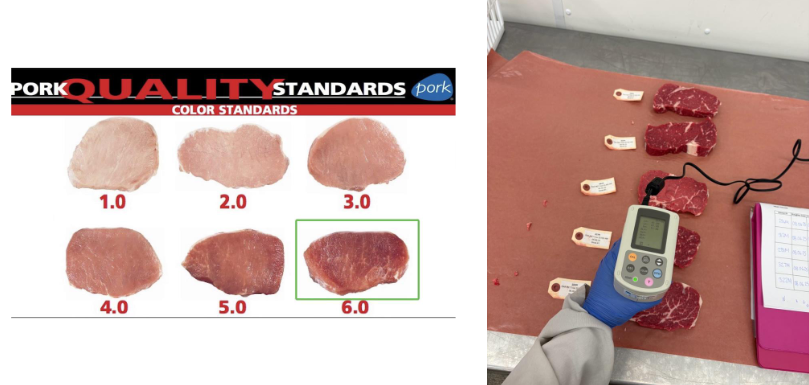
Factors of meat colour evaluation
Subjectively by meat graders
Instrumentally
Instrumental colour evaluations: a, b and L
L = whiteness
a = redness
b = yellowness
Instrumental colour evaluation: Negative values
a = intensity of green light reflectance
b = intensity of blue light reflectance
L = dark not much light reflectance
Instrumental colour evaluation: Positive values
a = intensity of red light reflectance
b = intensity of yellow light reflectance
L = higher intensity of light reflectance
Meat colour: Myoglobin
Varies depending on animal’s age
Muscles from older animals have more myoglobin contractions than younger animals
Why the meat is more tough, less tender
What affects myoglobin solubility
High temps and low pH
Causes denaturation of myofibrillar proteins
Cause denaturing of myoglobin
Decrease redness of meat
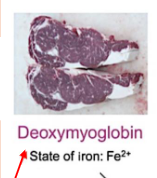
Lack of Oxygen
Iron is not bound to gas
High concentrations of deoxymyoglobin
Meat looks darker
Vacuum packaged meat
Lots of oxygen and iron in ferrous state
High affinity for oxygen
Form of myoglobin that is responsible for bright red colour
Observed in traditional retail meat packages
Oxygen permeable wrap
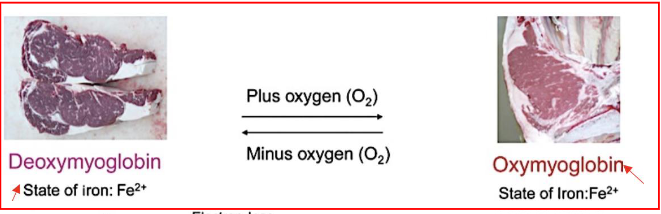
Both deoxymyogloin and oxymyoglobin
Iron ion remains in ferrous state (2+)
Change in colour is due to lack of oxygen
No changes in state of iron (stays 2+)
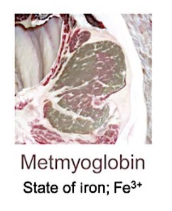
Metmyoglobin
Oxidation of heme iron results in transition from ferrous iron (2+) to ferric iron (3+)
Ferric iron does not have a high affinity for oxygen
Makes meat gray/brown
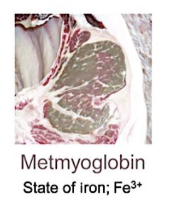
Why metmoglbin occurs
When meat has been exposed to environment for long period of time
Catalyzed by high temperature
Negatively impact retail sales