Chemistry topic 2 - Titration and S1.3
1/36
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
37 Terms
Solute
Substance being dissolved
Solvent
liquid in which dispersion occurs
Solution
homogenous mixture of 2+ substances
Dilute
solution containing small amount of substance
Concentrated
Solution with large amount of solute
Concentration
amount (moles) of substance in a given volume of solvent
Titration
Technique used to determine the unknown concentration of a solution by reacting with a solution of a known concentration
Isotope
Atom of an element, same number of protons but different number of neutrons, only changes physical properties
Volatile
Easily evaporated at normal temperatures
Ionization
Neutral electrically charged atoms/molecules connected to electrically charged atoms (ions) through gaining or losing electrons
Mass spectrometry
Analytical technique used to determine molecular mass of a compound and individually helped to prove the existence of isotopes
Ion
An atom or molecule with a net electrical charge
5 stages of mass spectrometry
vaporization, ionisation, acceleration, deflection, detection
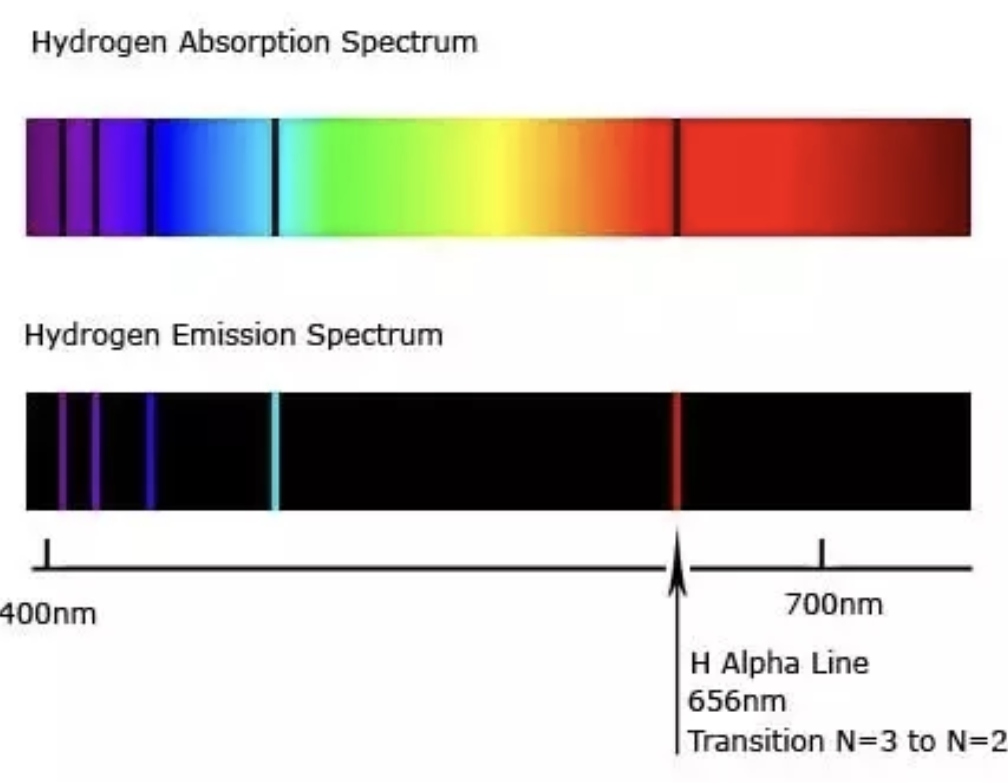
Continuous spectrum
Shows broad bands of electromagnetic radiation (emission) or shadow (absorption)

Emission line spectrum
contains only some discrete lines of electromagnetic radiation (emission)

Absorption line spectrum
or shadows (black lines superimposed on a continuous spectrum).

Hydrogen absorption/emission spectrum
If observed through a spectroscope each element gives a characteristic set of colours or lines, at fixed wavelengths.
First ionisation energy
Minimum required energy to remove an electron from an atom or a mole of electrons from a mole of atoms in its gaseous state
γ rays, x rays, UV, visible, IR, Microwaves, radiowaves - state 4 on left
left - high frequency, short wavelength, high energy, converge
Lyman series
Higher energy to n=1, viewed was UV radiation, highest energy transitions
Balmer series
Higher energies to n=2, visible light, medium energy transition
Paschen series
Higher energies to n=3 is IR, lowest energy transitions
Aufbau principle
When adding electrons to an atom, lowest energy levels are filled first
Pauli exclusion principle
Atomic orbital can only hold 2 electrons, and they must have opposite signs
Hunds rule
With degenerate orbitals, each orbital is filled with single electron before being doubly occupied (on bus)
Degenerate orbitals
Orbitals of the same energy
Strong nuclear force
Overcomes repulsion between positively charged protons, attracts subatomic particles such as protons and neutrons toward each other
Atomic radius
distance between nucleus and valence electrons
Convergence limit
frequency or wavelength that corresponds with ionisation
Successive ionisation energies
Energies required to remove more and more electrons from an ion that is becoming increasingly positive
He electron configuration
1s2
Ne electron configuration
1s2 2s2 2p6
Ar electron configuration
1s2 2s2 2p6 3s2 3p6
Kr electron configuration
1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6
Xe electron configuration
1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d10 5p6
Heisenberg uncertainty principle
not possible to know location and velocity of very small electrons, only probability that they will be in electron clouds 90% of time
shielding
the reduction in electrostatic attraction between the positive nucleus and outer-shell electrons, caused by the repulsive forces of inner-shell (core) electrons