Cell and Molec Bio (Exam 1)
1/407
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
408 Terms
What is the hierarchy of organizational levels? (5)
cell
multicellular
community
ecosystem
biosphere
Cell
an organized chemical system that includes many specialized molecules and subcellular structures
Multicellular Organism
an organism made up of many cells
Population
a group of organisms of the same kind that live together in the same place
Community
all of the populations of different organisms that live in the same place
Ecosystem
includes the community and the nonliving environmental factors with which it interacts
Biosphere
encompasses all the ecosystems of the Earth’s waters, crust, and atmosphere
Emergent Properties
new properties and rules emerge that cannot be predicted by full knowledge of a lower level
What is an example for an emergent property for a cell? (2)
a cell is alive if it is organized as a cell
as its individual components it is no longer alive and functional
Element
pure substance that cannot be broken down into simpler substances by normal chemical and physical processes
Atom
unit of an element
Protons (3)
subatomic particle with a positive charge
the number of protons in the core of an atom uniquely identifies the element
aka atomic number
Neutrons (2)
subatomic particle with no charge
exist in equal number to protons
How do you calculate the atomic mass?
proton # + neutron #
Electrons (2)
negatively charged subatomic particles
occupy 99% of the atom’s space, but accounts for little mass
Shells
orbit followed by electrons around an atom’s nucleus
Valence Electrons (2)
in the outer shell
determines an elements reactivity
Chemical Bonds
describe the chemical linkages between atoms that lead to formation of higher order structures containing more than one atom
How are chemicals formed and broken?
by chemical reactions
How are chemical reactions assisted in biological systems?
enzymes
Electronegativity (2)
value that describes the relative tendency of an atom to attract electron towards itself
determines an atom’s reactivity and type of bond formed between two atoms
What will an atom with 1 or 2 electrons in its valence shell want?
to lose them
What will an atom missing 1 or 2 electrons in their valence shell?
it will want to gain them
How can atoms get closer to having a full valence shell?
by sharing electrons
Ionic Bond
a bond between two atoms in which one has given some electrons to another atom
Covalent Bond
a bond formed when atoms share electrons
Molecule
entity with two or more atoms joined by covalent bonds
What are the two types of covalent bonds? (2)
nonpolar
polar
Nonpolar
equal electron sharing
Polar
unequal electron sharing
Intermolecular Forces
electrostatic attractions or repulsions between separate molecules
What are the important intermolecular forces for this class? (2)
dipole-dipole
hydrogen bonding
Dipole-dipole
force between polar molecules, where the partially positive end of one molecule attracts the partially negative end of another
Hydrogen Bond
hydrogen can be involved in such polarized covalent bond that it resembles an exposed proton and therefore a very positive surface
Water
a polar molecule with a number of emergent properties
What are the organizational properties of water? (4)
forms lattice of H-bonded molecules
can tolerate large changes in energy without huge temperatures
polar solvent
can dissociate into ions
pH
a measure of H+ ions relative to OH- ions
Why do we have buffers?
changes in pH can have dramatic consequences for living systems
Buffers
substances that compensate for changes in pH by absorbing H+ or OH- ions
Organic Molecules
those that include carbon
Hydrocarbons
molecules containing only carbon and hydrogen
Functional Groups
add specific properties to a molecule
What common reactions occur with water and functional groups? (2)
Condensation
Hydrolysis
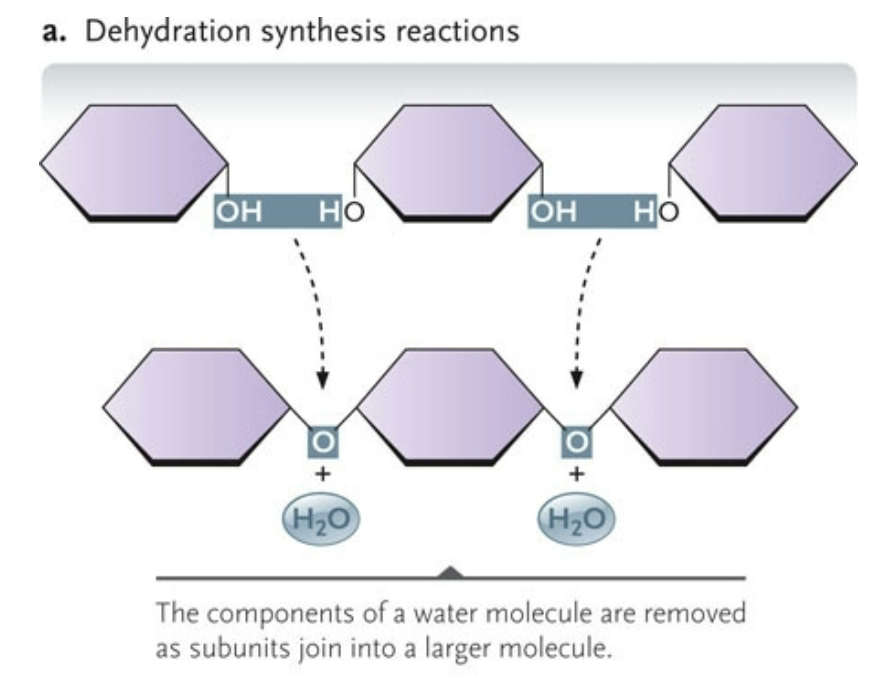
Condensation
the components of a water molecule are removed as subunits to join into a larger molecule
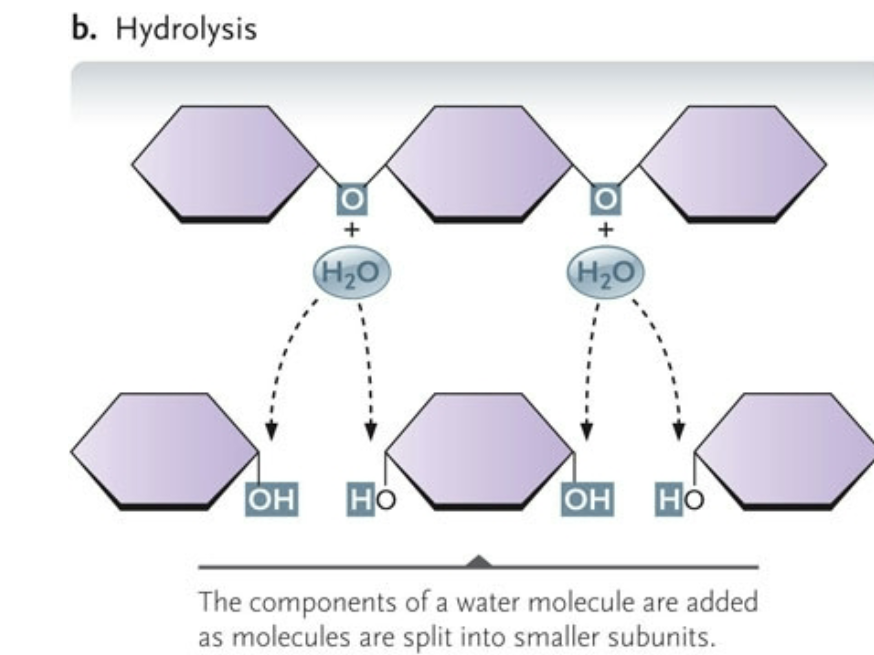
Hydrolysis
the components of a water molecule are added as molecules are split into smaller subunits
What are the types of macromolecules? (4)
carbohydrates
lipids
proteins
nucleic acids
Macromolecules (2)
made of atoms connected by covalent bonds
can interact with themselves and other molecules using intermolecular forces
Which macromolecules are polymer based? (3)
carbohydrates
proteins
nucleic acids
Polymer
a macromolecule assembled from smaller subunits through polymerization
Monomers
subunits of macromolecules
Monosaccharides (2)
monomer of carbohydrates
can exist in linear or ring form
What are carbohydrates used as? (3)
structural components
energy storage molecules
biochemical intermediates in making other molecules
Isomers
molecules with the same chemical formula, but different structure
Structural Isomers
same chemical formula, but atoms are connected in a different order
Aldehydes
carbonyl with a C and H
Ketone
carbonyl with two Cs
Stereoisomers (3)
same chemical formula and connectivity, but atoms are arranged differently in space around the central carbon
two molecules that are stereoisomers are mirror images
they have important biological implications
What kind of isomer is glucose?
stereoisomer
How is glucose a sterioisomer?
the OH on C1 can either be up or down
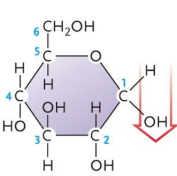
Is this an α or β glucose?
α
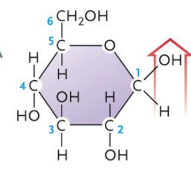
Is this an α or β glucose?
β
Disaccharides (2)
reaction between C1 and C4 in 2 monosaccharides yields two rings connected via covalent bonds
used for transient energy storage
What types of glycosidic bonds are there? (4)
1-4 linkage (more common)
1-2 linkage
1-3 linkage
1-6 linkage
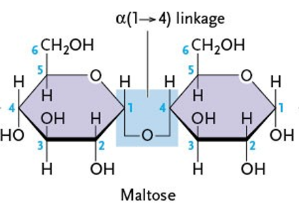
What type of linkage does maltose have?
α(1-4)
What are the components of maltose? (2)
glucose
glucose
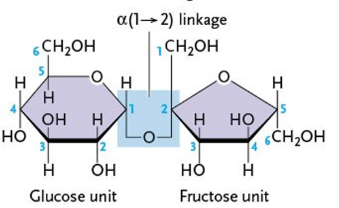
What is type of linkage does sucrose have?
α(1-2)
What are the components of sucrose? (2)
glucose
fructose
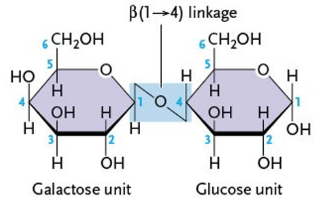
What type of linkage does lactose have?
β(1-4)
What are the components of lactose? (2)
galactose
glucose
Polysaccharides (3)
linear or branched polymers
used as energy storage molecules
important in building structures
Amylose (3)
Plant starch
Linear
α(1-4) linkage
Glycogen (3)
animal starch
branched
α(1-4) and (1-6) linkages
Cellulose (3)
primary plant cell wall components
linear
β(1-4)
Chitin (3)
structural component in insect exoskeleton and fungal cell walls
linear
β(1-4)
What is the monomer to chitin?
N-acetylglycosamine
N-acetylglycosamine
like a glucose molecule, but with N groups attached
Lipid (3)
water insoluble
nonpolar molecules composed of mostly hydrocarbons
amphipathic
Amphipathic
a molecule that contains polar and non polar regions
What are lipids used for? (3)
to form membranes
store energy
as biochemical intermediates
What are the types of lipids? (4)
fatty acids
triglycerides
phospholipids
sterols
Fatty Acids (2)
hydrocarbons of various lengths ~14-22
carboxyl group
How are fatty acids amphipathic? (2)
they have long hydrocarbon strings and a carboxylic acid, which makes a polar and nonpolar region
nonpolar is dominant
Saturated
all single bonds saturated with hydrogens
Unsaturated (2)
presence of one (mono) or multiple (poly) double bonds
a shape is imposed by double bonds
Triglycerides
three fatty acids bonded to a glycerol molecule by ester linkages
Ester Linkage
hydroxyl and carboxyl group bonded through a dehydration reaction
What are triglycerides used for? (2)
to store energy
triglycerides can store twice as much energy as carbs per gram
Phospholipid (2)
glycerol bound to two fatty acids along with another functional group linked through a phosphate
highly amphipathic
How does the phosphate change the character of this molecule? (2)
it makes it amphipathic
allows it to form special structures in an aqueous solution
What are the phospholipid head types? (4)
phosphatidic acid
phosphatidylinositol
phosphatidylserine
phosphatidylcholine
Steroids (3)
groups of lipids with structure based on framework of four carbon rings
differentiated by functional groups
important in signaling molecules operating at the level of the organism
Cholesterol (4)
steroid that is very important in biological membranes
embedded in the membrane
very rigid structure that enhances the selective barrier properties of the plasma membrea
keeps the plasma membrane in a packed and organized state
Which hormones demonstrate the significance of small differences in structure? (2)
estrogen and testosterone
important for sex determination in the development of mammals
Chiral
bound to 4 different entities
What gives rise to alpha and beta forms of glucose rings?
spontaneous ring interconversion
Protein
polymer made up of amino acid subunits
What are proteins used for? (3)
as structural molecules
enzymes
in biological processes
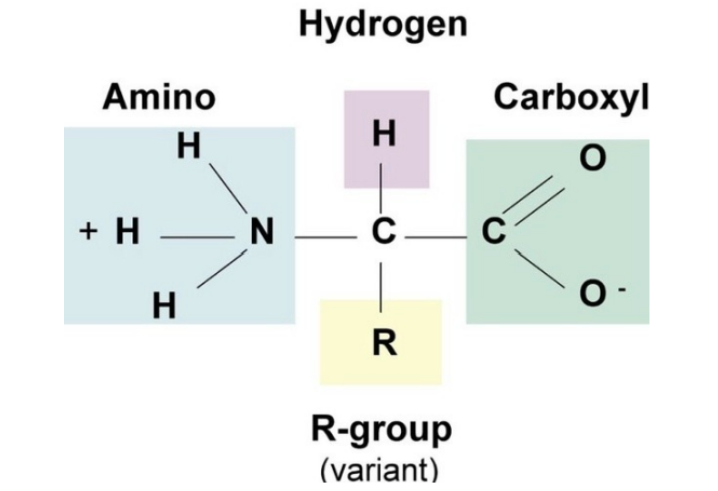
Amino Acids (3)
monomer of proteins
20 found in proteins
characterized by the different functional groups or side chains attached
How are amino acids used to form proteins?
they are polymerized into proteins by peptide bonds to form an amide bond
What does an amino acid contain? (5)
amino group
carboxyl group
hydrogen
R group
central carbon