DMI 115: Chapter 23 (Alder) and Venipuncture Combined
1/139
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
140 Terms
The use of contrast material as a means for visualizing human anatomy has a long history. Regardless of the type of contrast media, the purpose for using contrast media is to:
a. increase patient radiation dose and improve image quality.
b. enhance the low subject contrast of anatomic structures.
c. increase metabolism of the kidneys and liver.
d. improve the contrast between high contrast structures such as bone.
B. enhance the low subject contrast of anatomic structures
Pg. 318: Contrast are diagnostic agents that are instilled into body orifices or injected into the vascular system, joints, and ducts to enhance subject contrast in anatomic areas where low subject contrast exists.
Contrast agents can be generally classified as:
a. ionic and nonionic.
b. metallic and nonmetallic.
c. positive and negative.
d. aqueous and lipid based.
C. positive and negative
Pg. 318: Contrast media are generally classified as negative and positive…Negative contrast agents used in radiography decrease attenuation of the x-ray beam and produce areas of increased density on the radiograph, whereas positive contrast agents increase the attenuation and produce areas of decreased density on the radiograph.
Negative contrast agents:
a. appear white on images.
b. have low atomic numbers. (Low-attenuation + passed though easily)
c. provide high x-ray beam attenuation and absorption.
d. are materials such as bone and metal.
B.) have low atomic number
Pg. 318: X-ray photons are easily transmitted or scattered through radiolucent contrast media. As the name implies, these media are relatively lucent to X-rays.Because the anatomic areas filled by these agents appear dark (increased density) on radiographs, they are also called negative contrast agents. These media are composed of elements with low atomic numbers.
Think of the image like coloring with a black pencil:
If you press lightly, the paper looks light gray
If you press harder, the paper gets darker and darker
If you press REALLY hard, it becomes solid black
Negative contrast = DARK on the image
Positive contrast agents:
a. have high atomic numbers.
b. appear white on radiographs because of the degree of attenuation.
c. have a tendency to absorb x-ray photon energy to a great extent.
d. All of the above are correct.
D. All of the above are correct.
Pg. 318:
X-ray photons are absorbed by radiopaque contrast media because these media are opaque to X-rays. Because the anatomic areas filled by these agents appear light (decreased density) on radiographs, they are also called positive contrast agents. These media are composed of elements with high atomic numbers.
An air-contrast colon study would be an example of:
a. a high-contrast examination.
b. the use of both a negative and a positive contrast agent on the same image.
c. a barium sulfate and air examination.
d. all of the above.
D.) All of the above
Pg.318: In some instances, negative and positive agents are used together so that the lumen of organs, such as the colon, can be visualized or so that anatomic structures within a space,
such as the menisci of the knee, can be visualized.
An air‑contrast colon study uses:
Air → negative contrast
Barium → positive contrast
Together → double‑contrast
Double‑contrast = high‑contrast exam (because you have black vs white side‑by‑side)
For select examinations, air (negative contrast) and radiopaque contrast (positive contrast) are used as a pair to outline mucosal linings, joint interspaces, and passageways. These examinations are considered:
a. double-contrast studies.
b. extremely dangerous and rarely done.
c. single-contrast exams.
d. low-contrast studies.
A.) Double-contrast studies
Pg.319: Air alone provides negative contrast for laryngopharyngogrpahy because upper-respiratory tract structures contain air naturally. Otherwise, radiolucent contrast media are used in combination with radiopaque media to outline the lumens of, or spaces within, body structures.
Air + barium together = double‑contrast study.
All of the following are true of barium sulfate as a contrast agent EXCEPT:
a. when mixed with water it dissolves completely.
b. its atomic number (Z) is 56.
c. it is chemically inert.
d. to prevent flocculation, barium sulfate is often mixed with water and sodium citrate
A.) when mixed with water it dissolves completely
Pg.320:
-The element barium has an atomic number of 56 and is radiopaque.
-Because barium sulfate is
not soluble in water, it must be mixed or shaken into a suspension in water.
-In an acid environment, barium has a tendency to clump and come out of suspension, a process called flocculation.
-Stabilizing agents such as sodium carbonate or sodium citrate are usually used to prevent flocculation.
Think of sand in water:
Sand does not dissolve
If you stir it, it spreads out
If you leave it alone, it settles
water‑soluble = can dissolve in water.
Barium sulfate would be contraindicated (Should not used) for a patient with:
a. colonic polyps.
b. a suspected duodenal bulb perforation.
c. diverticulosis.
d. all of the above.
B. A suspected duodenal bulb perforation
Pg.320: If a patient is thought to have a perforation in the digestive tract, then barium sulfate is contraindicated because the body does not absorb barium sulfate naturally. If it enters the peritoneal or pelvic cavity, barium sulfate can cause peritonitis (means the belly lining is infected and very irritated.) and must be surgically removed.
Most adverse reactions to ionic, iodinated contrast agents are caused by:
a. their pH.
b. their hydrogen concentration.
c. their osmolality. (Higher)
d. the presence of iodine
C. their osmolality
Pg.322: Most adverse reactions to contrast media have been related to the osmolality of the media because the osmolality of a solution determines osmotic pressure, which controls
The movement of water in the body
Osmolality = number of particles in solution.
Ionic contrast dissociates (break apart) into a cation (+) and an anion (–).
More particles = very high osmolality.
High osmolality causes:
heat sensation
nausea
vomiting
vasodilation
pain
higher chance of adverse reactions
An undesirable characteristic of ionic contrast agents is their:
a. inertness.
b. neutral osmolality.
c. neutral pH.
d. dissociation into a cation and an anion when dissolved in plasma.
D.dissociation into a cation and an anion when dissolved in plasma.
Nonionic iodine contrast agents differ from ionic agents in that nonionics:
a. do not break down into a cation and an anion.
b. have a low pH.
c. have a small chemical structure and high osmolality.
d. are not triiodinated
A.) do not break down into a cation and an anion.
Pg.322: Efforts to decrease the many side effects of ionic iodine contrast media resulted in the development of molecules that do not dissociate in anions and cations (nonionic) or that ionic are too big to have osmotic effects such as ioxaglate (Hexabrix). These agents are identified as lower osmolality contrast.
The perfect iodinated contrast agent would have what characteristics?
1. High osmolality
2. Low osmolality
3. Nonionic chemical structure
4. Water solubility (hydrophilic)
5. Three or more iodine atoms per molecule
a. 1, 4, and 5 only
b. 1, 3, and 5 only
c. 2, 3, 4, and 5 only
d. 2 only
C.) 2,3,4, and 5 only
Pg.322:
-Recently, a nonionic dimer, iodixanol (VISIPAQUE), was introduced. Iodixanol is made to be osmolar (having the same number of particles) to blood plasma by the addition of electrolytes, small anions, and cations, which are normally present in the blood plasma.
-An additional advantage of the lower osmolality contrast media is that they are hydrophilic (water soluble) than higher osmolality contrast media. As a result, they may be less likely reactive with the cells that can trigger allergic effects.
-It is Triiodinated benzene ring and does not carry an acid group. Many oxygen-hydrogen hydroxyl groups surround the benzene ring. These groups increase the solubility of the media in blood plasma.
After the injection of an ionic iodinated contrast agent into a patient for a renal study, the patient complains of a warm feeling and is starting to itch. You notice the patient is developing urticaria. As you monitor the patient, you should:
a. casually watch the patient and note any more symptoms, as these are not unusual.
b. closely monitor the patient and look for worsening signs such as vomiting and bronchospasm.
c. inform the radiologist or physician of the patient's anaphylaxis. (Anaphylaxis = a very serious allergic reaction that affects breathing and blood pressure.)
d. do both b and c.
D. do both b and c.
Pg.323: Allergic reactions to water-soluble iodinated contrast media resemble allergic reactions to foreign substances, such as pollen grains. Reactions of typical allergic patients may be minor,
such as urticaria (hives). Some patients, however, experience wheezing and edema in the throat and lungs, with accompanying bronchospasm. Other anaphylactoid effects of water-soluble iodinated contrast media are nausea and vomiting. These patient reactions
can be typical but require close monitoring and communication with the radiologist.
Hives + itching = mild reaction → watch closely for worsening.
Laboratory tests to assess a patient's BUN and creatinine are used to determine the patient's:
a. liver function.
b. renal function.
c. pancreas.
d. thymus.
B. renal function
Pg.323: Increased Blood urea nitrogen (BUN) and creatine (waste product of metabolism) levels indicated that the patient may have renal disease and are good indicators for possible contrast medium induced renal effects.
Contrast media that are high in osmolality can be harmful to patients with compromised renal function. Patients who commonly have compromised kidney function include:
a. diabetics.
b. elderly patients.
c. renal dialysis patients.
d. all of the above.
D
What is “compromised kidney function”? (Simple words)
It means the kidneys are not working as well as they should.
Nonionic low-osmolality contrast agents have gained tremendous popularity with physicians as a safer choice for iodine contrast examinations. It is important to remember that nonionics:
a. are much less expensive than ionics.
b. still pose a risk to renal function.
c. are more difficult to inject owing to their higher viscosity.
d. can be used on all patients, regardless of cost and risk.
B. still pose a risk to renal function.
When using any type of contrast agent for a radiographic examination:
a. the patient should have the final choice on the type of contrast used.
b. a thorough medical history must be taken by the imaging professional.
c. the cost of the contrast agent is not a consideration.
d. anaphylactic reactions are rare and of little concern.
B.a thorough medical history must be taken by the imaging professional.
pg. 325: The most important patient care aspect before administration of water-soluble iodine contrast media is the patient history. The possibility of patient reaction is closely related to the
patient's disease state or age. The prevention of contrast media reactions depends on obtaining a thorough patient history that can indicate contrast media contraindications or an increased
likelihood of adverse reactions.
A radiopharmaceutical is:
a. a contrast agent.
b. any medication given to patients in radiology.
c. any medication given by a radiologist.
d. a radioactive material used for nuclear imaging with a gamma camera.
D.) a radioactive material used for nuclear imaging with a gamma camera.
Pg. 327: A radiopharmaceutical is not contrast agent. A radiopharmaceutical is a radionuclide that is attached (chemically bound) to a pharmaceutical that has a specific biodistribution in the human body…The radioisotope attached to the pharmaceutical is imaged using a gamma camera, which sees or detects where the radiopharmaceutical is in the body and forms an image of more or less concentration of the radiopharmaceutical.
A key distinction between radiographic studies and nuclear studies using radiopharmaceuticals is that:
a. nuclear studies are generally more functional in nature.
b. radiographic examinations are more structural in nature.
c. both studies use ionizing radiation.
d. all of the above are correct.
D. all of the above are correct.
Pg.327: The functionality of this procedure lies in the ability of many radiopharmaceuticals to be taken up by an organ of interest and then metabolized and used or eliminated body.
The responsible medical imaging professional will always maintain his or her focus on patients, particularly when any contrast agent is administered. A mandatory prerequisite to these studies is always:
a. laboratory tests to determine renal function.
b. a completed registration form.
c. a thorough medical history including allergies and diseases.
d. prepayment of the cost of the contrast agent.
C. a thorough medical history including allergies and diseases.
What is the normal range of blood urea nitrogen (BUN) values for adults.
A.) 4-8
B.)7-21
C). 10-20
D.) 15-30
B.) 7-21
What lab test evaluates the chemical byproduct of metabolism in the muscles?
a. Blood Urea Nitrogen (BUN)
b. Glomerular Filtration Rate (GFR)
c. Sedimentation rate d. Creatinine
d. Creatine
Correct answer: d. Creatinine
Creatinine is the chemical byproduct of muscle metabolism. It correlates with muscle mass and is an early indicator of impaired kidney function.
Which of the following creatinine mg/dL (milligrams per deciliter.) levels would be considered abnormal?
Noraml range: 0.7-1.5 mg/dL
a. 0.8
b. 1.0
c. 1.5
d. 2.0
Correct answer: d. 2.0
Normal creatinine range is 0.7–1.5 mg/dL
Anything above 1.5 mg/dL is strongly associated with impaired kidney function. So 2.0 mg/dL is clearly abnormal.
A glomerular filtration rate (GFR) test would provide information on which of the following?
a. Amount of creatinine in the blood
b. Amount of waste products that the kidneys can filter
c. Rate at which the kidneys filter waste from the blood
d. Rate of urine production
Correct answer: c. Rate at which the kidneys filter waste from the blood
GFR is calculated from creatinine levels and estimates how effectively the kidneys filter waste, making it essential for staging chronic kidney disease.
GFR = how fast your kidneys clean your blood.
What is the normal range for glomerular filtration rate (GFR)?
a. 90–120
b. 50–80
c. 20–40
d. 10–20
A GFR of 90–120
is considered normal. Values below 90 indicate decreased kidney function, and below 15 is associated with kidney failure.
Which of the following statements concerning kidney function is correct?
a. A high creatinine level is associated with a high GFR.
b. High BUN and creatinine levels are associated with a low GFR.
c. High BUN and creatinine levels indicate iodine contrast is acceptable.
d. Low creatinine and BUN levels mean the kidneys are filtering insufficient waste.
Correct answer: b. High BUN and creatinine levels are associated with a low GFR.
When creatinine and BUN rise, the kidneys are not filtering waste effectively, which means the GFR decreases. This situation is a contraindication for giving iodine contrast.
Which of the following lab tests would be a test relating to blood coagulation (blood clotting)? (Select all that apply)
a. Prothrombin time (PT)
b. Platelet count
c. Partial thromboplastin time (PTT)
d. International normalized ratio (INR)
Correct answers: a, b, c, d — ALL of them
PT & PTT measure how long it takes blood to clot.
INR is a standardized ratio used especially for patients on warfarin.
Platelet count is directly related to clotting ability:
High = clotting disorders
Low = bleeding disorders
What is the normal range for platelet count?
a. 1,000 – 4,000
b. 50,000 – 150,000
c. 150,000 – 400,000
d. 300,000 – 500,000
Correct answer: c. 150,000 – 400,000
Platelets are essential for clot formation, so their count is a key indicator of clotting ability. The normal range is 150,000–400,000 per mm³, and values below this increase bleeding risk.
A partial thromboplastin time (PTT) result of 50 seconds would indicate which of the following?
a. Shorter than normal blood clotting time
b. Normal clotting time
c. Longer than normal blood clotting time
Correct answer: c. Longer than normal blood clotting time
Normal PTT range is 25–35 seconds.
A value of 50 seconds means slower clotting, which may indicate excessive bleeding risk.
Low number = FAST clotting = more clotting risk
(blood clots too quickly)
High number = SLOW clotting = more bleeding risk
Which of the following would be indicated by a higher‑than‑normal prothrombin time (PT) or partial thromboplastin time (PTT) value?
a. The patient should not receive iodinated contrast media due to impaired kidney function
b. A procedure requiring a large spinal needle may need to be avoided
c. All injections should be completely avoided
d. All types of contrast media should be avoided
Correct answer: b. A procedure requiring a large spinal needle may need to be avoided. '
PT and PTT evaluate blood coagulation, and higher values mean excessive bleeding risk. Large‑lumen needles (like spinal needles) increase bleeding, so they may need to be avoided.
High PT/PTT = slow clotting = avoid big‑needle procedures.
. What is the normal range for a prothrombin time (PT) test?
a. 11–13.5 seconds
b. 15–25 seconds
c. 35–45 seconds
d. 50+ seconds
Correct answer: a. 11–13.5 seconds PT measures coagulation time. A normal PT is 11–13.5 seconds. Values above 13.5 seconds indicate slower clotting and increased bleeding risk.
12. What is the normal range for the international normalized ratio (INR) for reporting prothrombin time (PT)?
a. 0.8 – 1.2
b. 1.5 – 2.0
c. 2.0 – 2.5
d. 2.5 – 5.0
Correct answer: a. 0.8 – 1.2
The INR was created by the World Health Organization to standardize PT results across labs. A normal INR is 0.8–1.2. Patients on warfarin will have intentionally higher INR values.
Low PT/PTT/INR = FAST clotting = more clotting risk
(blood clots too quickly)
High PT/PTT/INR = SLOW clotting = more bleeding risk
Iodine contrast media is made less toxic and more tolerable to the human body by which of the following methods?
a. Removing the iodine content from the molecules
b. Binding the iodine atoms to ionic molecules
c. Binding the iodine atoms to a benzene ring
d. Breaking the molecule into individual ions
Correct answer: c. Binding the iodine atoms to a benzene ring
Attaching iodine to a benzene ring makes the molecule organic, more stable, and far better tolerated by the human body. This reduces toxicity and improves patient safety during contrast administration.
Iodine by itself is irritating and toxic to tissues.
When iodine is attached to a benzene ring, the molecule becomes:
organic
stable
non‑reactive
much less toxic
safe for injection
Which type of contrast media is the only one that is safe for intravenous (IV) injection?
a. Nonionic iodine
b. Ionic iodine
c. Benzene iodine
d. Water‑soluble iodine
Correct answer: d. Water‑soluble iodine
Water‑soluble contrast dissolves in blood and body fluids, allowing it to mix safely in the bloodstream. This makes it the only type appropriate for IV injection.
15. Which type of iodine‑based contrast media is less likely to cause adverse reactions when injected into a patient’s bloodstream?
a. Ionic
b. Non‑ionic
b.Non-ionic
More preferred
-When placed into a solution or fluid
-Molecule remains in one piece
-does not break apart
-no charged particles
-Has a low osmolarity that is closer to that of human blood
What is the main difference between ionic and non‑ionic iodine contrast media?
a. Non‑ionic solutions contain molecules that do not break apart upon injection
b. Non‑ionic solutions do not contain iodine
c. Non‑ionic solutions are considered more toxic to the body
d. Non‑ionic solutions are not water‑soluble
Correct answer: a. Non‑ionic solutions contain molecules that do not break apart upon injection.
Non‑ionic contrast does not dissociate into ions in the bloodstream, which makes it less reactive and better tolerated.
This is why non‑ionic contrast is associated with fewer adverse reactions.
Ionic contrast agent
-The undesirable contrast agent
-When placed in fluid the ionic contrast agent turn into multiple molecules (break apart first)
-forms charged particle in fluid
-higher toxicity
-higher osmolarity
Comparing ionic and non‑ionic iodine contrast media, which one is considered to have higher osmolarity?
a. Ionic
b. Non‑ionic
Correct answer: a. Ionic
Ionic contrast dissociates into more particles (ions) when injected, which increases the number of dissolved particles in solution.
More particles = higher osmolarity, which is why ionic contrast is more likely to cause reactions.
The term osmolarity refers to which of the following?
a. How a substance dissolves in water
b. The concentration of particles in a solution
c. Whether a molecule separates into ions in a solution
d. The thickness of a liquid
Correct answer: b. The concentration of particles in a solution
Osmolarity describes the number of dissolved particles in a given amount of solution. Higher osmolarity = more particles, which is why ionic contrast (which dissociates) has higher osmolarity.
Non‑ionic iodine contrast media will be less toxic to the body than ionic solutions due to which of the following?
a. Lower osmolarity
b. Stable chemical form that does not break apart into ions
c. Osmolarity closer to that of human blood
d. All of the above
Correct answer: d. All of the above
Non‑ionic contrast is less toxic because:
It does not dissociate into ions, making it chemically more stable.
It has lower osmolarity, meaning fewer dissolved particles.
Its osmolarity is closer to human blood, making it less reactive and better tolerated.
Which of the following are desired characteristics of iodine contrast media to make it appropriate for injection and to decrease the likelihood of adverse reactions? (Select all that apply)
a. Ionic form
b. Water‑soluble
c. Low osmolarity
Correct answers: b and c — Water‑soluble & Low osmolarity
Water‑soluble: All injectable contrast must be water‑soluble so it can mix safely with blood.
Low osmolarity: This is a property of non‑ionic contrast and is associated with fewer adverse reactions.
Ionic form is not desired because ionic contrast has higher osmolarity and causes more reactions.
Iodine contrast media that is described as non‑ionic is also associated with lower osmolarity.
a. True
b. False
Correct answer: a. True
Non‑ionic contrast does not break apart into ions, meaning it produces fewer particles in solution. Fewer particles = lower osmolarity, which is why non‑ionic contrast is better tolerated and causes fewer reactio
Recommendations for dietary instructions prior to an exam involving injection of iodine contrast media include which of the following?
a. No restrictions are recommended
b. Nothing by mouth (NPO) after midnight prior to the day of the exam
c. Nothing by mouth (NPO) for one hour prior to the exam only
d. Increased hydration the day before the exam and NPO for one hour prior to the exam
Correct answer: d. Increased hydration the day before the exam and NPO for one hour prior to the exam
The combination of good hydration the day before and short‑term NPO helps reduce adverse effects and improves patient tolerance during iodine contrast exams.
Increased hydration prior to an injection of iodine contrast media is recommended for what reason?
a. Decrease the chance of nausea and vomiting
b. Ease the burden on the kidneys when the contrast media is filtered
c. Lessen the chance of an anaphylactic response
d. Decrease the concentration of the contrast media after injection
Correct answer: b. Ease the burden on the kidneys when the contrast media is filtered
Hydration helps protect kidney function by diluting the iodine load as the kidneys filter the blood. This reduces the risk of contrast‑induced nephrotoxicity.
Which of the following lab tests are important for evaluating renal function before injection of iodine contrast media? (Select all that apply)
a. Creatinine
b. Complete Blood Count (CBC)
c. Glomerular Filtration Rate (GFR)
d. Blood Urea Nitrogen (BUN)
Correct answers: a, c, and d — Creatinine, GFR, and BUN
These three tests directly evaluate renal function, which is essential because the kidneys must be able to safely filter iodine contrast. CBC does not assess kidney function.
What is the normal range for blood urea nitrogen (BUN)?
a. 5 – 10 mg/dL
b. 7 – 21 mg/dL
c. 20 – 35 mg/dL
d. 30 – 50 mg/dL
Correct answer: b. 7 – 21 mg/dL
This is the typical normal range for BUN. BUN may increase slightly with age, and elevated levels are associated with decreased kidney function.
5. Which of the following is a byproduct of the metabolic activity in the muscles?
Creatine
What is the normal range for creatinine levels in a healthy adult?
a. 0.7 – 1.5 mg/dL
b. 1.5 – 3.0 mg/dL
c. 7.0 – 20.5 mg/dL
d. 20.0 – 35.0 mg/dL
Correct answer:
a. 0.7 – 1.5 mg/dL Creatinine should generally not exceed 1.5 mg/dL in most adults. Higher levels indicate the kidneys are not filtering waste effectively, which is critical to assess before giving iodine contrast.
Which of the following is the best description of the glomerular filtration rate (GFR) measurement?
a. Measurement of how much waste is filtered from the kidneys
b. Measurement of how much waste is left in the bloodstream
c. Estimate of the rate at which kidneys filter waste from the blood
d. Estimate of how long it will take for contrast media to be filtered from the blood
Correct answer: c. Estimate of the rate at which kidneys filter waste from the blood
GFR reflects how efficiently the kidneys are filtering, and it is calculated using creatinine, age, and gender. It is one of the most important indicators of renal function before giving iodine contrast.
What is the normal glomerular filtration rate (GFR) for an adult?
90-120
Premedication is required for which type of patient undergoing IV injection of contrast media?
a. Patient with diabetes
b. Patient with elevated BUN or creatinine
c. Patient with known allergy to iodine
d. Patient with chronic or acute renal failure
Correct answer: c. Patient with known allergy to iodine
Premedication (usually steroids + antihistamines) is used to suppress the immune response in patients with a known iodine allergy. Patients with impaired kidney function (elevated BUN/creatinine or renal failure) should not receive IV contrast at all, so premedication is not appropriate for them.
10. What drugs are commonly used as premedication to prevent allergic reactions to intravenous (IV) contrast in a patient with a known allergy?
a. Antihistamines and corticosteroids
b. Antihistamines and antibiotics
c. Corticosteroids and antibiotics
d. Analgesics and anti‑inflammatories
Correct answer: a. Antihistamines and corticosteroids
These two drug classes work together to reduce or prevent allergic reactions:
Antihistamines block histamine release
Corticosteroids suppress the inflammatory response
This combination is the standard premedication approach for patients with a known iodine contrast allergy.
During an intravenous (IV) bolus injection of contrast media, the injection of contrast into the tissues surrounding the vessel is called:
a. Absorption
b. Resorption
c. Extravasation
d. Infiltration
Correct answer: c. Extravasation
Extravasation = contrast injected outside the vessel into surrounding tissues.
It is most often associated with a direct injection, not an IV line.
Infiltration is similar but refers specifically to IV solution leaking from a catheter, not contrast from a direct injection.
Extravasation = contrast accidentally leaks out of the vein into the tissue
During an intravenous (IV) injection of contrast media, your patient complains of tightness and burning in the area of the injection. What should your immediate response be?
a. Place a cold compress on the area and continue the injection
b. Discontinue the injection and check for signs of extravasation
c. Ensure you are injecting into the vein and if so, continue the injection
d. Stop the injection and pull back on the plunger to remove what has been injected
Correct answer: b. Discontinue the injection and check for signs of extravasation
Tightness, burning, and swelling are classic signs of extravasation. Your immediate action is to stop the injection and assess the site for leakage of contrast into surrounding tissues.
Which two of the following are the recommended treatments for a patient who has experienced extravasation of intravenous (IV) contrast media? (Select two)
a. Elevation of the affected area
b. Cold or hot compresses
c. Administration of an antihistamine
d. A four‑hour observation of the patient
Correct answers: a and b — Elevation & Cold/Hot compresses
Elevation helps increase venous return and promotes resorption of the leaked contrast.
Cold or warm compresses help with pain relief and also support resorption.
Antihistamines are not indicated because extravasation is not an allergic reaction.
Routine four‑hour observation is not required unless the extravasation is severe.
Resorption = the body slowly absorbs the leaked contrast back into the bloodstream.
What is the most common reaction to iodinated contrast media?
a. Hives
b. Itching
c. Vomiting
d. Edema
Correct answer: a. Hives
Hives are the most common mild reaction to iodinated contrast. They fall under the mild reaction category, which also includes itching and limited rash, but hives occur most frequently.
1st level Mild reaction
-Scattered hives (the most common reaction to iodinated contrast)
-Pruritus (itching)
-Rhinorrhea (running nose)
-Coughing
Immediately after Mr. Miller receives an IV injection of iodinated contrast media, he complains of pruritus and you observe two hives on his neck. What should your response be?
a. No response is necessary since these are signs of a mild reaction
b. Monitor Mr. Miller’s vital signs and be alert for further reactions for at least 20–30 minutes
c. Recognize a moderate reaction and make sure that IV access is maintained
d. Recognize the onset of a severe reaction and alert the radiologist immediately
Correct answer: b. Monitor Mr. Miller’s vital signs and be alert for further reactions for at least 20–30 minutes
A few scattered hives + pruritus = mild reaction.
No treatment is required, but close monitoring is essential because mild reactions can progress.
Very important b/c a mild reaction can become a more serious reaction
-Monitor 20-30 min (Very important b/c a mild reaction can become a more serious reaction)
-Monitor vital signs
-Administered a medications like Antihistamine
Diphenhydramine (Benadryl)
Which of the following represents a moderate reaction to iodinated contrast media?
a. Scattered hives
b. Rhinorrhea
c. Pruritus
d. Persistent vomiting
Correct answer: d. Persistent vomiting
Scattered hives, rhinorrhea, and pruritus = mild reactions
Persistent vomiting is classified as a moderate reaction, which may require intervention and can delay or alter the exam sequence.
2nd level: Moderate reaction to iodinated reaction
-Diffuse Hives
-Persistent Vomiting
-Laryngeal edema (swelling of the throat)
-Tachycardia
Which of the following drugs would most likely be administered to a patient experiencing an allergic reaction to iodinated contrast media?
a. Diphenhydramine
b. Naproxen
c. Cephalexin
d. Hydromorphone
Correct answer: a. Diphenhydramine
Diphenhydramine (Benadryl) is a commonly used antihistamine, the drug class used to treat mild to moderate allergic reactions to iodinated contrast. The other options are not used for allergic reactions.
How should you respond to a patient experiencing a moderate reaction to iodinated contrast media?
a. Stop the injection and discharge the patient immediately
b. Monitor the patient, check vital signs, maintain IV access, and prepare an antihistamine
c. Begin CPR and call a code
d. Apply warm compresses and continue the exam
b. Monitor the patient, check vital signs, maintain IV access, and prepare an antihistamine
19. Which of the following drugs would most likely be used as emergency treatment for a severe allergic reaction to iodinated contrast media?
a. Benadryl
b. Diazepam
c. Epinephrine
d. Atropine
Correct answer: c. Epinephrine
Epinephrine (adrenaline) is the drug of choice for severe or life‑threatening allergic reactions. It rapidly reverses airway swelling, bronchospasm, and cardiovascular collapse.
How should you respond to a patient experiencing a severe reaction to iodinated contrast media?
a. Monitor the patient and prepare an antihistamine
b. Apply warm compresses and continue the exam
c. Stop the injection and observe the patient for 30 minutes
d. Call a CODE BLUE; the patient will receive epinephrine
d. Call a CODE BLUE; the patient will receive epinephrine
As you progress with an IV injection of iodinated contrast media, your patient states she is having trouble breathing and is visibly struggling to breathe. What should your immediate reaction be?
a. Call a code
b. Stop the injection
c. Take her vital signs
d. Prepare emergency medications
Correct answer: b. Stop the injection Stopping the injection is the first and most critical action.
Continuing to inject contrast would worsen the reaction and delay emergency treatment. Once the injection is stopped, you can escalate to emergency response steps as needed.
Which of the following lists the correct order of actions a radiographer should take when responding to any contrast reaction?
a. Assess ABCs → Call CODE BLUE → Prepare O₂ → Discontinue contrast
b. Acquire vital signs → Call for nurse → Prepare reaction kit → Discontinue contrast
c. Discontinue contrast → Call for nurse and physician → Assess ABCs → Acquire vital signs → Prepare reaction kit and O₂ → Be prepared to call CODE BLUE
d. Prepare O₂ → Discontinue contrast → Assess ABCs → Call for nurse and physician
c. Discontinue contrast → Call for nurse and physician → Assess ABCs → Acquire vital signs → Prepare reaction kit and O₂ → Be prepared to call CODE BLUE
ABC’s (airway, breathing, circulation)
As the radiographer, you are performing an IVP exam on Mrs. Jefferson. During your conversations and history‑taking with her, she seems alert and able to answer your questions. Why is it important to continue asking Mrs. Jefferson questions as you inject IV contrast media?”
a. To ensure the patient is aware of what you are doing
b. To ensure the patient is satisfied with their experience
c. To monitor the patient’s mental status, since changes can indicate a severe reaction
d. To make sure your history is complete
Correct answer: c. To monitor the patient’s mental status, since changes can indicate a severe reaction
Ongoing communication is a clinical assessment tool. A sudden change in the patient’s ability to answer questions or stay alert can be an early sign of a severe contrast reaction, making continuous verbal interaction essential during injection.
A decrease in renal function following administration of IV contrast media is called:
a. Renal failure
b. Nephrotoxicity
c. Nephrolithiasis
d. Iodine hypersensitivity
Correct answer: b. Nephrotoxicity
Nephrotoxicity is the term for contrast‑induced reduction in kidney function. It is more common in patients with pre‑existing renal impairment, dehydration, diabetes, or cardiac disease. This reaction can lead to kidney damage if not monitored and managed appropriately.
AI: Nephrotoxicity = kidney damage caused by a toxic substance.
In this case, the toxic substance is iodinated IV contrast.
So if kidney function drops after contrast, that’s nephrotoxicity.
Which of the following include a risk factor for nephrotoxicity?
a. Elevated BUN or creatinine
b. Diabetes
c. Heart disease
d.All of the above
Correct answer: d. All of the above
All three conditions — elevated BUN/creatinine, diabetes, and heart disease — increase the patient’s risk for contrast‑induced nephrotoxicity because each can impair renal perfusion or filtration. Patients with these risk factors require careful screening, hydration, and sometimes contrast dose modification.
Kidney damage caused by a toxic substance.
. Which of the following are characteristics of barium sulfate? (Select two)
a. It is water‑soluble
b. It is a suspension
c. It has a low anatomic number
d. It has a high atomic number
Correct answers: b. It is a suspension & d. It has a high atomic number
Barium sulfate is not water‑soluble, so it is not absorbed by body tissues.
It is administered as a suspension, not a solution.
Barium has an atomic number of 56, which is high, making it an excellent positive contrast agent.
Which statement about barium sulfate contrast media is true?
a. It is water‑soluble and can be used as IV injection
b. It attenuates radiation more than normal body tissues
c. It dissolves in solution
d. It has an atomic number similar to that of body tissues
correct answer: b. It attenuates radiation more than normal body tissues
Because barium has a high atomic number, it absorbs (attenuates) more radiation than surrounding tissues. This is why barium appears white on radiographs and functions as an excellent positive contrast agent.
Barium sulfate is used for what type of x‑ray examinations?
a. Urinary system
b. Gastrointestinal system
c. Vascular system
d. Endocrine system
Correct answer: b. Gastrointestinal system
Because barium sulfate is not water‑soluble, it cannot enter the bloodstream.
It stays within the GI tract, making it ideal for imaging the:
Esophagus
Stomach
Small bowel
Large bowel
It is not used for urinary, vascular, or endocrine studies.
Which of the following methods for introduction of contrast media into the body would be safe for barium sulfate?
a. Intrathecal
b. Parenteral
c. Intravenous
d. Enteral
Correct answer: d. Enteral
Barium sulfate is only safe when administered enterally, meaning it must stay within the GI tract and never enter the bloodstream. Intrathecal, parenteral, and intravenous routes would be dangerous because barium is not water‑soluble and cannot be absorbed or transported by body fluids.
As the radiographer for a patient having an upper gastrointestinal (GI) exam, you notice that the patient history states that there is a possibility of bowel perforation. Which of the following situations would most likely be a result of this information?
a.Perform the exam as usual with no modifications
b. Perform the exam with a less toxic form of barium sulfate
c. Perform the exam with an iodine‑based contrast media for the GI tract
d. Do not perform the exam at all since no contrast media could be used
Correct answer: c. Perform the exam with an iodine‑based contrast media for the GI tract
Because barium sulfate is not water‑soluble, it must never be used when bowel perforation is suspected. If barium leaks into the abdominal cavity, it can cause severe inflammation and complications. A water‑soluble iodinated contrast is the safe alternative for suspected perforation.
For which of the following exams would barium sulfate be the contrast media of choice?
a. Intravenous pyelogram
b. Myelogram
c. Arthrogram
d.Small Bowel
Correct answer: d. Small bowel
Small bowel studies keep the contrast inside the GI tract, which is safe for barium.
All other listed exams (IVP, myelogram, arthrogram) require contrast to be introduced into spaces where it mixes with body fluids, which means barium cannot be used.
Barium is not water‑soluble, so it must never enter the bloodstream or spinal canal.
Barium sulfate contrast media is categorized as a suspension. Because of this fact, the radiographer must follow which practice regarding the use of it?
a. It must be kept chilled to keep the particles suspended in the liquid
b. It must be mixed with distilled water to evenly distribute the particles
c. It has very short shelf‑life since the particles will not dissolve in the liquid
d. It must be shaken or mixed prior to use to redistribute the particles within the solution
Correct answer: d. It must be shaken or mixed prior to use to redistribute the particles within the solution
Because barium sulfate is a suspension, the particles naturally settle to the bottom. Before administration, the radiographer must shake or mix the container to ensure the contrast is evenly distributed for accurate imaging.
What is the atomic number of barium?
a. 6
b. 15
c. 26
d. 56
Correct answer: d. 56
Barium has an atomic number of 56, which is significantly higher than the atomic numbers of normal body tissues. This high atomic number means barium absorbs more radiation, making it appear bright white on radiographs and providing excellent contrast in GI imaging.
What is the main reason barium is combined with a sulfate molecule for use as a contrast media?
a. It lessens the toxicity of barium by forming a non‑water‑soluble solution
b. It increases the attenuation characteristics of the barium
c. It improves the taste of barium for drinkable forms
d. It increases the water‑solubility of the barium so that it is less toxic
Correct answer: a. It lessens the toxicity of barium by forming a non‑water‑soluble solution
Pure barium is toxic if absorbed.
When bound to sulfate, it becomes barium sulfate, which is not water‑soluble and therefore not absorbed by body tissues.
This makes it safe for GI imaging while still providing excellent radiographic contrast.
Which of the following methods of introducing contrast media into the body is considered an enteral method?
a. Intravenous
b. Intrathecal
c. Oral
d. Intra‑articular
Correct answer: c. Oral
“Enteral” means the contrast passes through the intestines. Only oral administration enters the GI tract. All other options (IV, intrathecal, intra‑articular) are parenteral routes.
Which of the following represents the chemical formula for the barium sulfate compound used in radiographic imaging?
A. BaCl₂
B. BaSO₄
C. CaSO₄
D. NaI
B. BaSO₄
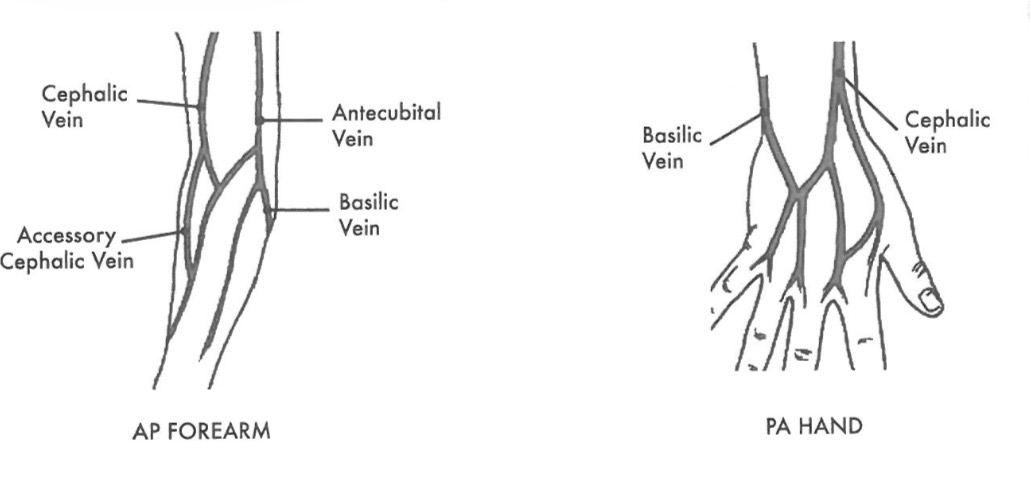
Sites for insertion of venous catheters or needles 4 type
What is the most common sit for injection and second?
1.Antecubital (most common site for injection
2.Accessory cephalic vein
3.Basilic (back of hand-second most common site for injection)
4.Cephalic vein
1. What is the primary purpose of administering contrast media during radiographic examinations?
A. To increase patient dose and improve spatial resolution
B. To artificially alter tissue attenuation and enhance visualization of specific structures
C. To decrease subject contrast and reduce image density
D. To eliminate the need for proper exposure technique
B. To artificially alter tissue attenuation and enhance visualization of specific structures
Supported by: “Through the introduction of contrast agents… the operator artificially alters the ability of selected organs and structures to attenuate the X‑ray beam… allowing enhanced recording of the contrast‑filled structures.”
Contrast = makes certain structures easier to see by changing attenuation.
1. Negative contrast agents typically involve the use of gases to do which of the following to the attenuating ability of the structures they fill?
A. Increase attenuation and increase radiographic density
B. Increase attenuation and decrease radiographic density
C. Reduce attenuation and increase radiographic density
D. Reduce attenuation and decrease radiographic density
c
Supported by: “Negative contrast agents… involve the use of gases to reduce or decrease the attenuating ability… resulting in an increase in radiographic density.”
AI:Negative contrast = gas = low attenuation = dark image.
Air is like a clear window. X‑rays go right through it, so the picture turns dark in that spot.
1. Room air used during a double‑contrast enema functions as a negative contrast agent because it does which of the following to the attenuating ability of the structures it fills?
A. Increases attenuation and decreases radiographic density
B. Increases attenuation and increases radiographic density
C. Reduces attenuation and increases radiographic density
D. Reduces attenuation and decreases radiographic density
C. Reduces attenuation and increases radiographic density
Supported by: “Negative contrast agents… involve the use of gases to reduce or decrease the attenuating ability… resulting in an increase in radiographic density.”
1. Positive contrast agents are used because they do which of the following to the attenuating ability of the structures they fill?
A. Reduce attenuation and increase radiographic density
B. Reduce attenuation and decrease radiographic density
C. Increase attenuation and decrease radiographic density
D. Increase attenuation and increase radiographic density
C. Increase attenuation and decrease radiographic density
Supported by: “Positive contrast agents increase the attenuation ability… resulting in a decrease in radiographic density.”
3. Positive contrast agents rely on which property to alter beam attenuation?
A. Low atomic number
B. High gas expansion
C. Increased specific gravity
D. Rapid absorption
C. Increased specific gravity
Rely on their ability to increase the specific gravity of the objects they fill to alter beam attenuation. → Their heaviness/density is what changes how x‑rays interact.
Positive contrast (barium, iodine) works because it is heavy.
When something is heavy, it:
👉 Blocks more x‑rays
👉 Stops the beam
👉 Makes the image turn white
1. Water‑soluble iodinated contrast media may be classified as which of the following?
A. Only ionic
B. Only non‑ionic
C. Ionic or non‑ionic
D. Neither ionic nor non‑ionic
C
Water‑soluble iodine contrast media:
a. May be ionic or non‑ionic
b. Readily absorbed by the body
c. Used for IVP, cystograms, intrathecal, and hysterosalpingogram studies
d. all of the above
d. all of the above
Oil‑based contrast agents are:
A. Rapidly absorbed and commonly used
B. Used for CT angiography
C. Slowly absorbed and not commonly used
D. Used for intrathecal studies
C. slowly absorbed and not commonly used
1. The kilovoltage used for an exam that utilizes iodinated contrast media is typically limited to which of the following values to prevent over‑penetration of the contrast?
A. 60 kVp or less
B. 80 kVp or less
C. 100 kVp or more
D. 125 kVp
B. 80 kVp or less
Non‑ionic Iodinated Contrast Media — Select All That Apply
Non‑ionic contrast media are designed to reduce the frequency of adverse reactions because they:
Do not dissociate into anions and cations
Limit the production of ions within the bloodstream when administered
Have no effect on iodine allergy reactions
Include examples such as Isovue, Omnipaque, and Optiray
All of the above
All of the above
Ionic iodinated contrast media:
a. Dissociate into two particles (a cation and an anion)
b. Increase the number of particles in solution (high osmolarity)
c. Are associated with more adverse reactions
d. Are water‑soluble
e. all of the above
e. all of the above
2. Which type of contrast media is associated with a higher osmolarity and therefore a greater likelihood of causing a reaction?
A. Non‑ionic contrast media
B. Oil‑based contrast media
C. Ionic contrast media
D. Gas‑based negative contrast agents
C. Ionic contrast media
Viscosity refers to which characteristic of a contrast agent?
A. Its osmotic pressure
B. Its thickness of contrast media
C. Its atomic number
D. Its rate of absorption in the bloodstream
B. Its thickness of contrast media
Barium sulfate must NOT be used in which clinical situation?
A. When the patient is NPO
B. When constipation is present
C. When a perforated bowel is suspected
D. When the patient is undergoing a small‑bowel follow‑through
C
When imaging with barium sulfate, which kilovoltage range is typically recommended to ensure adequate penetration while maintaining high subject contrast for a double‑contrast study, and which for a single‑contrast study?
A. Double‑contrast: 70–80 kVp Single‑contrast: 80–90 kVp
B. Double‑contrast: above 90 kVP Single‑contrast: 100-125 kVp
C. Double‑contrast: 60–70 kVp Single‑contrast: 70–80 kVp
D. Double‑contrast: 100–110 kVp Single‑contrast: 110–120 kVp
B.Double‑contrast: above 90 kVP Single‑contrast: 100-125 kVp
Gastrografin is preferred over barium sulfate when a perforated bowel is suspected because it is:
A. Oil‑based and slowly absorbed
B. Non‑absorbable and coats the mucosa better
C. Gas‑based and used as a negative contrast
D. Water‑soluble and iodine‑based positive contrast
D. Water‑soluble and iodine‑based positive contrast
Gastrografin is a water‑soluble, iodine‑based positive contrast agent, making it safer when perforation may exist.
Gastrografin is typically administered by which route when used as a positive contrast agent for GI studies?
A. Intravenously
B. Orally or through nasogastric tube
C. Intramuscularly
D. Subcutaneously
B
Special considerations or factors that may alter the type of volume of contrast used on an individual patient?
Select all that apply.
☐ Age and weight (pediatric & elderly sensitivity)
☐ Patient condition (perforated bowel
☐pts with impair liver or renal—
☐ Patients on medications (metformin, nephrotoxic drugs, beta‑blockers, anticoagulants)
☐ Use of contrast media in children and elderly being particularly hazardous
All of the above
Which of the following laboratory values are most important to review before administering iodinated contrast media?
A. Creatinine, BUN, and GFR
B. Liver enzymes (AST/ALT) and bilirubin
C. Coagulation values (PT/INR, aPTT)
D. Electrolytes (sodium, potassium, chloride)
A. Creatinine, BUN, and GFR
✔ Iodinated contrast is cleared by the kidneys
So we must check kidney function before giving it.
Which laboratory value is BEST for measuring kidney function and determining the stage of kidney disease, and what is it calculated from?
A. BUN; calculated from blood urea and hydration status
B. Serum creatinine; calculated from urine output
C. Estimated GFR (eGFR); calculated from the results of Creatine test, body size, age and gender
D. Urine specific gravity; calculated from urine concentration
C. Estimated GFR (eGFR); calculated from the results of Creatine test, body size, age and gender
eGFR is the BEST overall measure of kidney function
It tells you how well the kidneys filter blood
It is used to stage kidney disease
It is calculated from:
Serum creatinine
Age
Gender
Body size
1. Which term is defined as “when fluid infuses the tissue surrounding the venipuncture site”?
A. Extravasation
B. Phlebitis
C. Air embolism
D. Infiltration
D. Infiltration
Extravasation = contrast leak (dangerous)
Infiltration = IV fluid leak (mild)
Local effect
Which term is defined as the “accidental infiltration of a vesicant (contrast media) into the interstitial tissues at the site of injection”?
A. Infiltration
B. Phlebitis
C. Extravasation
D. Air embolism
C