Chemistry Unit 12: Reaction Rates and Equilibrium
1/51
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
52 Terms
Reaction Rate
the rate at which reactants change into products
Reaction Rate Equation
Δ[A]/Δt
Products have a ________ rate.
positive
Reactants have a ________ rate.
negative
What is Kinetics?
the study of how fast a reaction happens
What must happen for a reaction to occur?
-Particles must collide
-Must have enough energy (to break bonds)
Effective Collisions
Meet two conditions and result in a reaction.
Activated Complex
-an unstable arrangement of atoms that forms momentarily at the peak of the activation-energy barrier.
-Between reactants and products.
Potential Energy
Energy that is stored and held in readiness
Activation Energy
-Energy required to start a reaction
-Difference in potential energy
The larger the activation energy...
the slower the reaction
How is energy lost in a reaction
heat
If energy of products is lower than the energy of reactants...
exothermic (-ΔH)
If energy of products is higher than the energy of reactants...
endothermic (+ΔH)
How many factors affect reaction rate?
7
What are the factors that affect reaction rate?
Nature of Reactants, Concentration, Particle Size (SA), Agitation, Temperature, Pressure, and Catalyst.
Nature of Reactants
Some substances react faster than others
Elementary steps
Individual steps in a reaction (reactants to products)
All elementary steps together
Reaction Mechanism
Which step determines the reaction rate?
Slowest
Rate Determining Step
The slowest step determines the reaction rate
The slowest step determines the reaction rate
Rate Determining Step
Intermediate
products in one step, reactants in another
Concentration
Larger concentration = more collisions = faster reaction
Particle Size (Surface Area)
More particles on surface = more collisions = more activated complex = more product
Agitation
Particles more exposed = more correct collisions = more activated complex = product faster
Pressure
More pressure = less volume = particles closer = more collisions = more activated complex = product faster
Temperature
-Higher temp = more KE = more collisions
-Collisions are harder (overcome Activation Energy easier)
Catalyst
-lowers activation energy
-Is not used up in reaction
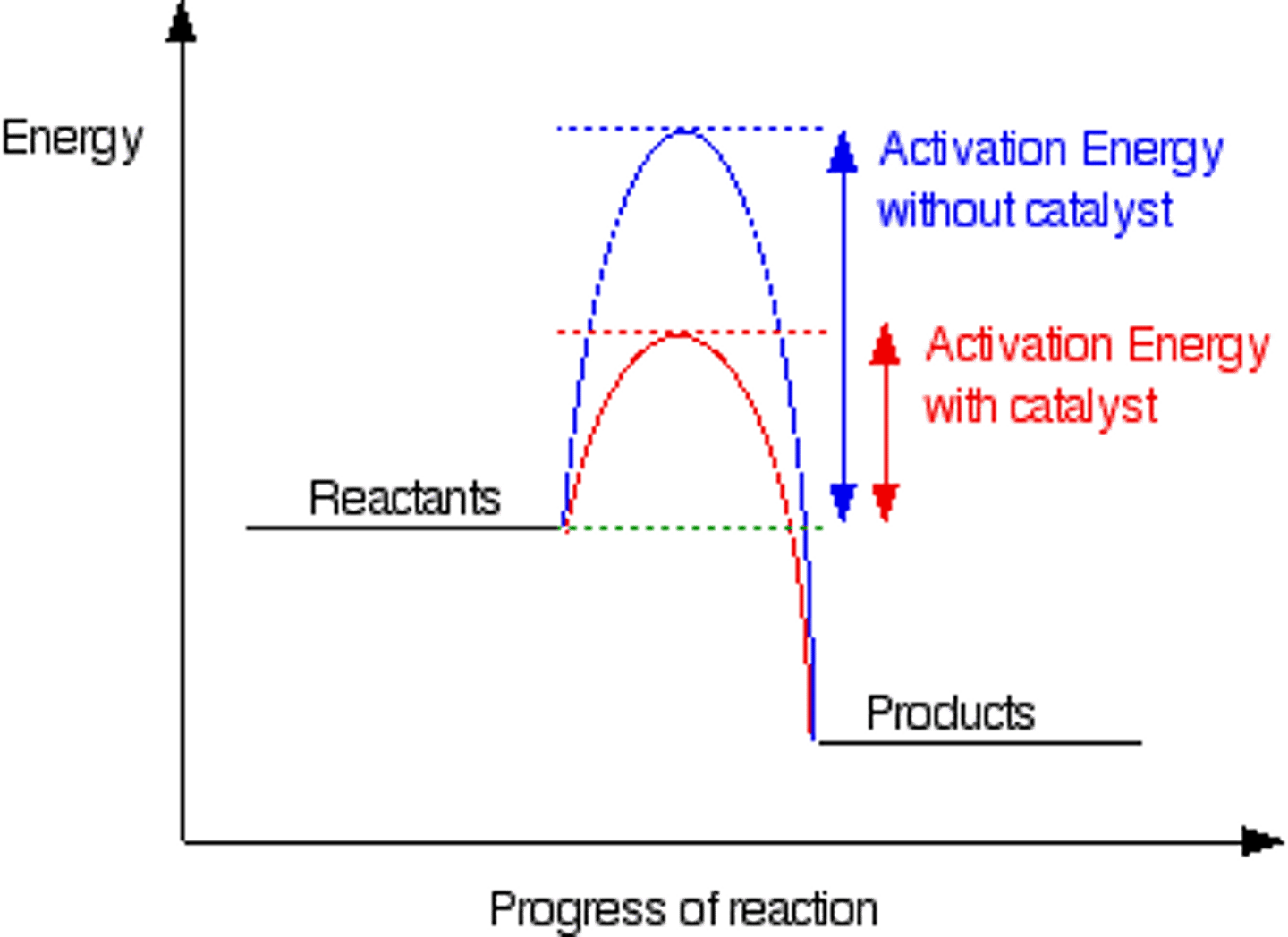
Most effective factor to increase reaction rate?
Temperature
Reversed Reaction
products can turn back into reactants (goes both ways)
Chemical Equilibrium
-rate(forward)=rate(reverse)
-Only occurs in a closed system
-Only solutions and gases
Chemical Equilibrium can only occur in...
A closed system
Chemical Equilibrium Equation
K(eq)=[C]c[D]d/[A]a[B]b
Capital letters in chemical equilibrium equation
Reactants/Products
Lowercase letters in chemical equilibrium equation
Coefficients
Chemical Equilibrium equation must be
Products/Reactants
Chemical Equilibrium > 1
Products are favored at equilibrium
Chemical Equilibrium < 1
Reactants are favored at equilibrium
Le Châtelier's Principle
If a stress is applied to a system at equilibrium, the system shifts in the direction that relieves the stress.
3 Main Stressors
Concentration, Temperature, and Pressure!
What effect does the addition of a catalyst have on equilibrium?
no effect
More product is added (Equilibrium)
Shifts left
More reactant is added (Equilibrium)
Shifts right
Product removed (Equilibrium)
Shifts right
Reactant removed (Equilibrium)
Shifts left
Exothermic reactions (Equilibrium)
Shifts left (away from heat)
Endothermic reactions (Equilibrium)
Shifts right (away from heat)
Cooling (Equilibrium)
Has opposite effects (moves towards heat)
Increased Pressure (Equilibrium)
Shifts towards the side with fewer gas molecules
Decreased pressure (Equilibrium)
Shifts towards the side with more gas molecules
20. What information is NOT given by an overall equation for a chemical reaction?
a. the relative numbers of molecules used
b. the probable order of the reaction
c. the number of atoms participating in the reaction
d. the reaction mechanism
d. the reaction mechanism