Chem 122 Master Flashcards
1/83
Earn XP
Description and Tags
All clickers, old exams, and mock exams mc
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
84 Terms
Which has atoms in it?
Cells, Air, Gold
Which is biggest?
A cell
How big do you think an atom (H) is?
About 0.1 nanometer
Elements are composed of small indivisible, indestructible particles called atoms.
No
All atoms of an element are identical and have the same mass and properties.
No
All atoms of a given element are different from atoms of other elements.
Yes
Compounds are formed by combinations of atoms of two or more elements.
Yes
Chemical reactions are due to the rearrangements of atoms, atoms (matter) are neither created nor destroyed during a reaction.
Yes
How many protons in atom of cobalt-60?
27
How many neutrons in atom of cobalt-60?
33
How many electrons in atom of cobalt-60?
27
What forces are acting on the ball when it is in the air?
Gravitational and Electromagnetic
What happens to the force of attraction as the mass of the interacting objects increase?
Increase
What happens to the force of attraction as the distance between the objects increase?
Decrease
What happens to objects with the same charge?
They repel each other
What forces act on a ball in someones hand?
Gravitational and Electrostatic
As a ball moves towards the ground what happens to the kinetic energy?
Increase
As the ball moves towards the ground what happens to the potential energy of the system (ball+Earth)?
Decrease
When the ball is falling, what causes the potential energy to decrease?
The gravitational interaction between the Earth and the ball
As the ball moves towards the ground what happens to the total energy of the system (ball+Earth)?
Stays the same
How many molecules must be present for LDFs to exist?
Two or more
As the helium atoms approach what happens to the total energy?
Stays the same
As the helium atoms approach what happens to the potential energy?
Decreases
As the helium atoms approach what happens to the kinetic energy?
Increases
As the helium atoms get very close (overlapping electron clouds) what happens to the total energy?
Stays the same
As the helium atoms get very close (overlapping electron clouds) what happens to the kinetic energy?
Decreases
As the helium atoms get very close (overlapping electron clouds) what happens to the potential energy?
Increases
How do He atoms “know” the temperature is rising?
The energy is transferred from other atoms that have collided with the walls of the container (that were directly heated)
What is true about a single chemical bond breaking?
Energy is absorbed
What is the strongest type of interaction?
Covalent Bonds
At 5 K, what holds the nuclei together within an H2 molecule
Covalent bond
At 5 K, why do molecules of H2 stick to other molecules of H2?
London Dispersion Forces
What is true about the forming of a single chemical bond?
Energy is released
What types of bonds and/or interactions are represent for H2 in the solid phase?
Both Covalent bonds and London dispersion forces
The boiling point of hydrogen is 20K. When hydrogen boils, what interactions and/or bonds are overcome?
London dispersion forces
Molar Mass Units
g / mole

Calculate the mass
18.2
Two models are used to describe the behavior of light (electromagnetic radiation).
A wave and a particle
What is the energy of a 422 nm photon?
4.71 × 10^-19
How much energy is there in one mole of 422 nm photons?
284 kJ/mol
Can atoms of a particular element absorb or emit any wavelength of light?
No
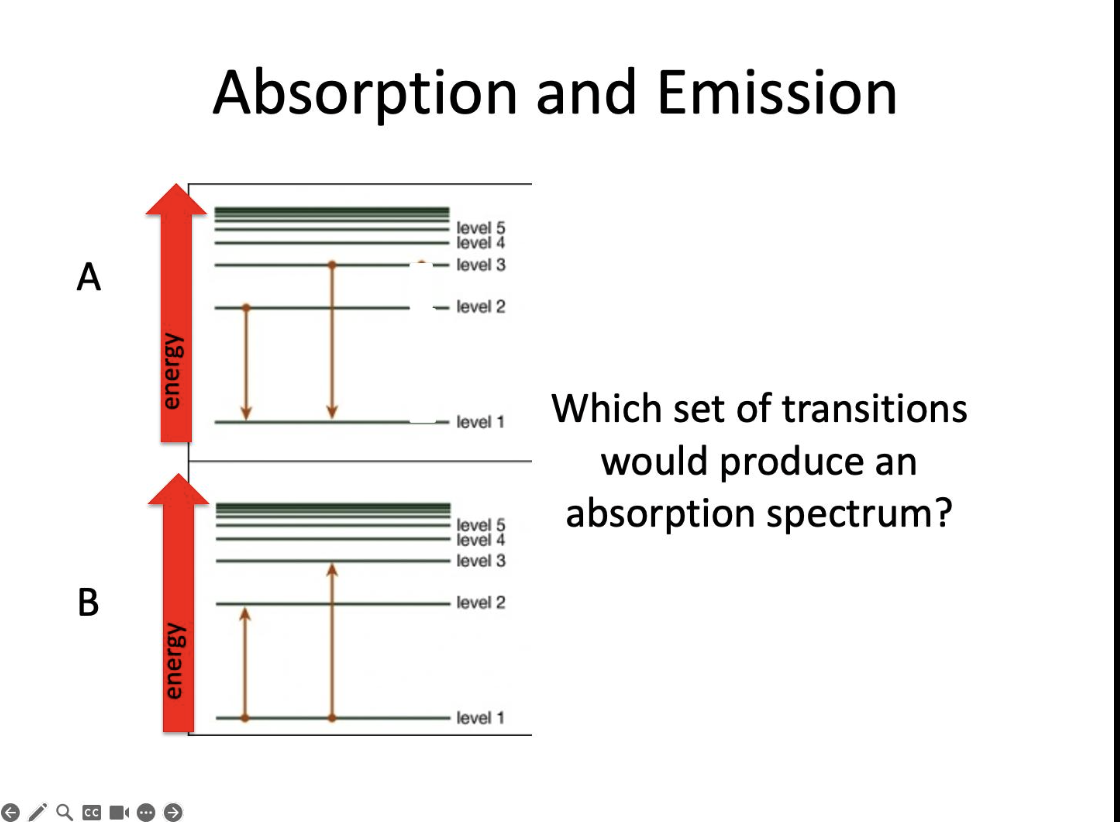
Which set?
B

Which transition is most likely from level 2 to 1?
Yellow
How many core electrons & valence electrons does N have?
2 core, 5 valence
How many core electrons & valence electrons does Si have?
10 core, 4 valence
Do you predict the atomic radius of Li is larger or smaller than that of Na?
Smaller
Do you predict the atomic radius of Li is larger or smaller than that of Ne?
Smaller
What is the effective nuclear charge of Carbon?
+4
What is the effective nuclear charge of Nitrogen?
+5
What is the effective nuclear charge of Oxygen?
+6
If an electron is removed from an atom, what is the charge of the ion?
Positive
If an electron is added to an atom, what is the charge of the ion?
Negative
Compare Li and Na. Which is easier to remove an electron from?
Na
The ionization energy of Na is _____ than that of Li?
Smaller
Compare Li and Ne. Which is easier to remove an electron from?
Li
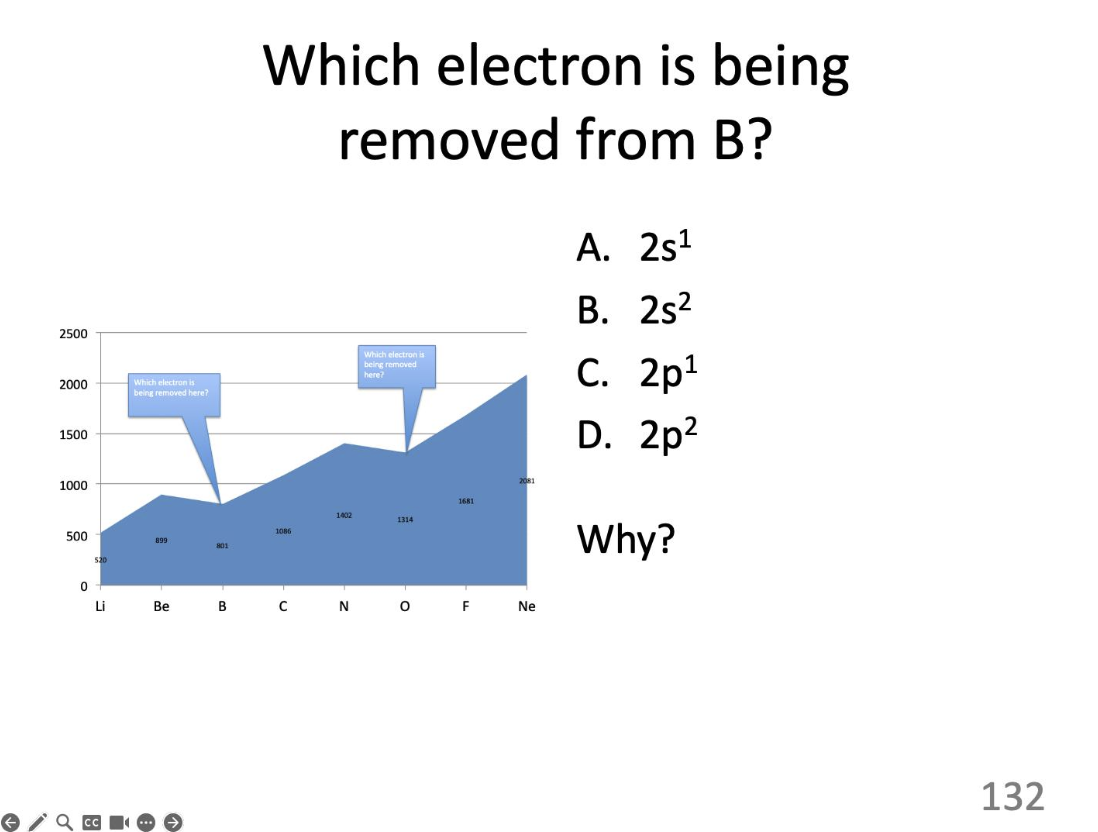
Which electron?
2p²
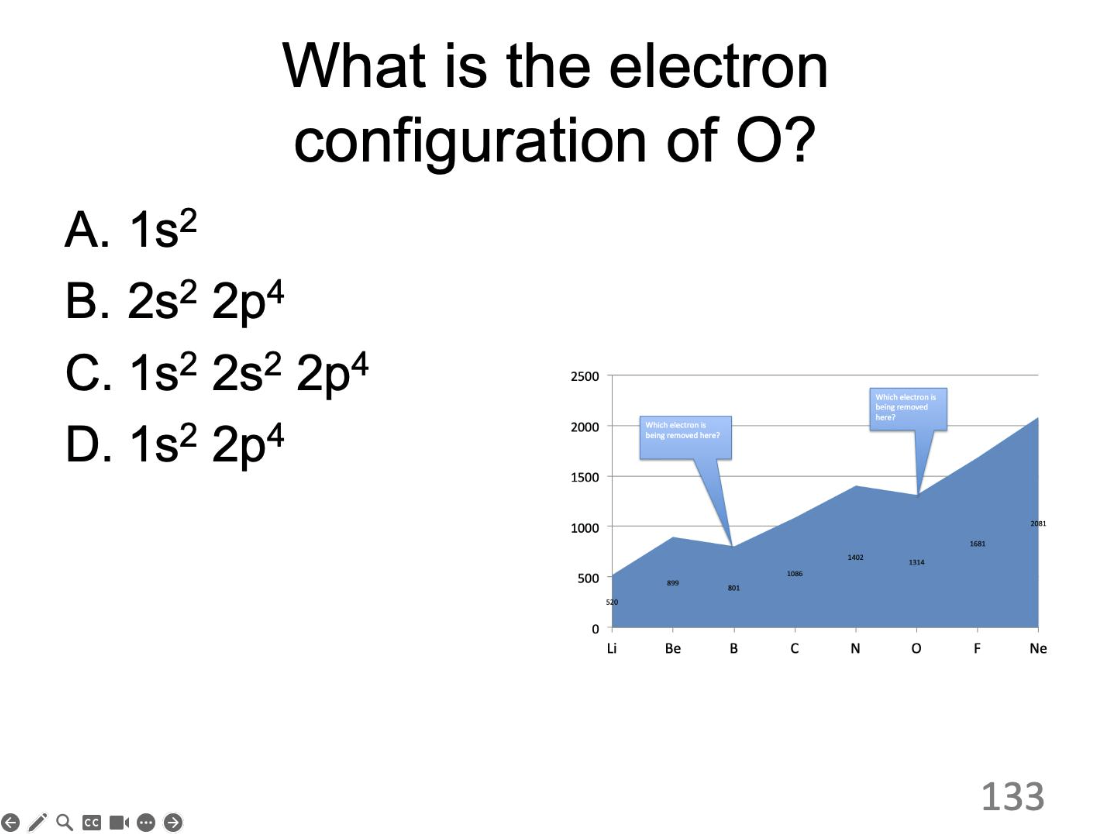
What is the electron configuration of O?
1s²2s²2p^4
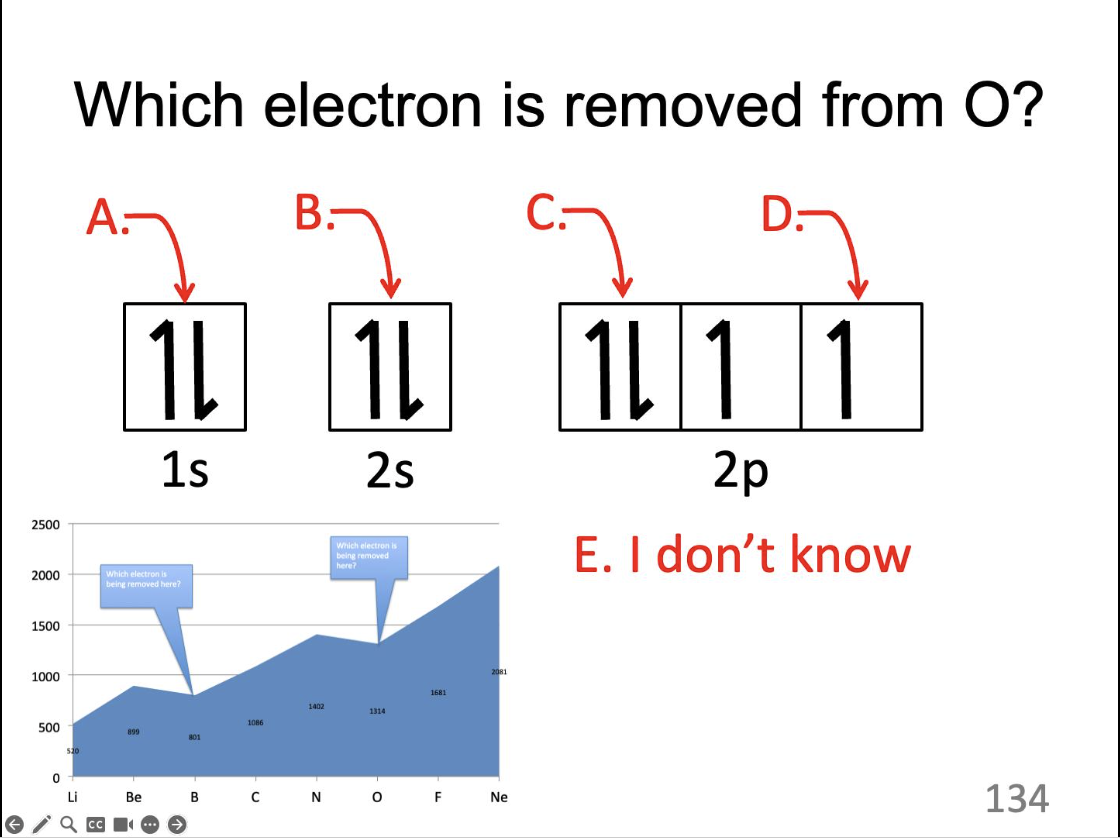
Which electron is removed from O?
C

Which Ionization energy is larger?
C. Third IE
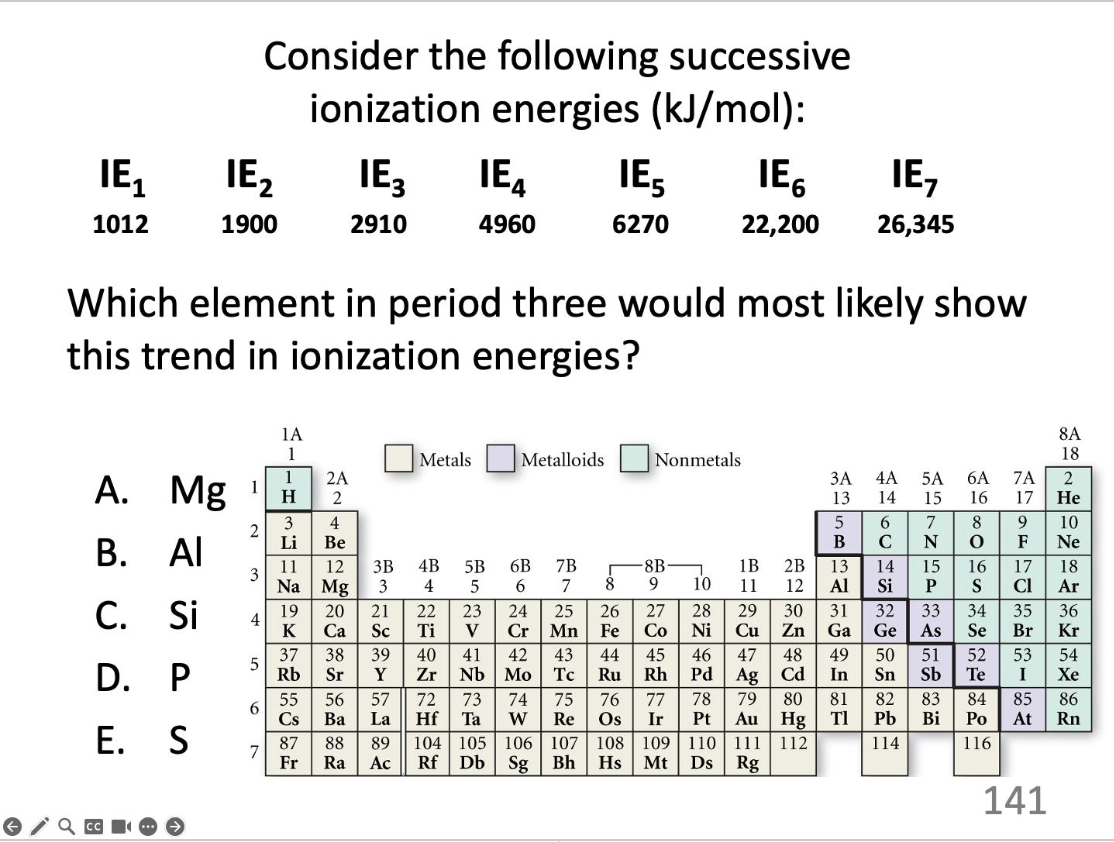
Which element in period three would most likely show this trend in ionization energies?
P
As two nuclei move toward each other (but are still separated by distances greater than the size of the nucleus) what force affects them?
Electrostatic
As two nuclei move toward each other (but are still separated by distances greater than the size of the nucleus) what happens to the potential energy of the system?
Increases
As two nuclei move toward each other (but are still separated by distances less than the size of the nucleus), the strong nuclear force comes into effect. What happens to the potential energy of the system?
Decreases
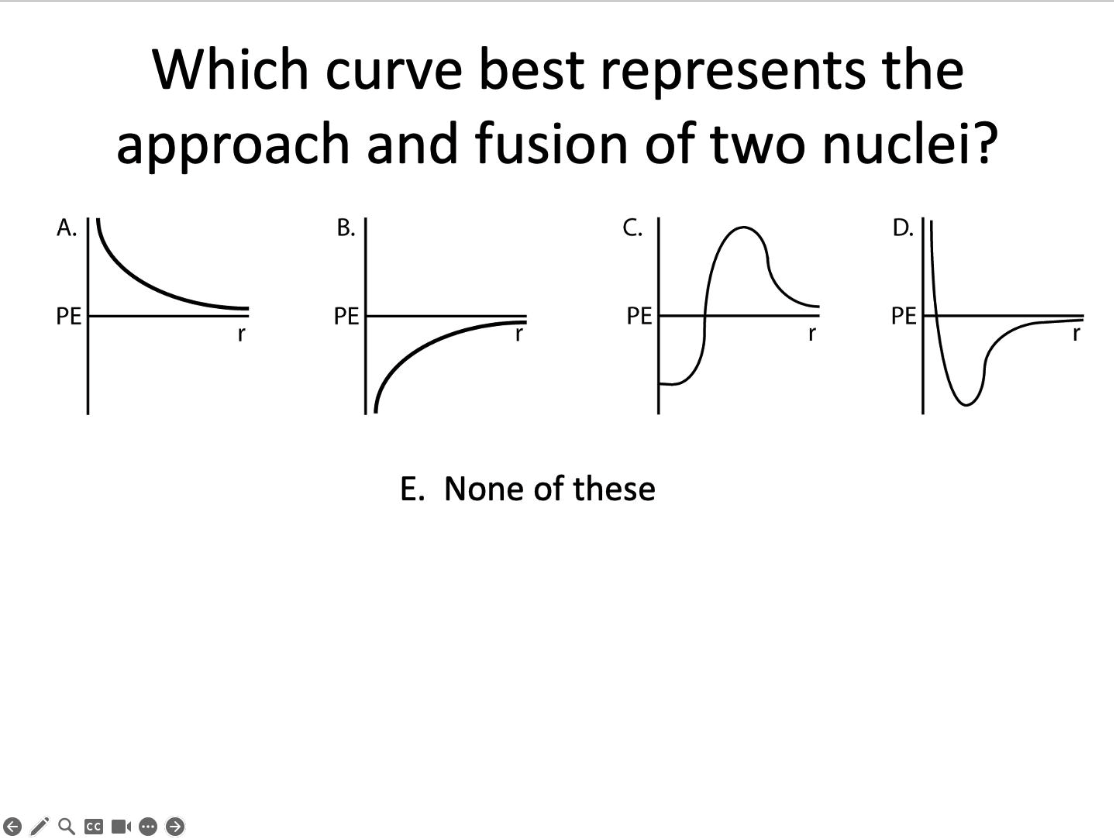
Which curve?
C

Which?
A
How many protons and neutrons does 12N have?
7p, 5n

Balance
B
Why do bonds form between atoms?
Because the potential energy is at a minimum
If two elements in the second row (eg two O atoms) were to interact, how many molecular orbitals would be generated by the atomic orbitals in the core and valence shells?
10
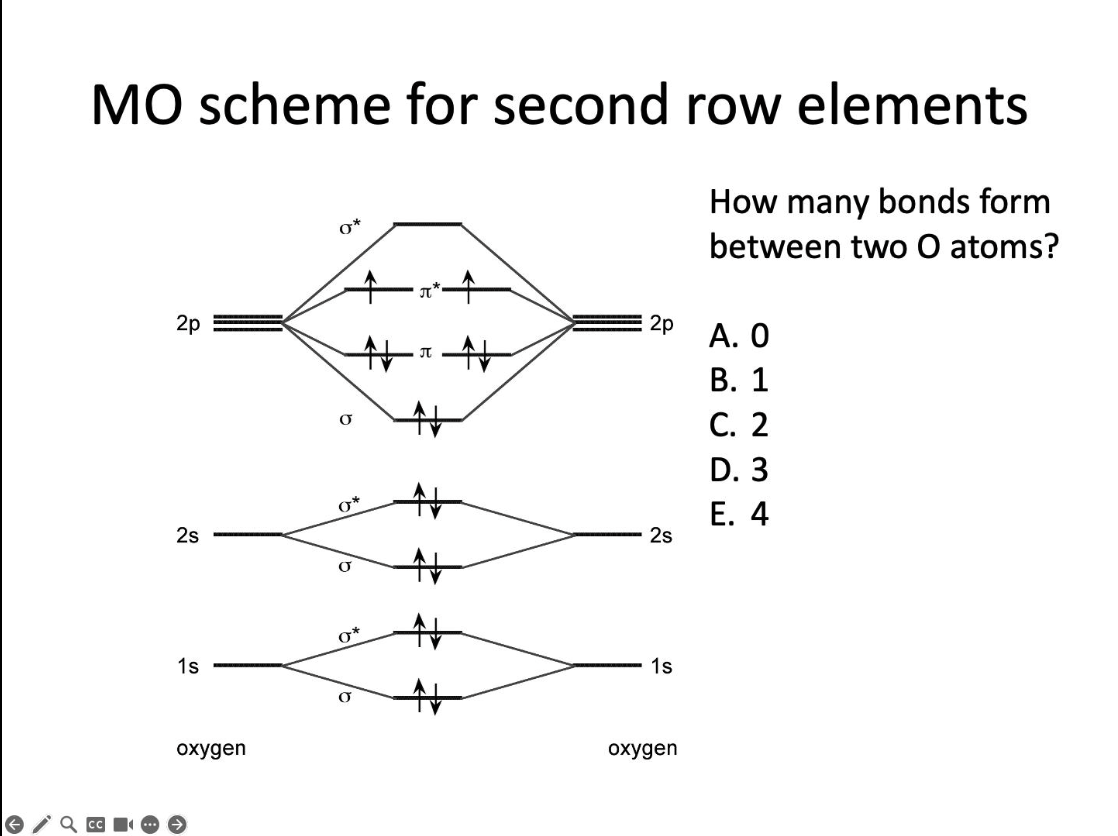
How many bonds form between two O atoms?
C
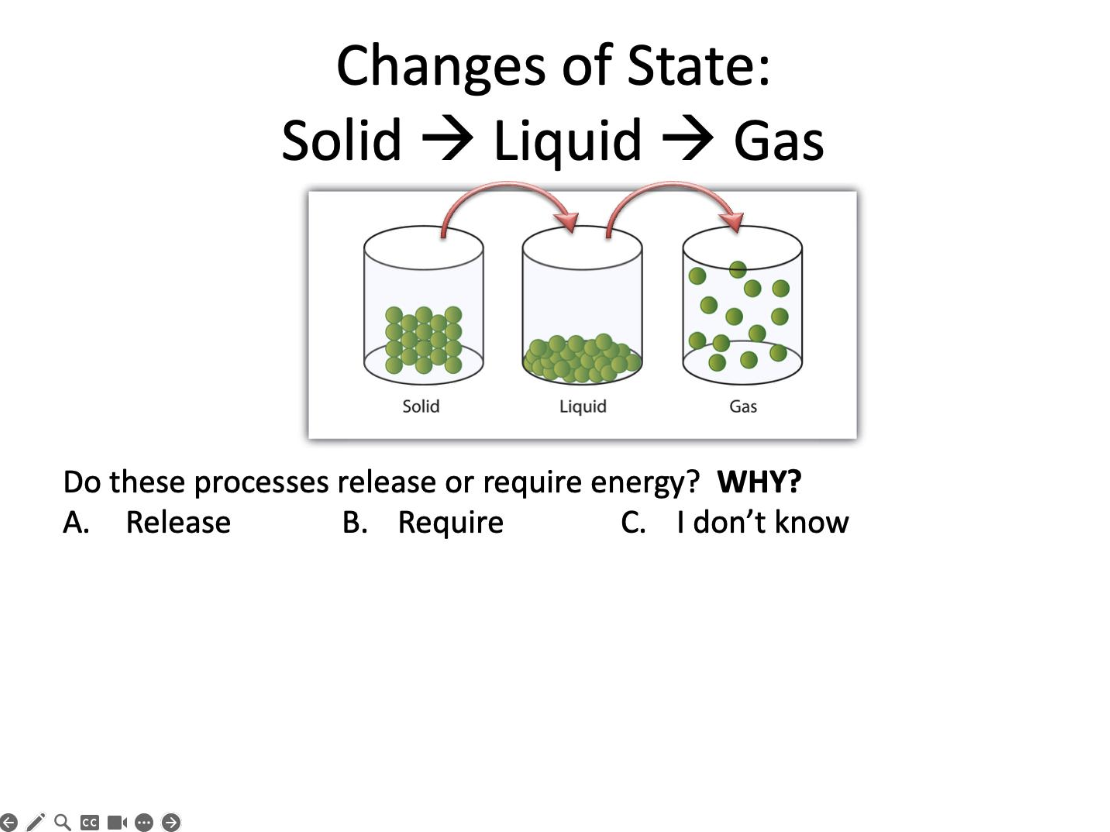
Do these processes release or require energy?
Require
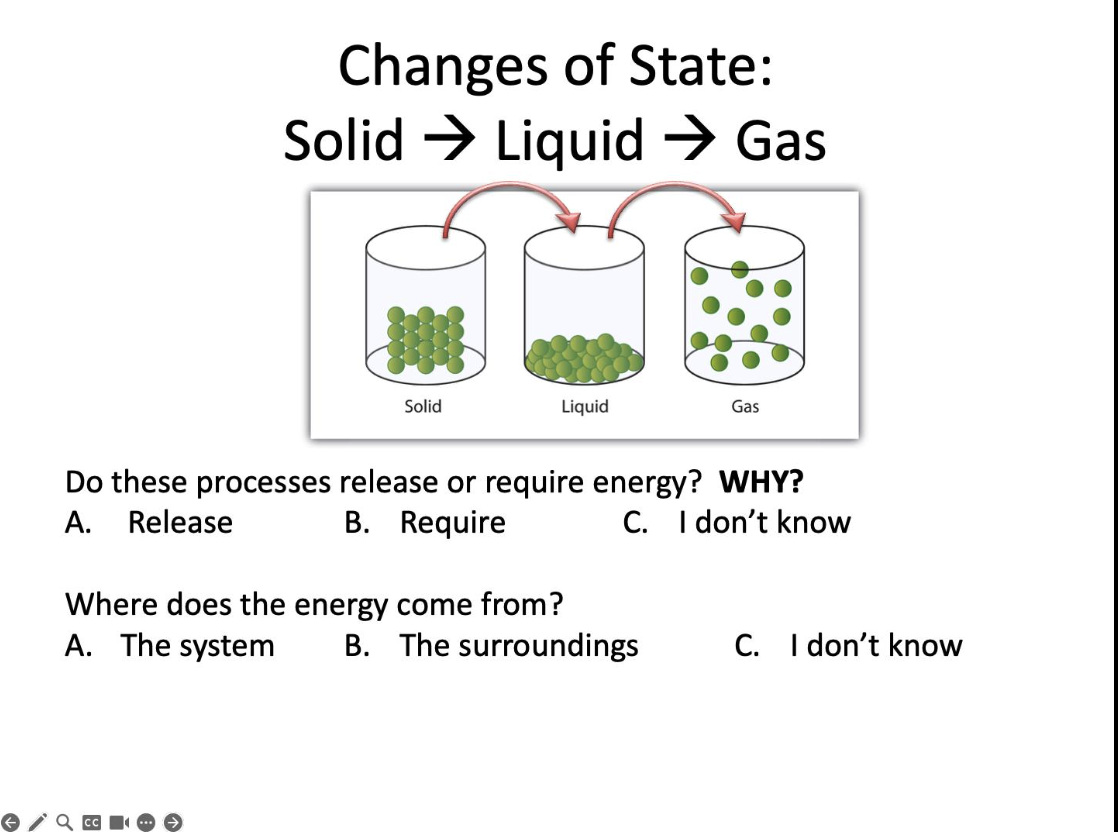
Where does the energy come from?
The surroundings
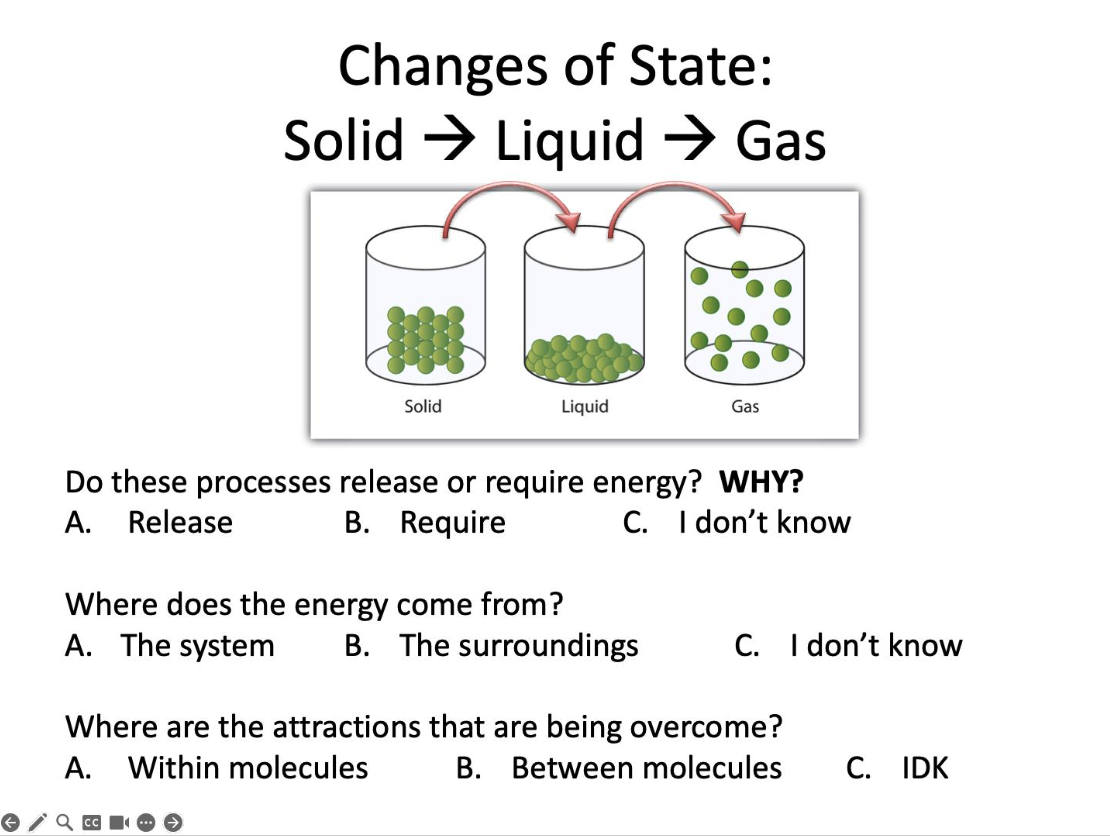
Where are the attractions that are being overcome?
Between molecules
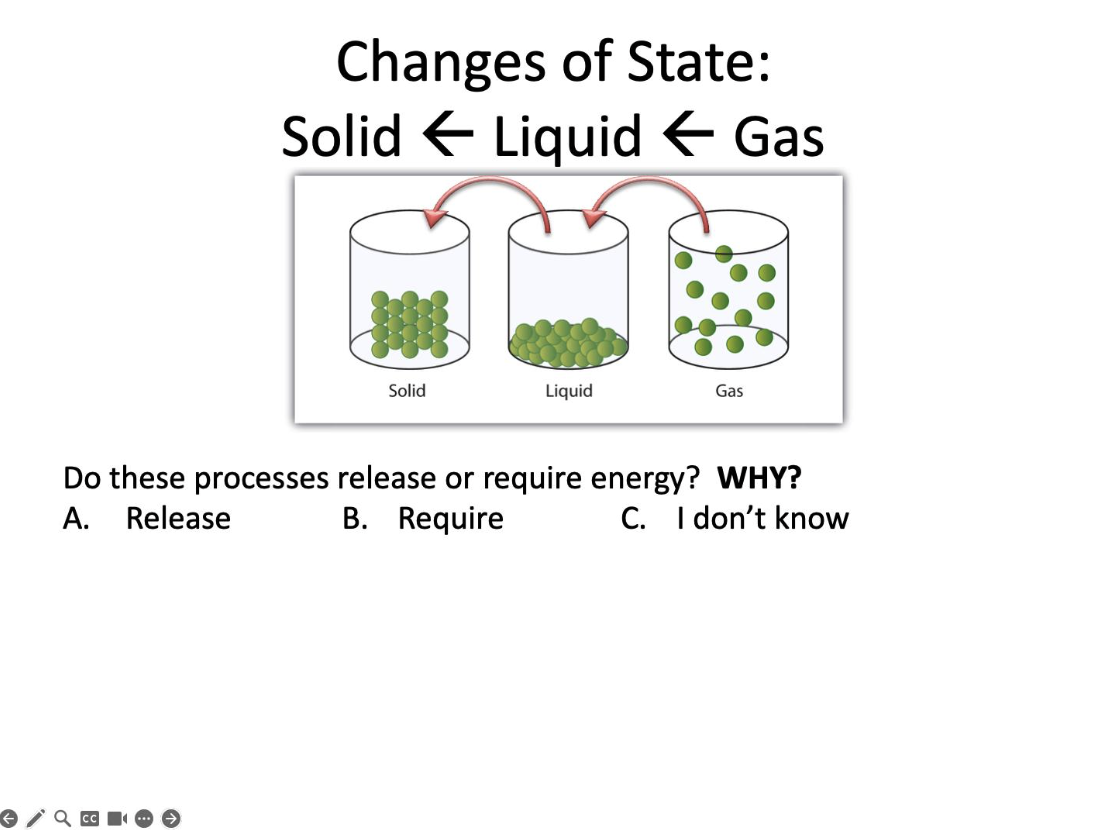
Do these processes release or require energy?
Release
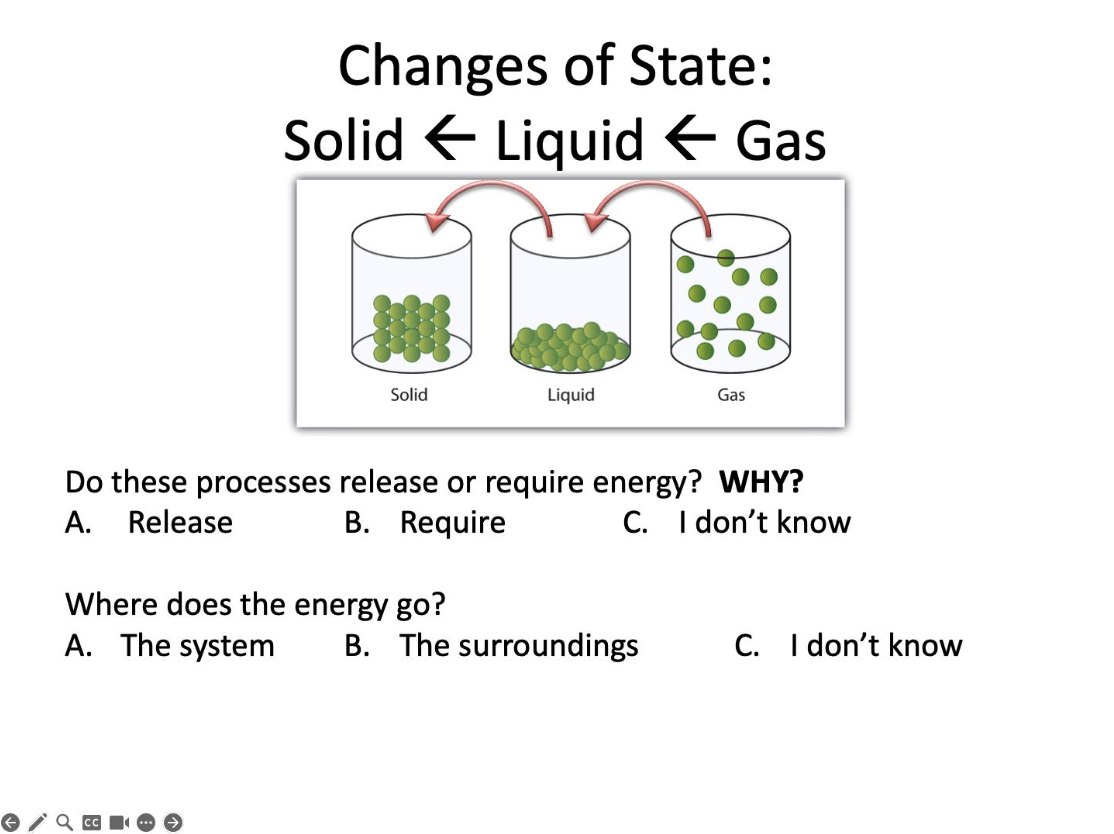
Where does the energy go?
The surroundings
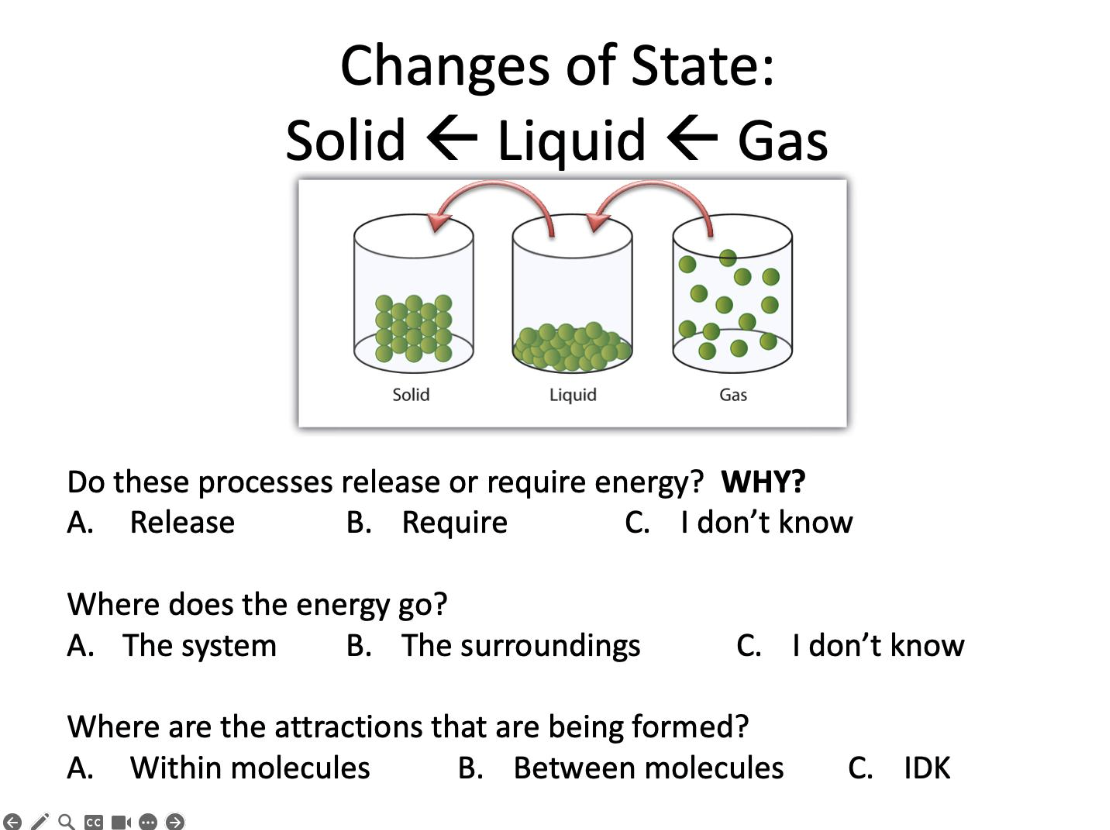
Where are the attractions that are being formed?
Between molecules
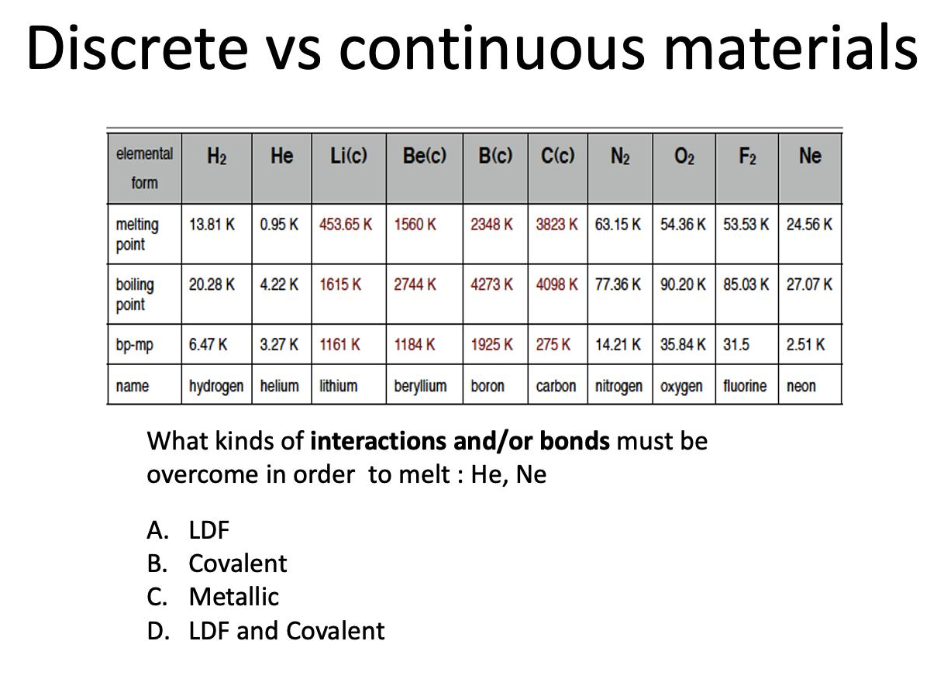
What kinds of interactions and/or bonds must be overcome in order to melt : He, Ne?
LDF
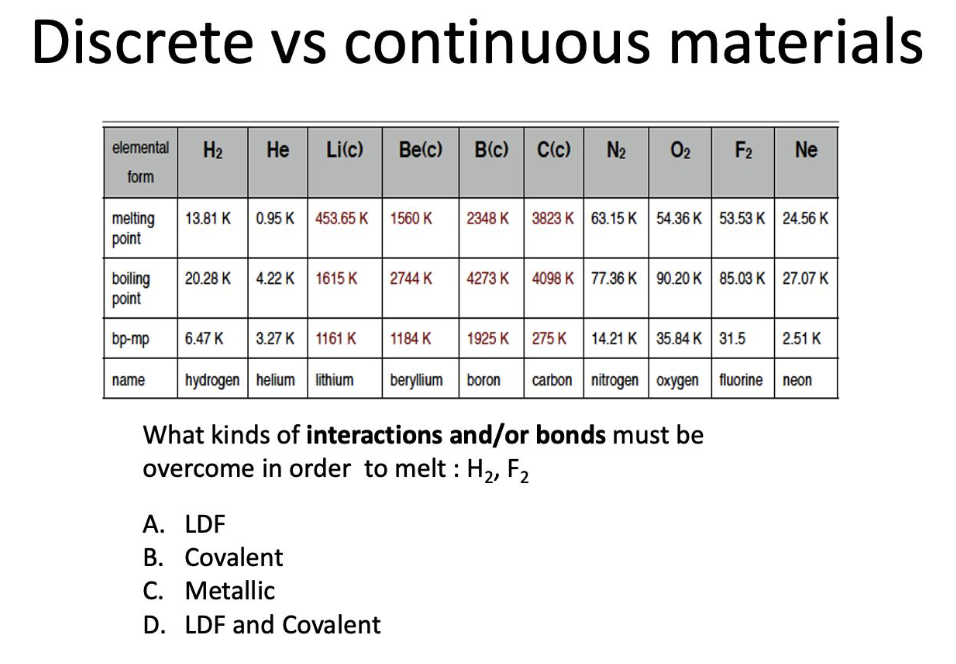
What kinds of interactions and/or bonds must be overcome in order to melt : H2, F2
LDF