Lecture 1: Organic Metabolism (Absorptive State Reactions, Fat in blood, Postabsorptive state reactions)
1/34
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
35 Terms
absorptive state vs postabsorptive state
absorptive state: period during which ingested nutrients are entering the blood
postabsorptive state: period during which the GI tract is empty of nutrients
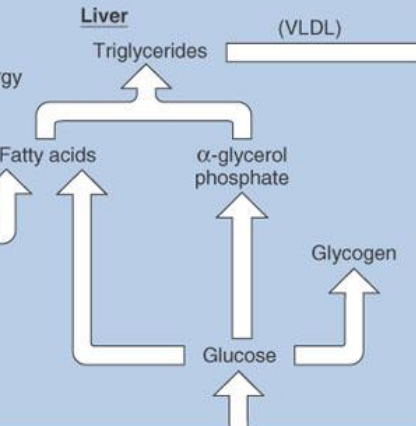
during the absorptive state, glucose taken into liver is…
Used for energy
Converted to glycogen
Converted to alpha-glycerol phosphate or fatty acids to be made into triglyceride
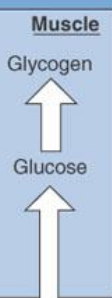
during the absorptive state, glucose taken into skeletal muscle is…
used for energy
converted to glycogen
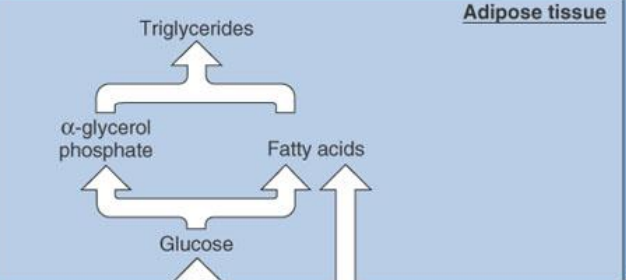
during the absorptive state, glucose taken into fat is…
Used for energy
Converted to alpha-glycerol phosphate or fatty acids to be made into triglyceride
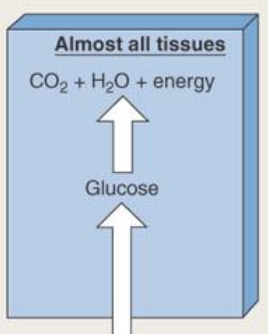
during the absorptive state, glucose taken into other cells are…
used for energy
during the absorptive state, amino acids taken liver are...
Used to synthesize proteins (e.g., plasma proteins made in liver)
Converted to alpha-keto acids which either enter glycolytic pathway, replace intermediates in the Krebs Cycle, or are converted to fatty acids
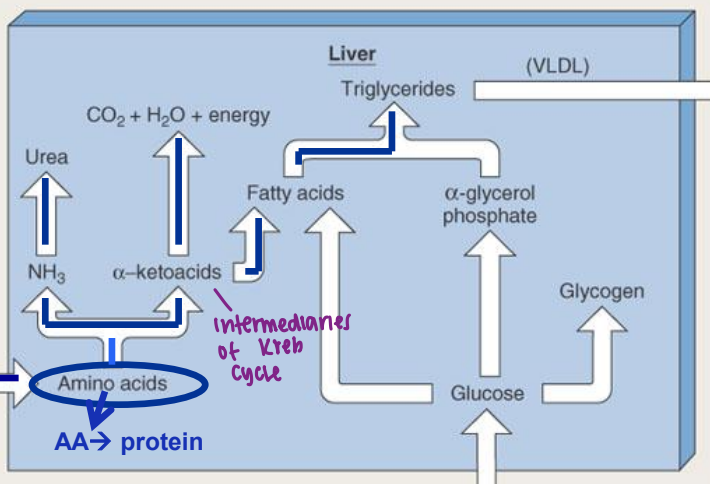
during the absorptive state, amino acids taken into muscle (and other) cells are
Used to synthesize proteins
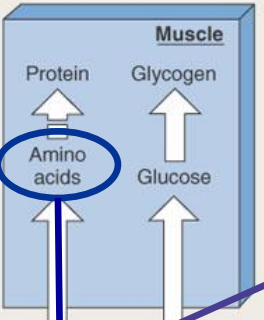
during the absorptive state, circulating fat comes from...
Chylomicrons absorbed from the GI tract, then into lacteals go into blood (do not first go to liver)
VLDL (lipoproteins) made in the liver
during the absorptive state, what primarily happens to circulating triglycerides?
Lipoprotein lipase breaks them down to monoglyceride and 2 free fatty acids
Free fatty acids enter adipocytes and are remade into triglyceride and stored
Small amount of monoglycerides enter adipocytes and can form triglycerides
Most monoglycerides return to the liver

nutrients enter liver via
hepatic portal vein
VLDLs and chylomicrons express a form of
ApoB
VLDLs and chylomicrons gain expression of
ApoC-II while circulating. ApoC-II binds the lipoproteins to the enzyme lipoprotein lipase
ratio of fat/protein by smallest to largest
HDL (1.5) < LDL (3.5) < VLDL (9) < Chylomicron (99)
HDL
moves excess cholesterol from blood and tissue to the liver for excretion
LDL
have high % of cholesterol and only ApoB100 on surface; they provide cholesterol to tissues
VLDLs
(from liver) have ApoB100. contain high % of TGs and gain ApoC-II in plasma which allows them to interact with lipoprotein lipase to deliver fatty acids to cells
Chylomicrons
(from GI tract) have ApoB48. contain high % of TGs and gain ApoC-II in plasma which allows them to interact with lipoprotein lipase to deliver fatty acids to cells
cholesterol is used
in membranes (all cells)
to create bile salts (hepatocytes)
to create vitamin D (skin)
to create steroid hormones (adrenal gland and gonads)
what happens to chylomicrons and VLDL if triglyceride content drops
they are then called chylomicron and VLDL remnants, and they lose ApoC-II and gain ApoE which targets them to the liver
ApoB48 remnants (from chylomicrons) are completely removed by the liver
ApoB100 remnants (from VLDL) are not completely removed. Some lose the ApoE and become LDL
where does HDL come from
primarily from liver
ApoA-I is secreted from liver. within the plasma, this low lipid ApoA-I incorporates membrane phospholipids and un-esterified cholesterol and becomes nascent HDL
how does HDL lower cholesterol levels
mature HDl particles interact with a receptor on hepatocytes (SR-BI), promoting uptake of lipids (cholesterol and cholesterole esters) into the liver
the liver then incorporates these cholesterole products into bile, either by creation of bile salts or by secretion of cholesterol itself in bile
to improve cholesterol levels,
lower LDL and higher HDL is better
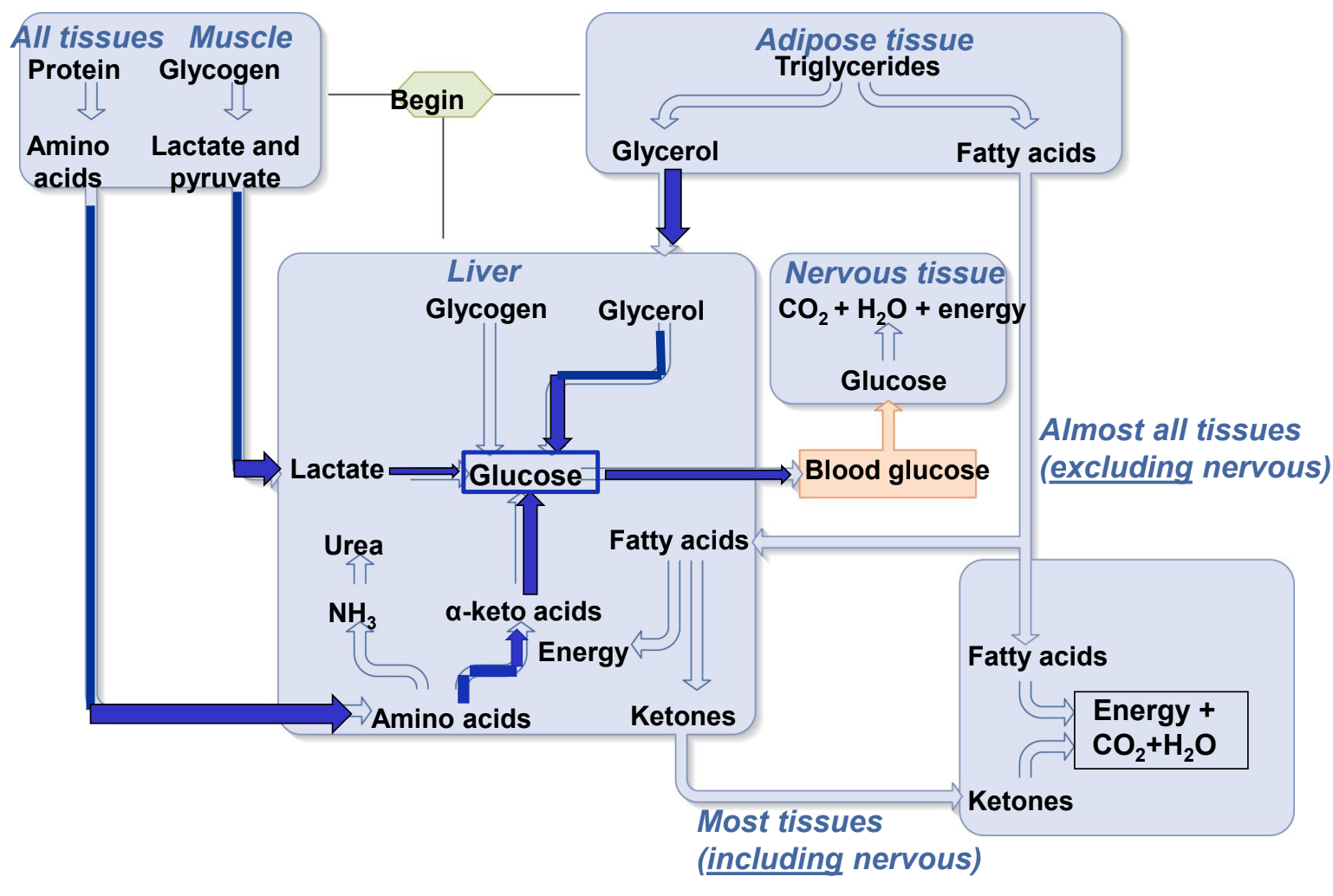
net catabolism or anabolism in postabsorptive state?
net catabolism (breakdown) of glycogen, fat, and proteins begin
glycogenolysis (what is it and where does it occur)
breakdown of glycogen. occurs in liver and skeletal muscle.
glycogenolysis in liver
glycogen is broken down into glucose-6-phosphate and converted to glucose which can be released to the blood
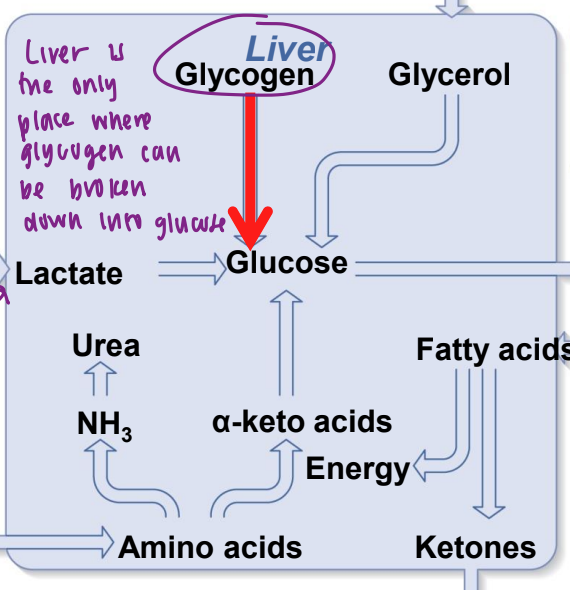
glycogenolysis in skeletal muscle
glycogen is broken down into G6P, but muscle does not have the enzyme to convert G6P to glucose, so G6P undergoes glycolysis to pyruvate in skeletal muscle cell, which has 2 fates
Kreb’s Cycle (ATP production)
converted to lactate which is released to the blood, taken up by the liver and converted into glucose by liver (gluconeogenesis)
what are the substrates in gluconeogenesis
lactate, pyruvate, amino acids, glycerol
how is lactate and pyruvate substrate used to make glucose in gluconeogenesis
lactate is from glycogen breakdown in muscle
lactate → pyruvate in liver and pyruvate is used to generate glucose
how is glycerol used to make glucose in gluconeogensis
TG breakdown (lipolysis) → glycerol + fatty acids
glycerol and fatty acids enter blood by diffusion
glycerol is taken up by liver and converted to glucose
where does gluconeogenesis occur
liver
how are amino acids used to make glucose in gluconeogensis
protein breakdown (proteolysis) yields amino acids
amino acids are released to the blood and taken into liver
liver converts amino acids (particularly alanine) to glucose (by converted them to alpha-keto acids then oxaloacetate or pyruvate)
glucose-sparing
metabolic adjustment by which most cells in the body increase fat utilization and decrease glucose utilization during the postabsorptive period
fatty acids are metabolized for energy by all cells other than CNS and RBCs
glucose sparing in liver
fatty acids converted to ketones = acetoacetic acid (acetoacetate + H+), beta-hydroxybutyric acid (beta-hydroxybutyrate + H+), and acetonine.
ketones then enter the Krebs cycle to make ATP
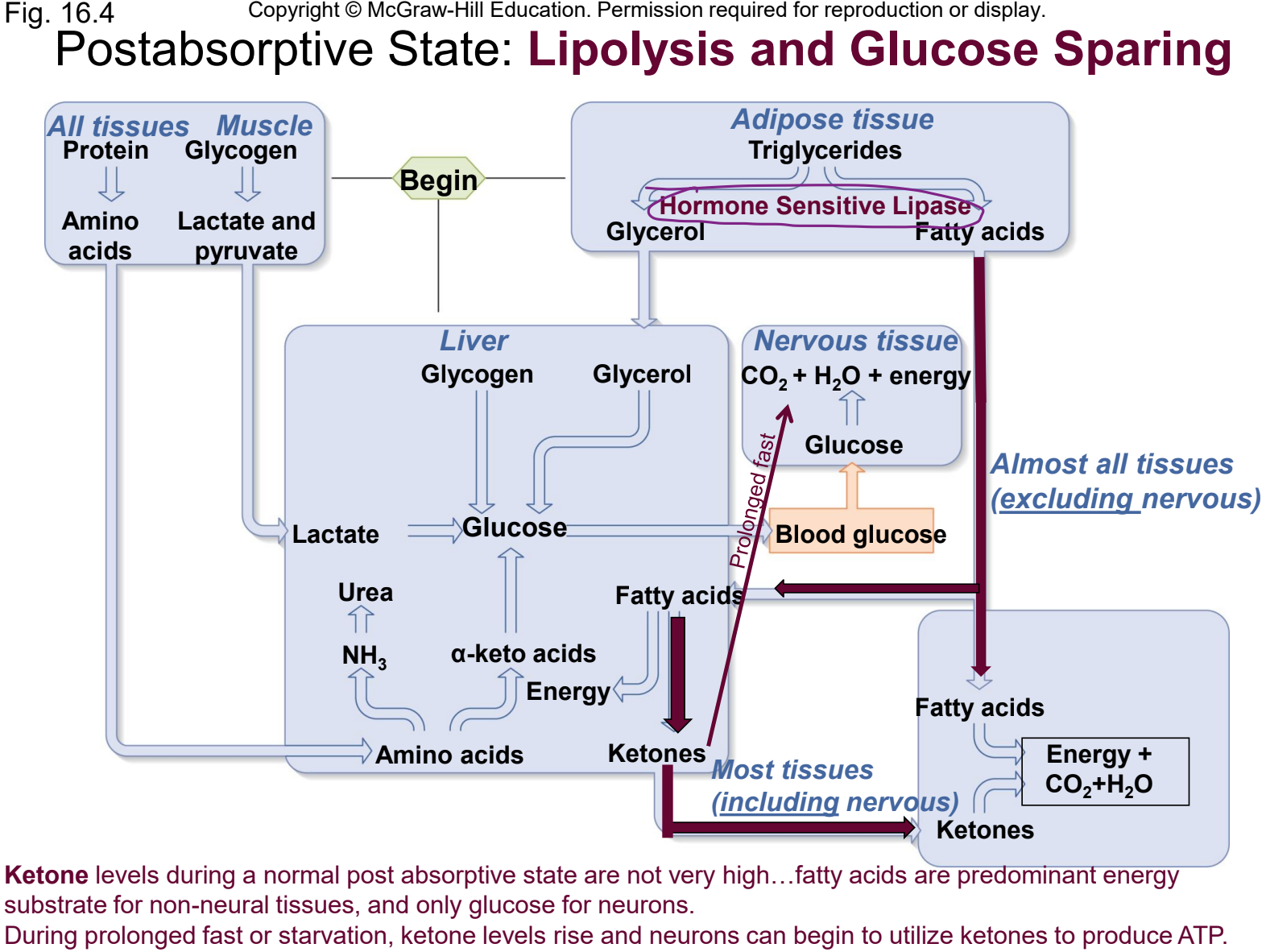
glucose sparing in body cells
fatty acids undergo beta-oxidation to (1) acetyl CoA which enters Kreb cycle; and to (2) NADH + H+ and FADH2 which enter oxidative phosphorylation

what happens with prolonged fasting or starvation in glucose sparing
CNS will begin to utilize ketones for energy which thereby minimizes protein breakdown
