[L20] Hydrogen
1/9
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
10 Terms
Current situation
used to be on the rise
lot of investment in the past
present: less talk
appeared in 1800s
Properties
lightest element (atomic mass = 1)
gas at atmospheric conditions
colourless, odourless, tasteless, non-toxic, highly combustible
stable in form H2
not naturally available alone, abundant in compounds with other elements
melting point: -259°C
boiling point: -252°C
high pressures needed to keep H2 liquid at room temperature or cryogenic temperatures
energy carrier ➔ high energy density but low mass density (0.08 kg/m3, 120 MJ/kg, 9.6 MJ/m3)
Sources of hydrogen
Green hydrogen → 1%
water electrolysis & renewable electricity
Blue hydrogen
fossil fuel but CO2 captured & stored underground
carbon capture, storage and utilisation (CCSU) → expected to be carbon neutral
Gray hydrogen → 99% (95% natural gas by SMR, 4% partial oxidation)
from fossil fuel & commonly uses steam methane reforming method (extracts H2 from methane using steam)
CO2 produced & released
Pink hydrogen
from nuclear energy
Future
many more uses to come
planned H2 in the world is much larger than current H2
From electrolysis
separation of H20 into H2 & O2
efficiency ~ 70-85%
From methane
several options w/ CO2, high T &/or high P
Steam-methane reforming reaction
CH4+H2O (+heat) → CO +3H2
Partial oxidation of methane reaction
CH4 + 1/2O2 → CO + 2H2 (+heat)
95% natural gas by SMR, 4% partial oxidation
Storage
can be stored either as a gas or as a liquid.
gas storage done in high pressure tanks (350-700 bar or 5000-10,000 psi)
liquid hydrogen storage done in cryogenic temperatures to prevent it boiling back into a gas
hydrogen can also be stored on the surface of (adsorption) or within solid materials (absorption)
Energy extraction
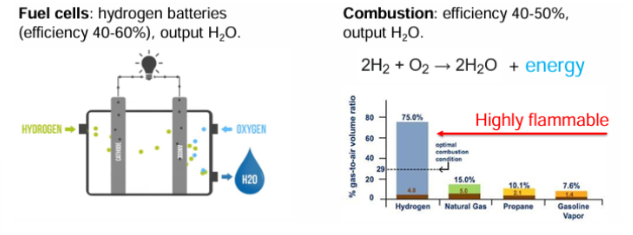
Fuel cells
hydrogen batteries
efficiency 40-60%
output H2O
Combustion
efficiency 40-50%
output H2O
Opportunities
Isolated systems (no access to grid) where changing infrastructure may result cost-effective: steel production, shipping and planes; but not consumer cars (electric grid is more available already).
Highly volatile future, subject to regulations against fossil fuels or technology breakthroughs.
There can be a strong industry lobby (from CH4 owners) to push further.
Challenges
Production of H2: energy demanding (only 1% is currently green hydrogen) = mainly non green
Difficult storage (and transport): high pressure or cryogenic conditions
Highly leak able and flammable – difficulties for infrastructure adaptation
Production technology not deployed at market level and infrastructure clearly lagging behind