Chemistry Week 2
1/36
Earn XP
Description and Tags
Atoms and the Periodic Table
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
37 Terms
Dalton’s Atomic Theory
Law of Conservation of Mass
Law of Definite Proportions
Law of Multiple Proportions
An element only consists of one type of atom that is unique to the element and found in all other exampled of that element
Law of Conservation of Mass
The total mass present before a chemical reaction is equal to the amount of mass present after the reaction has finished
This is sometimes changed by mass being converted into large amounts of energy instead, Ex. Uranium Decomposing
Law of Definite Proportions
Elements always combine in the same proportions by mass
Percent of atoms from each element also combine in set proportions

Law of Multiple Proportions
Great majority of molecules combine into small number ratios that are fixed
“Small” is relative to what is possible in the universe
Cathode Ray Experiment
Discovered the electron in 1897
Conducted by shooting a cathode ray between a positive and negative magnet
The ray bent slightly towards the positive magnet, proved the ray had something negative in it
Through trigonometry it was found that the mass of the negatively charged thing had a mass that was 1/12 that of an atom
Thompson’s Plum-Pudding Model
Early / false model of an atom
Atom is a cloud of positive electricity with negative electrons inside of it
The positive electricity is the “pudding”
The negative electrons are the “plums”
Logic follows below
The Cathode Ray Experiment proves that an atom has negative particles (modern-day electrons) in it
The atom makes up all matter
Mater with the same charge repel each other
Everything in the universe doesn’t repel each other
There must be a positive component to the atom
The positive component was theorized to be a positive electrical field “pudding”

Alpha Particels
He²+
Positively charged particle
Beta Particels
High energy electrons
Gama Rays
High energy electromagnetic radiation
Gold Foil Experiment
Discovered a positively charged nucleus at center of atom made of protons and neutrons
Proton
Subatomic particle
Part of all nuclei
Postive, +1 charge
1.007 amu*, replace with 1 for the purposes of Chem 101
Neutron
Subatomic particle
Part of nearly all nuclei, not present in most H nuclei
Neutral charge
1.009 amu, replace with 1 for the purpose of Chem 101
Electron
Subatomic particle
Found outside the nucleus
Negative, -1 charge
5.486 × 10^-4 amu, replace with 0 for purpose of Chem 101
Amount present in atom equal to that of protons
Subdivisions: Core electrons and valence electrons
Atomic Structure
99% empty space
Early on in periodic table neutrons and protons equal each other, causing the mass number to be double the atomic number
Later on in the periodic table neutrons start to greatly outnumber the amount of protons present
Atomic number (Z)
Mass number (A)
Atomic Number
Part of atomic structure
Decides the identity of the element
Count of number of protons in the atom
Mass Number
Number of protons plus the number of neutrons
Isotope
Different versions of the same element
Contain different number of neutrons
Ion
Different version of the same element
Contains different number of electrons
Subdivided into cation and anion
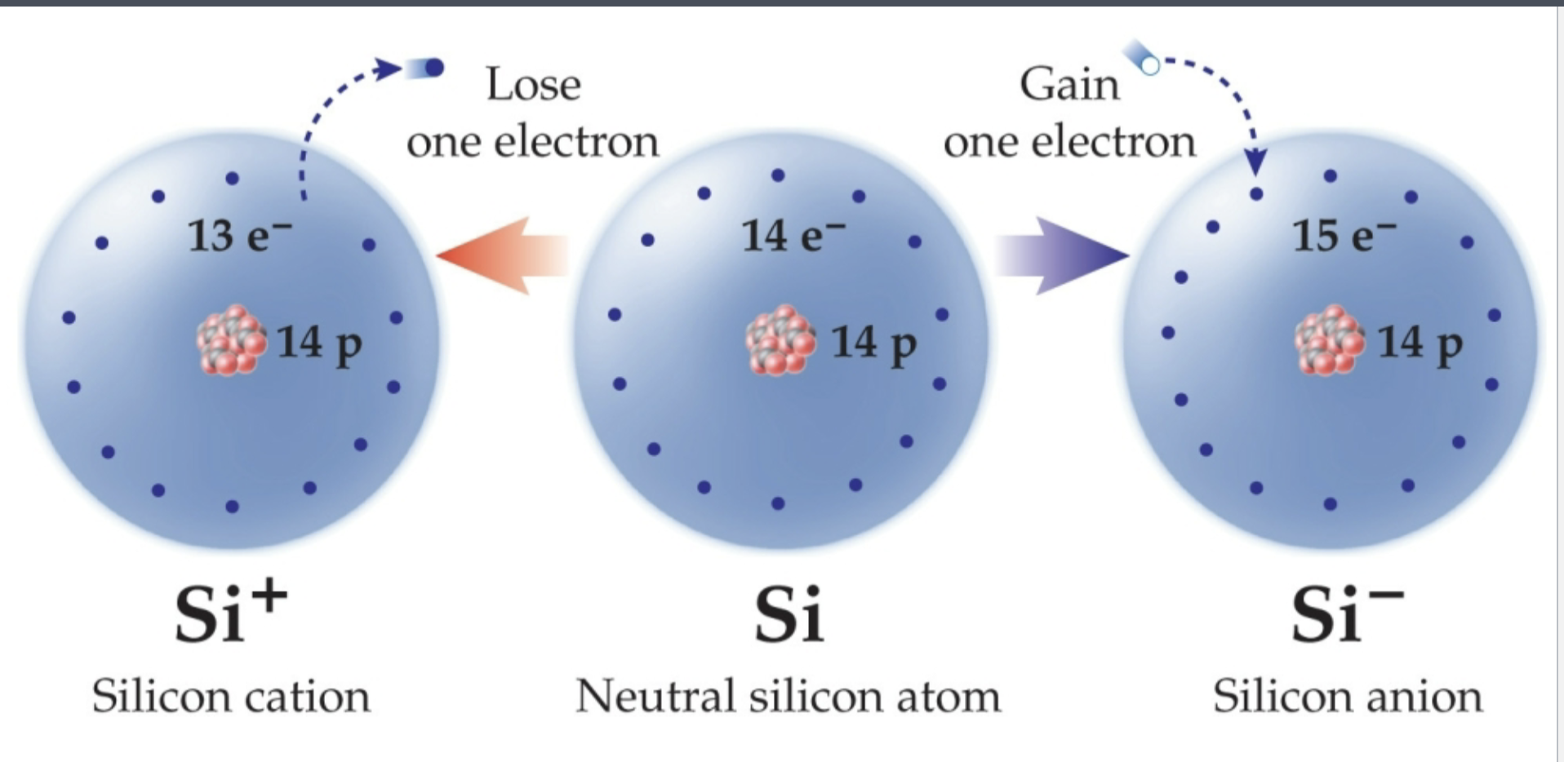
Cation
A positively charged ion
Has lost an electron
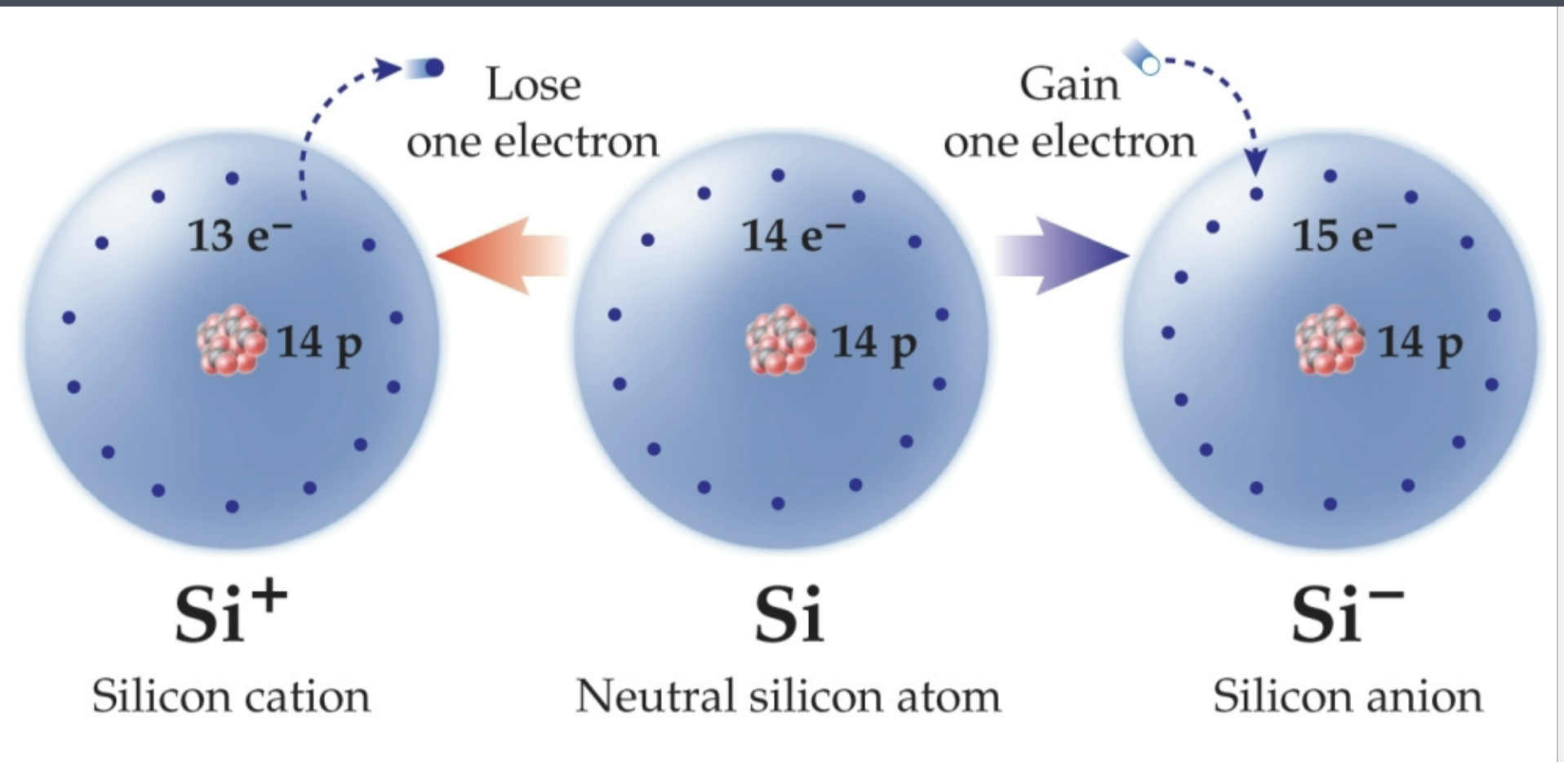
Anion
A negatively charged ion
Has gained an electron
Atomic Mass
Actual mass of an atom
Atomic Mass Unit (AMU)
1/12 of the atomic mass
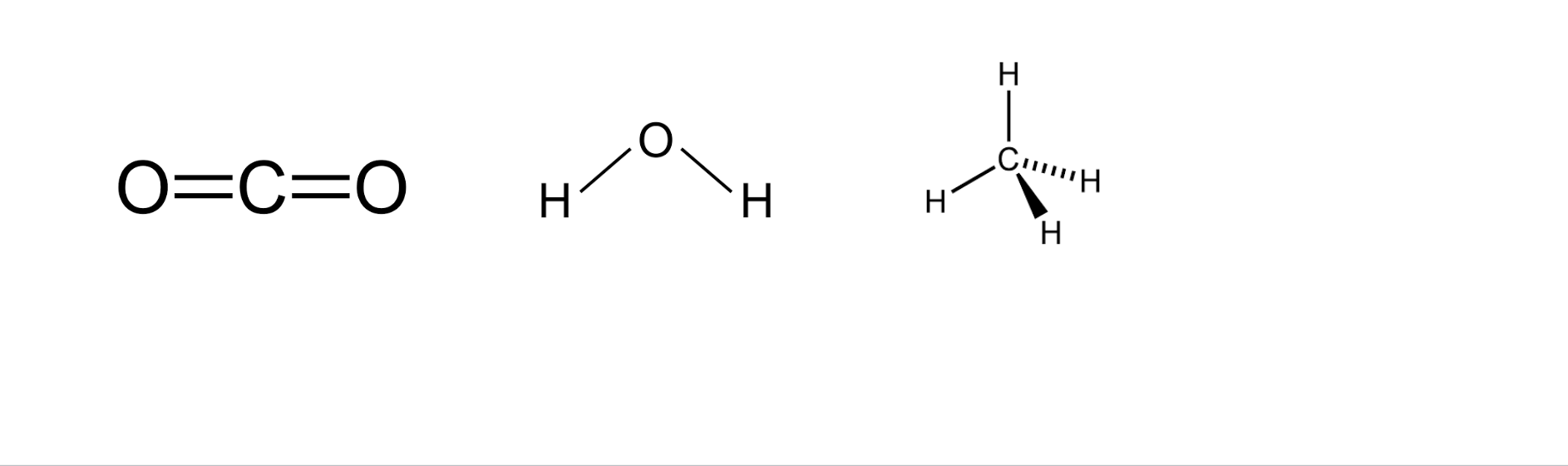
Chemical formulas
Represent molecules
Molecular formula and structural formula
For the most part the element that appears first on the periodic table leads
Ex: CO
Molecular formula
Used the chemical symbol to indicate type of atom followed by a subscript that shows the amount of that atom present in each molecule

Structural formula
Gives same info as molecular formula but also shows how atoms are connected
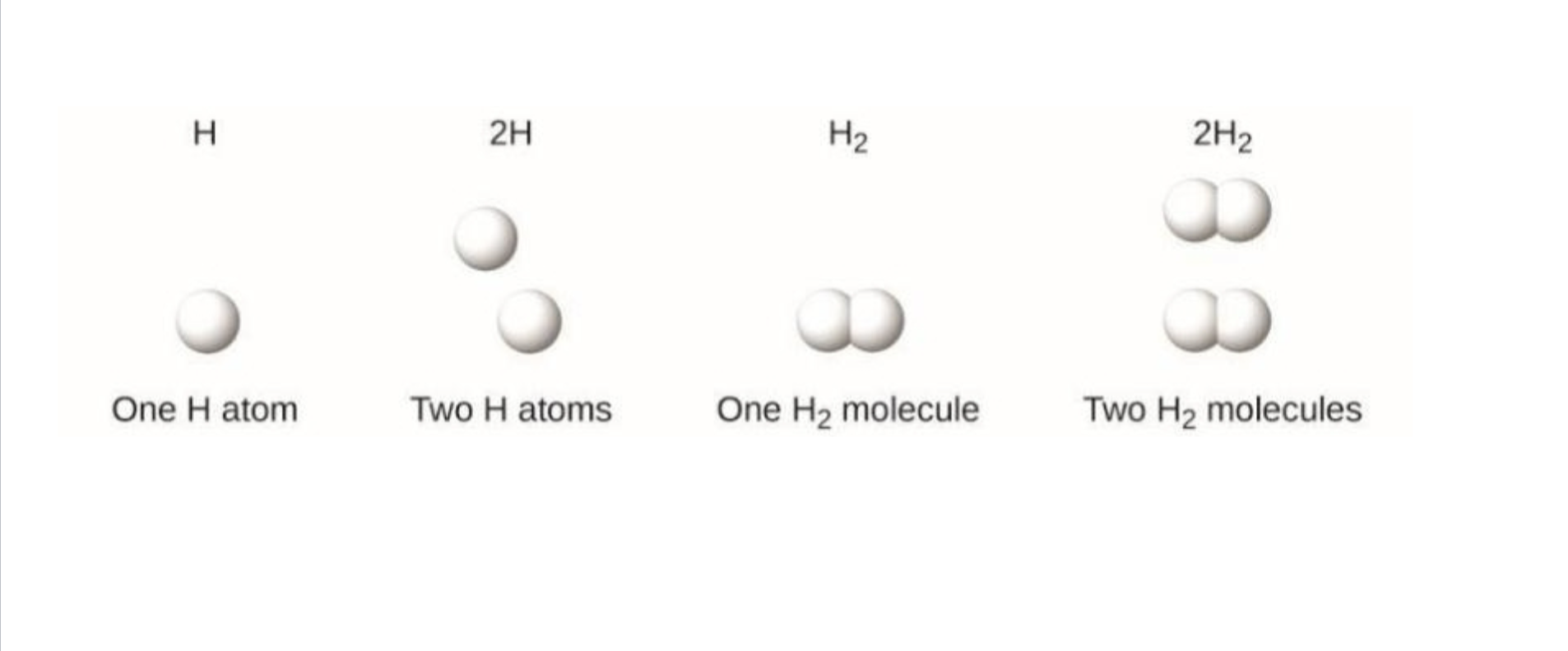
Coefficient
Comes before a molecular formula
Indicates the number of molecules present
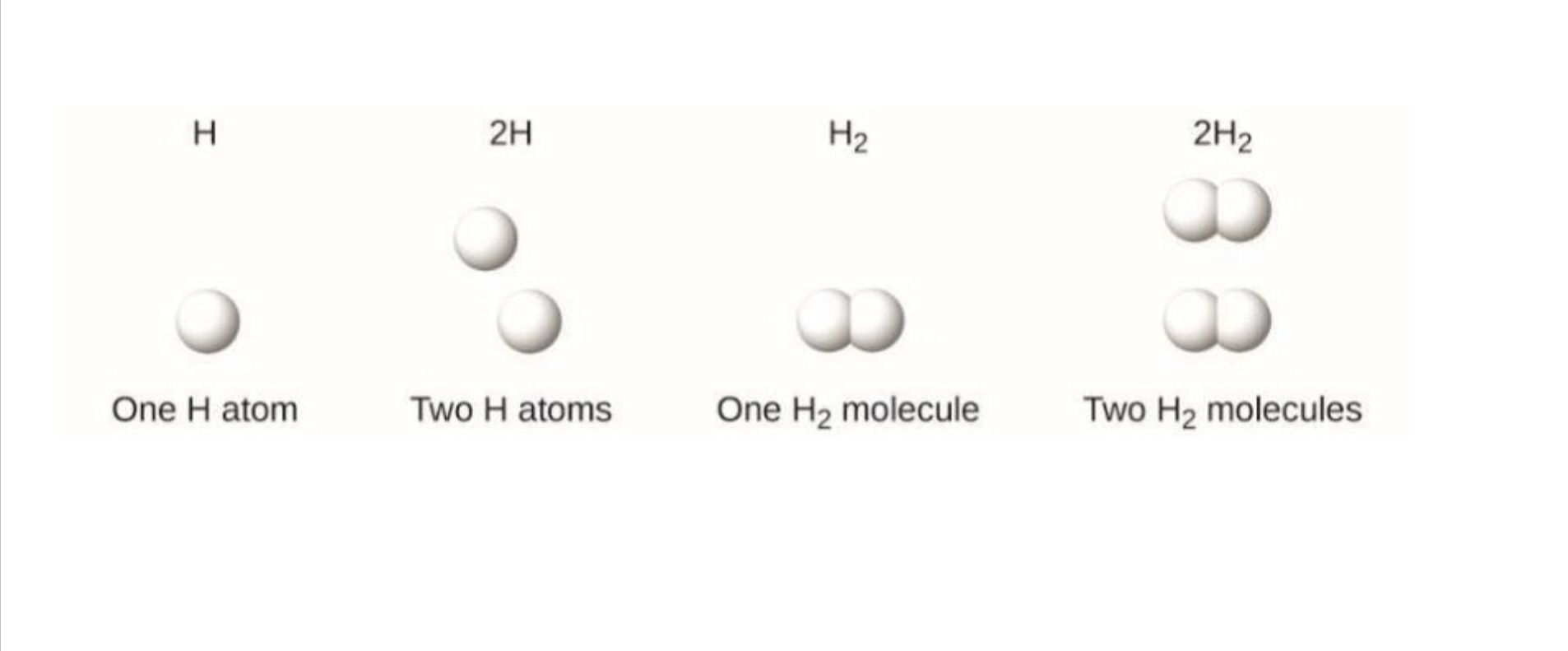
Subscript
Comes after a molecular formula
Indicates the ratio elements with a molecule
Empirical formula
Indicates the simplest whole-number ratio of the number of atoms (or ions) in the compound
Ex: C6H6 turns into CH
Isomers
Compounds with the same chemical formula but different molecular structures
One isomer has great differences in properties compared to the other
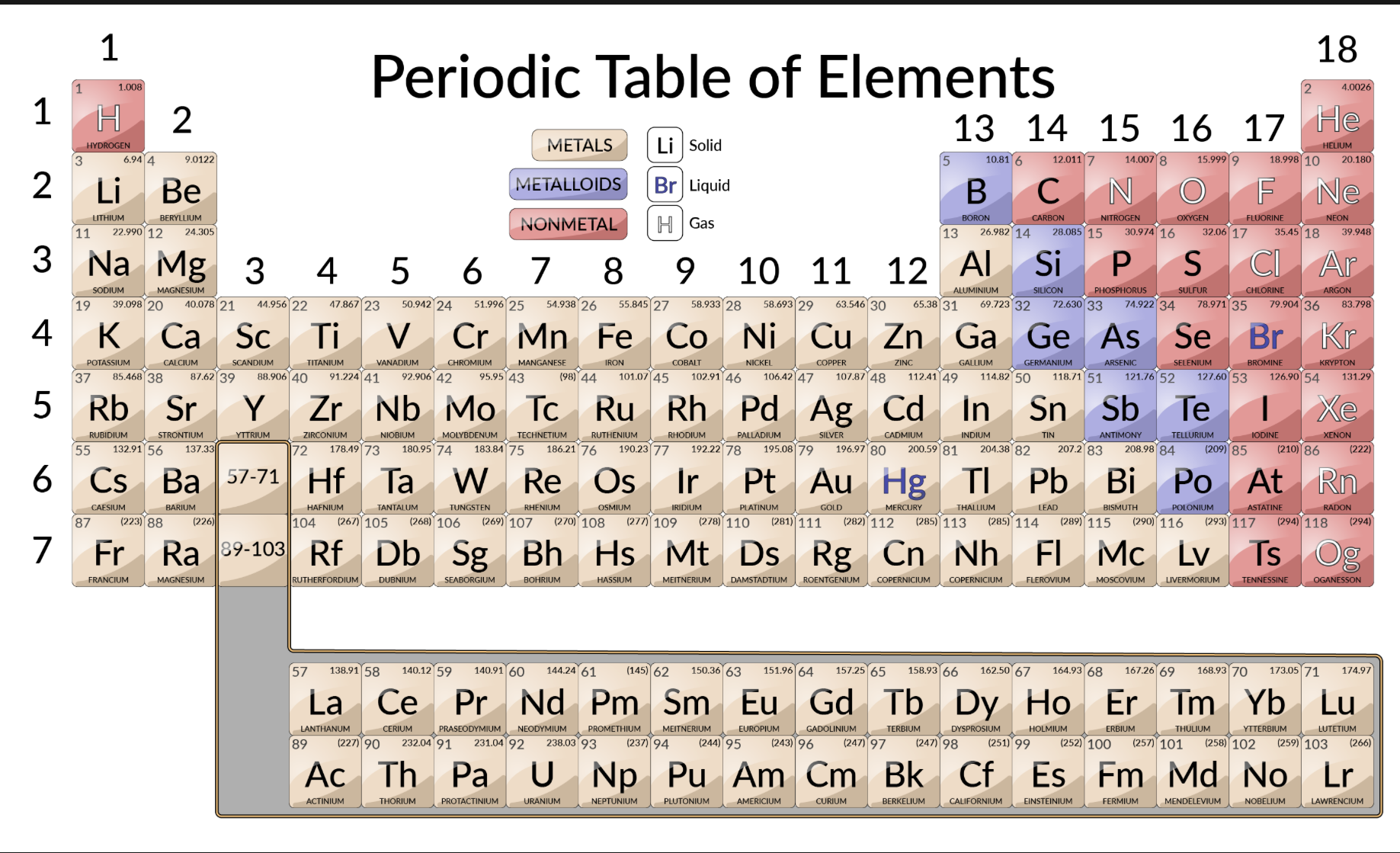
Metal classifications
Shiny solids, malleable, bendable, conductors, high melting and boiling point
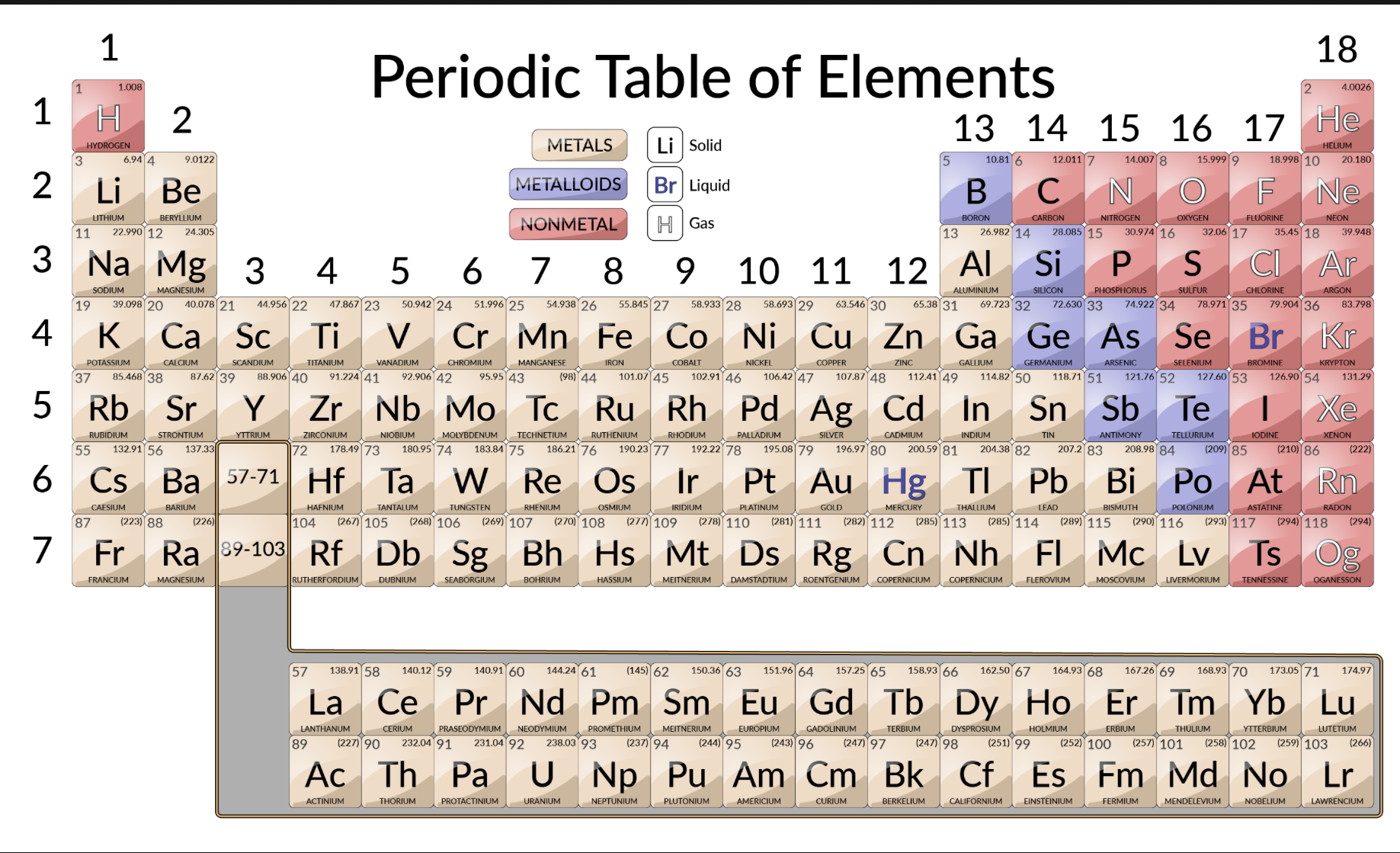
Nonmetal classifications
Brittle, insulators, low melting and boiling point
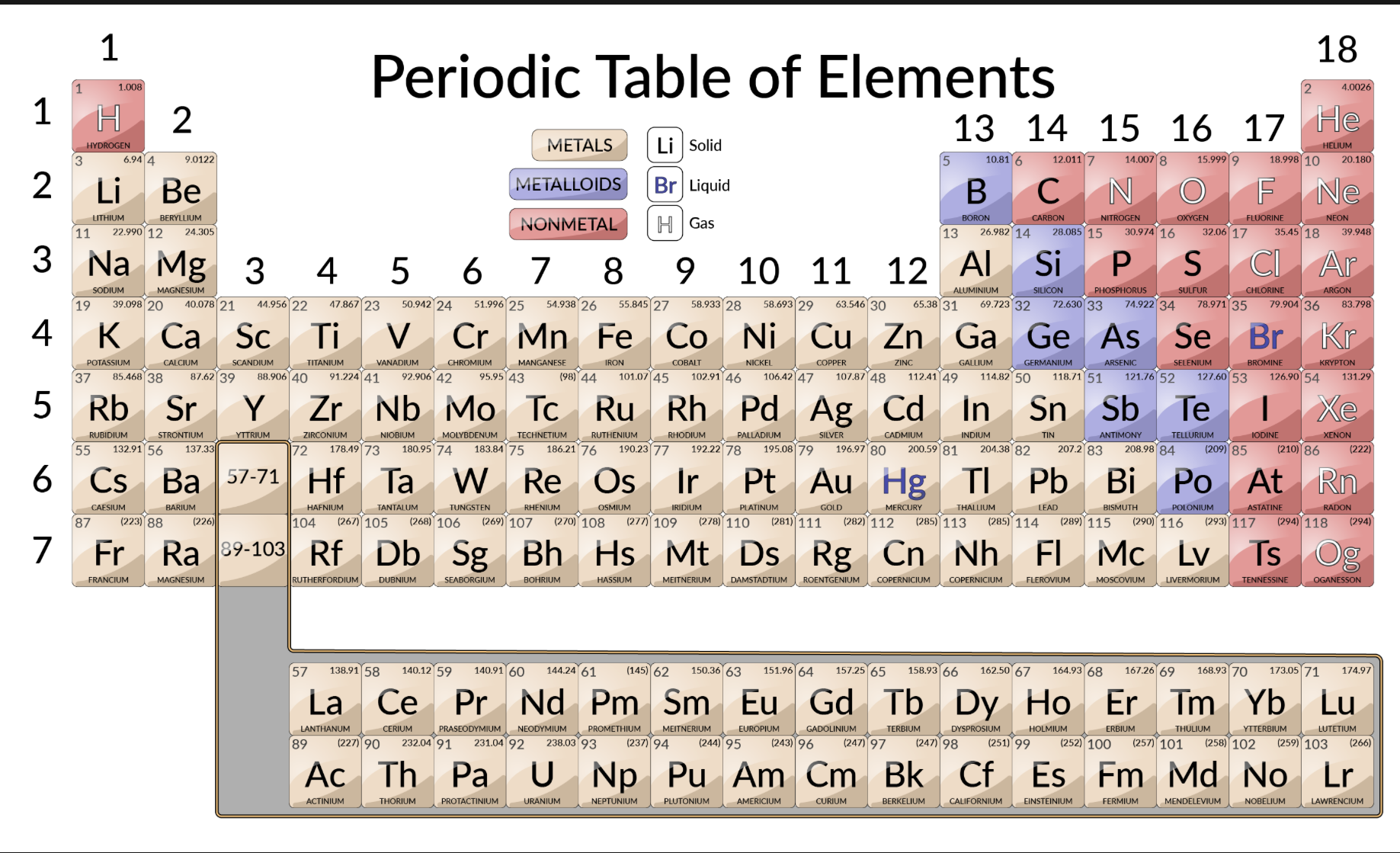
Semimetals
“Metalloid”
Can act as metal or nonmetal, semiconductors
Core electrons
Electrons in every orbital that isn’t the outermost
More tightly bound to the nucleus
Rarely involved in chemical reactions
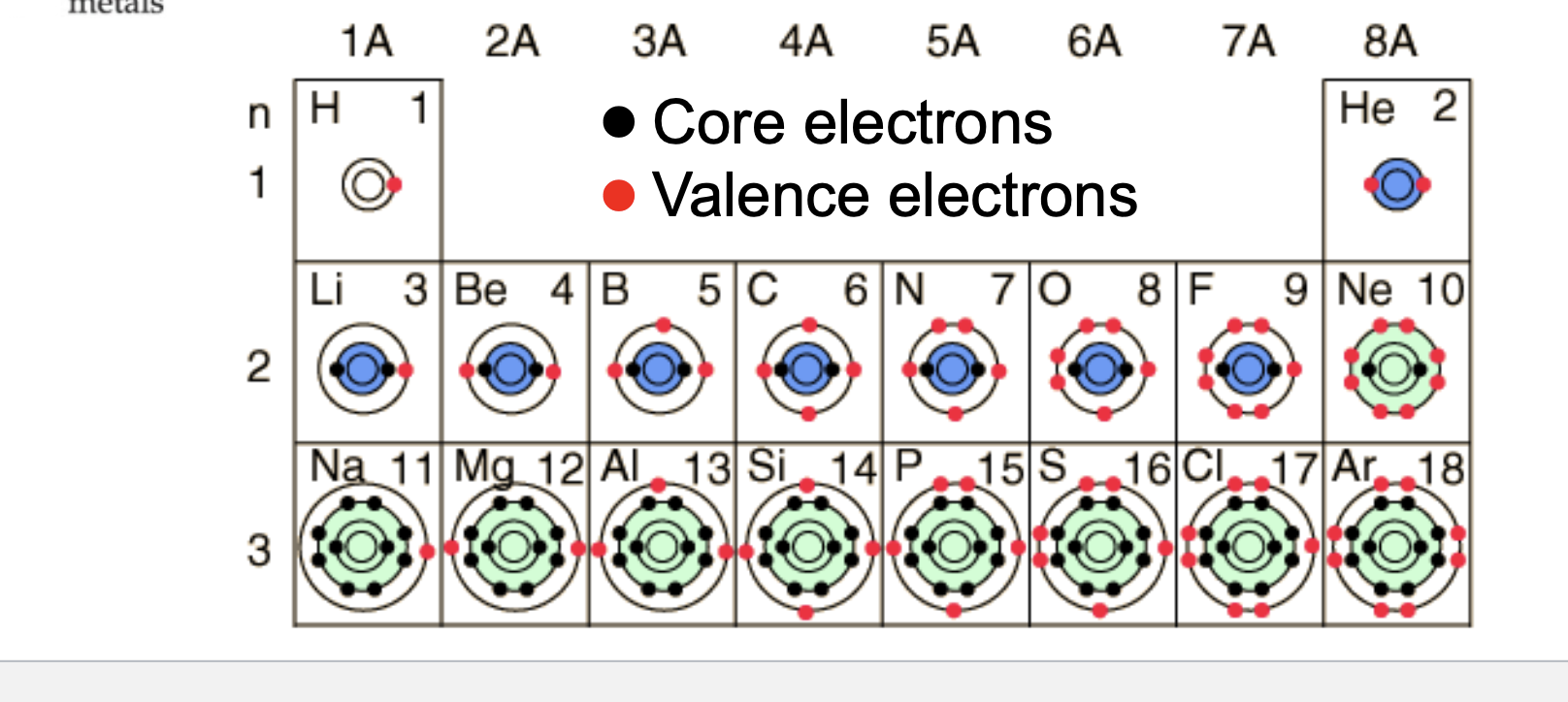
Valence electrons
Electrons in the outermost orbital
Responsible for chesty of atom
Amount an atom has equals it’s row number on the periodic table
Orbital
Pathway that electrons orbit the nucleus on
Very similar to the way planets orbit the sun
Amount an atom has equals it’s row number on the periodic table
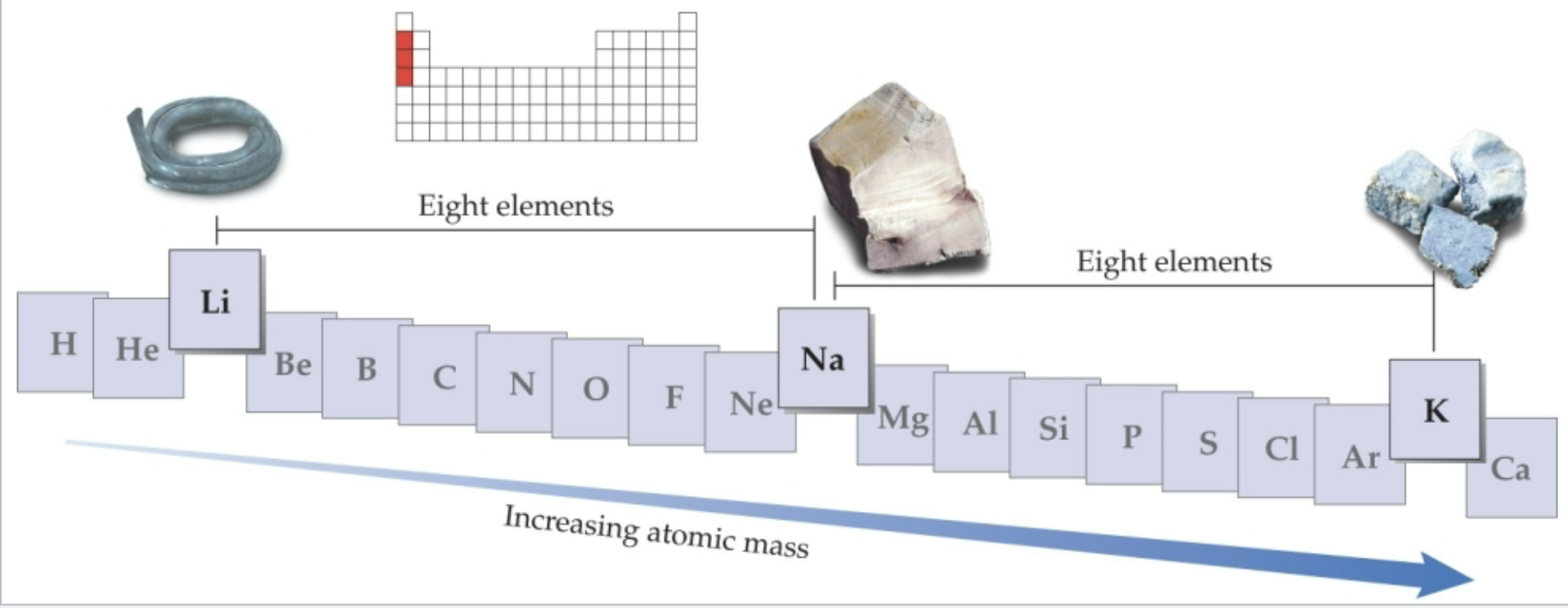
Law of Mendeleev - Chemical Periodicity
When elements are arranged in the periodic table by atomic mass properties repeated every 8 blocks
Ex. Lithium (Li) Sodium (Na) and Potassium (K) are all very soft metals and located 8 blocks away from one another
Magic Numbers
Ratio of neutrons to protons in a nucleus that makes the atom stable
Unknown to why there numbers exist