science revision
1/19
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
20 Terms
what is an Element?
A ‘pure’ substance, is comprised of 1 type of atom.
What is a molecule?
2 or more type of atoms that are bonded together
What is a compound?
2 or more different types atoms that are combined by chemical bonds
what type of charge does a proton have?
positive
What type of charge does a neutron have?
No charge, neutral
What type of charge does an Electron have?
Negative
How to calculate amount of neutrons using the atomic chart
mass number - atomic number = neutrons
How to calculate amount of protons / electrons using the atomic chart
atomic number = protons / electrons
Where are electrons held in Bhor’s model?
In the electronic shells around the nucleus
How many electrons can be held in each shell?
2, 8, 8, 18
where are the neutrons and protons in Bohr’s model?
In the nucleus
What is an Ion?
An ion is when the number of electrons in an elements are changed, changing the number of electrons won’t change the element but will change it’s chemical properties
Why do Ions occur?
Atoms always want to have full electron shells, an atom can gain or loose an electron to make a full outer shell
What is a positively charged ion called?
A Cation
What is a negatively charged ion called?
An Anion
How to write the symbol when an element gains an electron.
X 1-
X stands for the element symbol, the number 1 is changed for however many electrons are gained
How to write a symbol for when an atom looses an electron
X 1+
X stands for the element symbol, the number 1 is changed for however many electrons are lost
Nuclide symbol formation
Refer to image attached
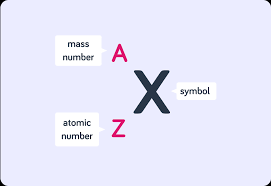
name the weight for protons, neutrons, and electrons
protons = 1 amu
neutrons = 1 amu
electrons = 0 amu
What is an isotope?
When an element protons stay the same but it’s atomic mass and neutrons differ