Chemistry- Ch 18: Amines and Amides
1/13
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
14 Terms
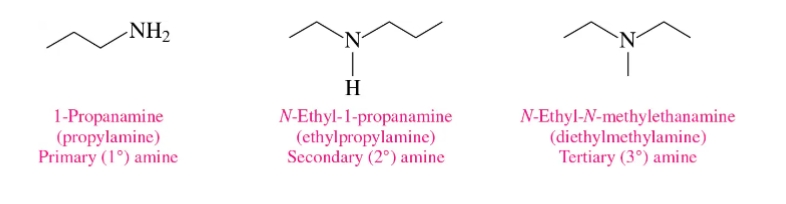
Naming Amines
“e” to “amine” ex. Methenamine
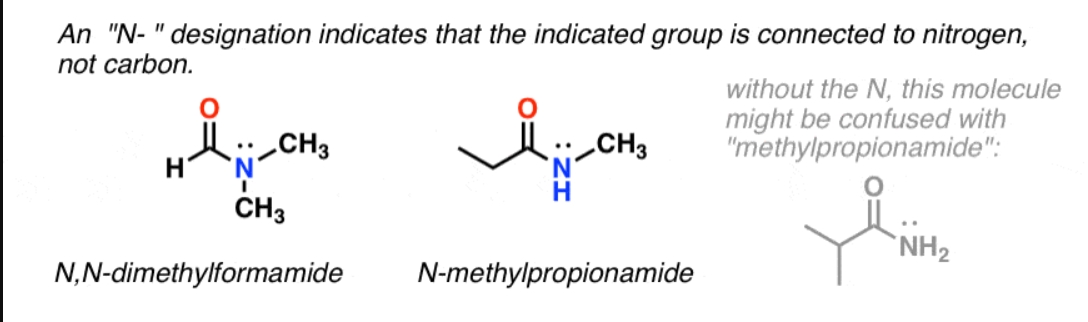
N- substituted amines
An N-substituted amine is a secondary () or tertiary () amine where one or more hydrogen atoms on the nitrogen are replaced by alkyl or aryl groups (R).
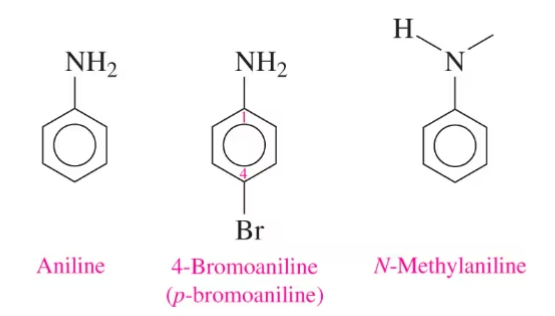
Aromatic Amines
compounds where a nitrogen group is attached directly to an aromatic ring (e.g., aniline).
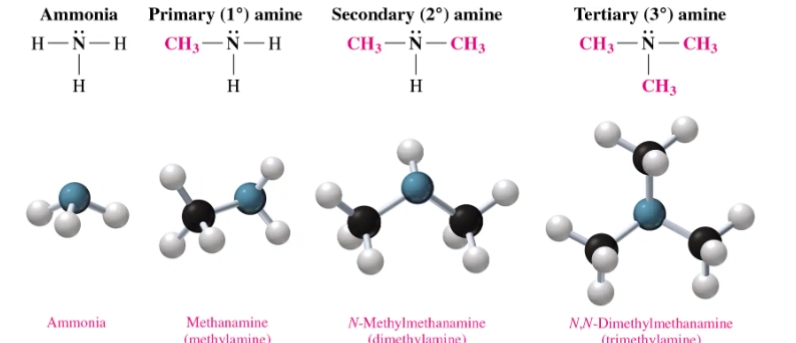
Substituted Amines
Substituted amines are organic compounds derived from ammonia () where one or more hydrogen atoms are replaced by alkyl or aryl (carbon-based) groups. They are classified as primary, secondary, or tertiary based on the number of organic groups attached to the nitrogen atom.
Skeletal Structures of Amines
When drawing amine skeletal structures, we always show the
hydrogens attached to the amine
• (Just like with alcohols)
Amines and hydrogen bonding
some bond w/ hydrogen
Amine Solubility
Hydrogen bonding increases solubility in water.
Amines
are usually bases and form ammonium salts (or ammonium ions) in water
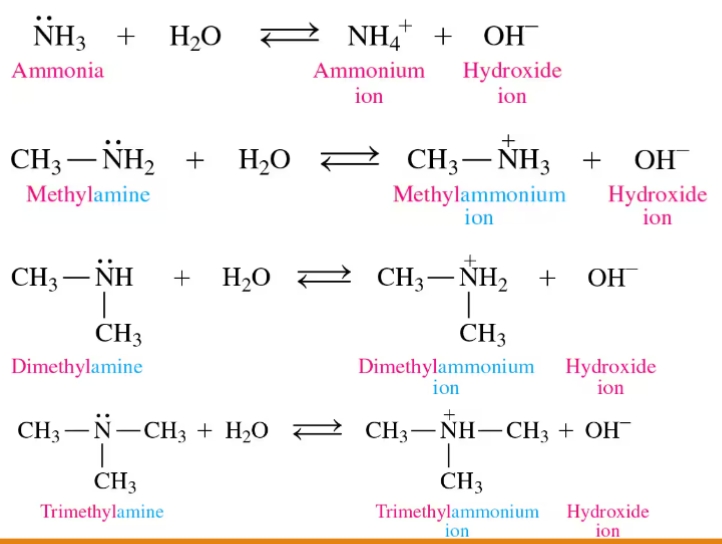
Neutralization of Amines
Neutralizing amines with acids leads to the formation of only ammonium salts. Ammonium salts do not boil in most cases. Common salts are ammonium
chlorides and ammonium bromides
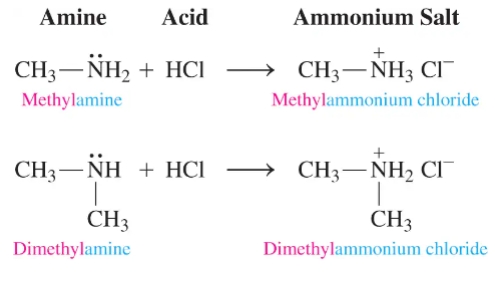
Ammonium Salts
highly water-soluble, generally white crystalline ionic compounds formed by the reaction of ammonia with acids.
Heterocyclic Amines
mutagenic and carcinogenic compounds containing at least one heterocyclic ring
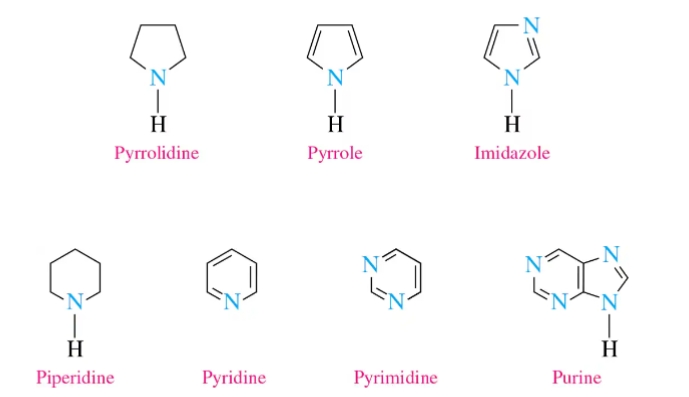
Alkaloids
They contain at least one nitrogen atom, usually in a heterocyclic ring, derived from amino acids.
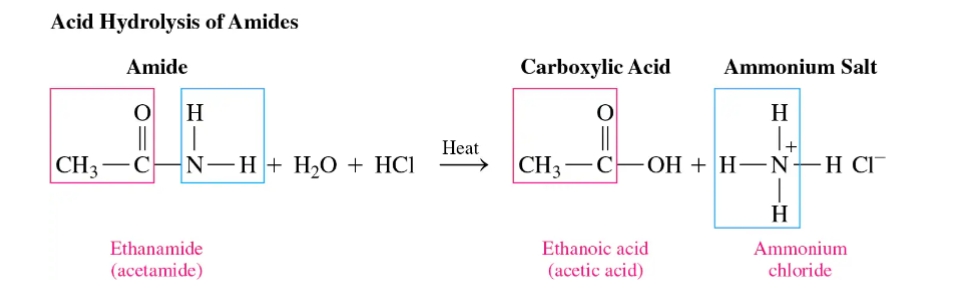
Acid Hydrolysis of Amides
involves heating it with an aqueous acid (like HCl) to break the amide bond, yielding a carboxylic acid and an ammonium salt
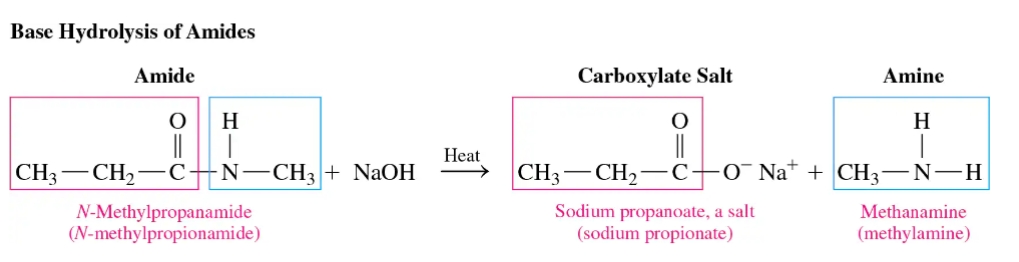
Base Hydrolysis of Amides
Basic hydrolysis of an amide involves heating it with a strong base (e.g., NaOH) to yield a carboxylate salt and an amine or ammonia.