Acid-base balance
1/30
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
31 Terms
Acid
release H+ ions. aka a proton donor (a Hydrogen atom that has lost an electron to become an ion)
Base
a proton acceptor
any substance that binds to H+ ions
important bases in the human body: bicarbonate ion (HCO3-)
pH scale
• A means of referring to the H+ concentration in a solution
• Pure water defined as neutral (pH 7) – Equal concentrations of H+ and OH-
• Change of 1 pH unit represents a 10-fold change in the H+ concentration
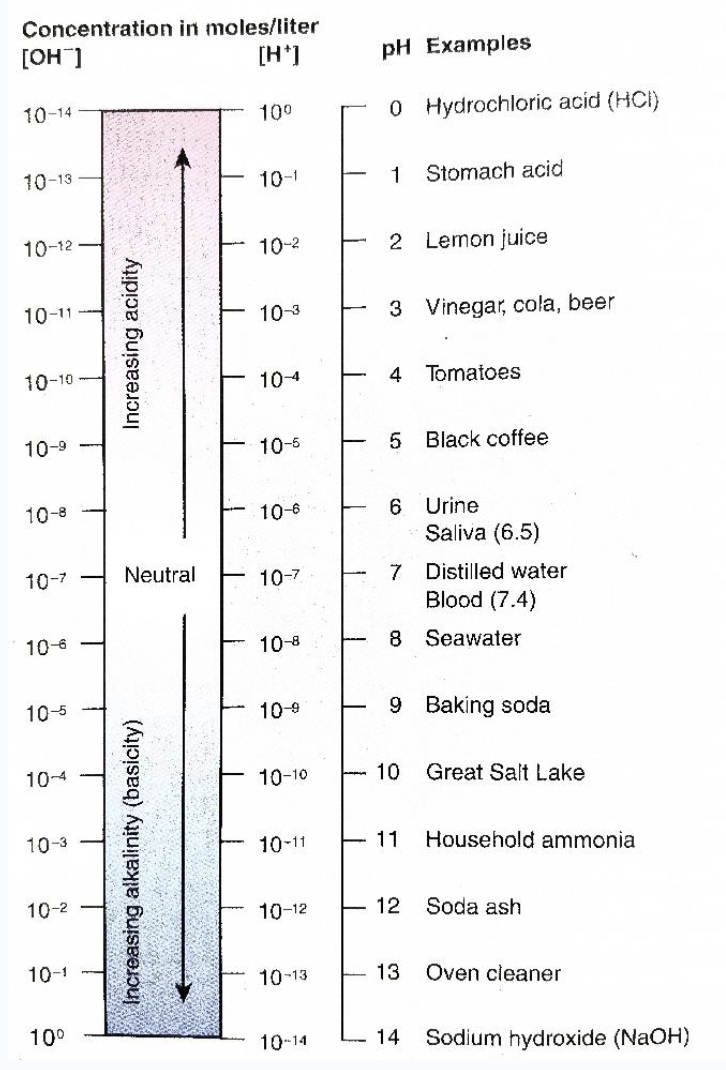
Strength of acids and bases
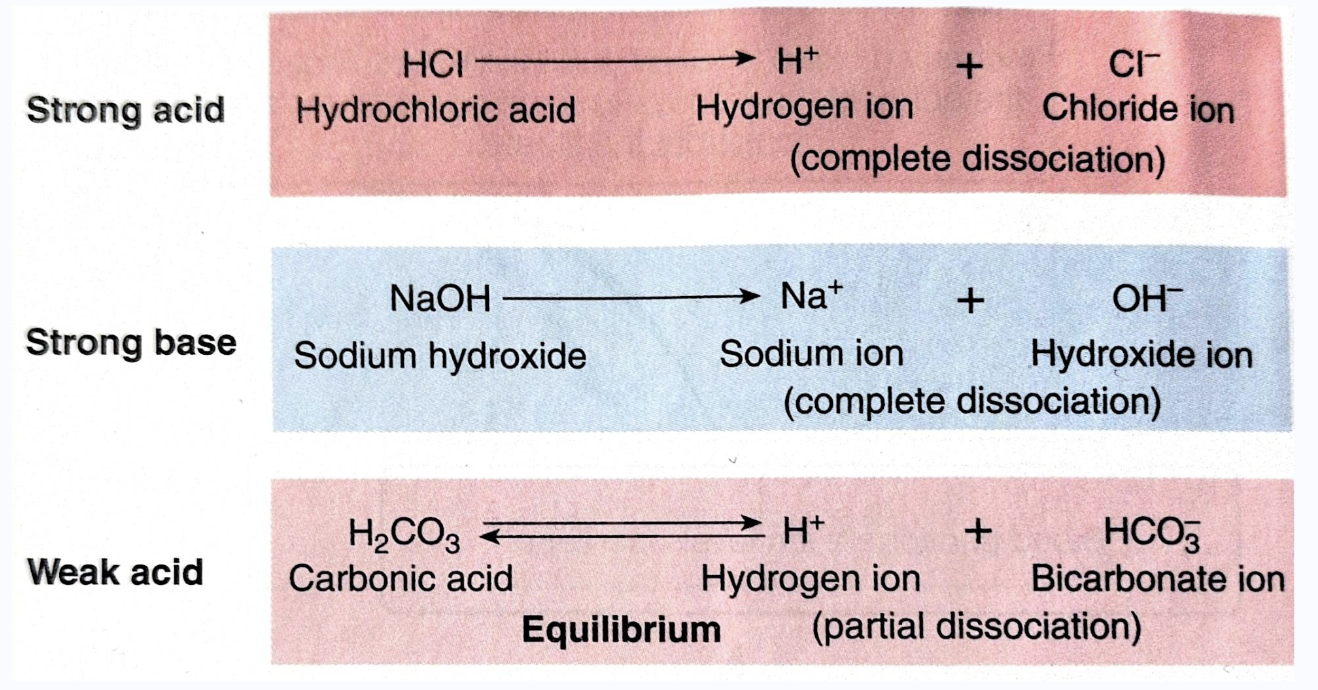
strong: lots of H+ ions dissociate into solution
weak: some H+ ions released (not all molecules dissociate entirely, some remain intact)
Functions of water
– Stabilising body temperature
– Protection
– Chemical reactions
– Mixing medium
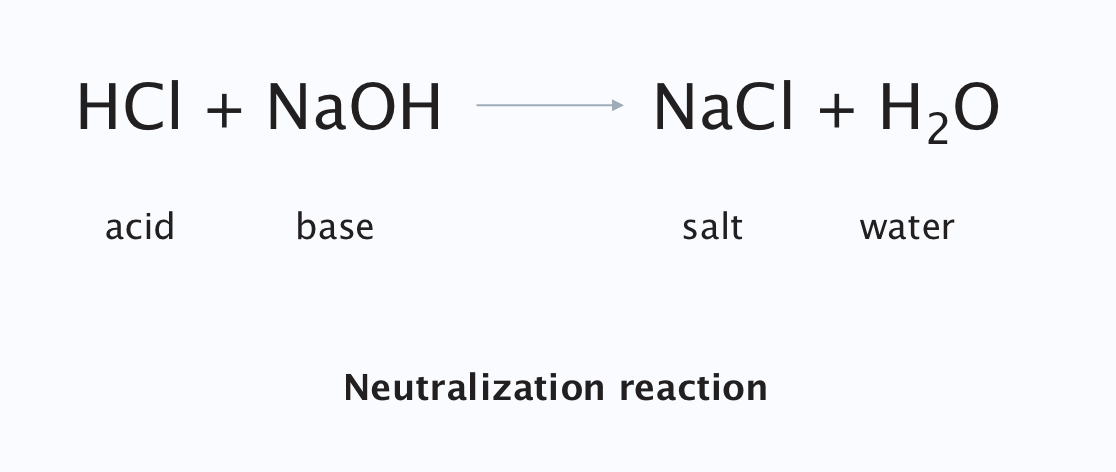
When acids and bases mix
Neutralisation reaction
How do buffers work?
Resist swings in pH by:
releasing H+ ions
Binding H+ ions
(depending on whether pH rises or falls)
Main chemical buffer systems
carbonic acid/ bicarbonate buffer system
protein buffer system
Phosphate buffer system
Carbonic acid/ bicarbonate buffer system
• Carbonic acid (H2CO3) = weak acid = dissociates easily
• Depends on the REVERSIBLE reaction
When the pH is low, more H+ ions bind to bicarbonate to form carbonic acid - then converted into CO2 to breathe out
when pH too high = carbonic acid dissociates into H+ and bicarbonate
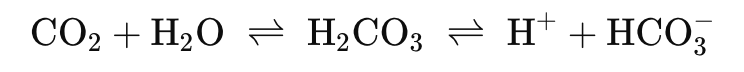
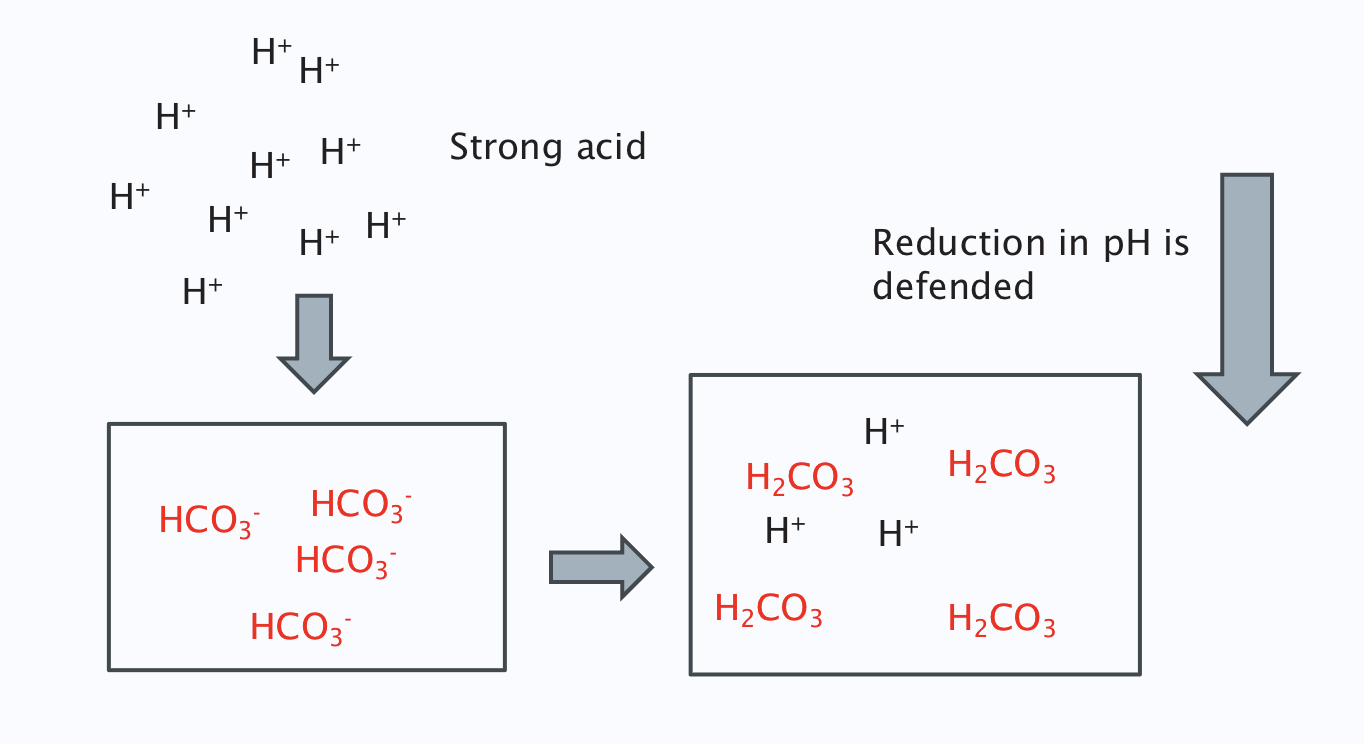
When a strong acid is added to the solution
H+ binds to bicarbonate to form carbonic acid which dissociates into CO2 and water
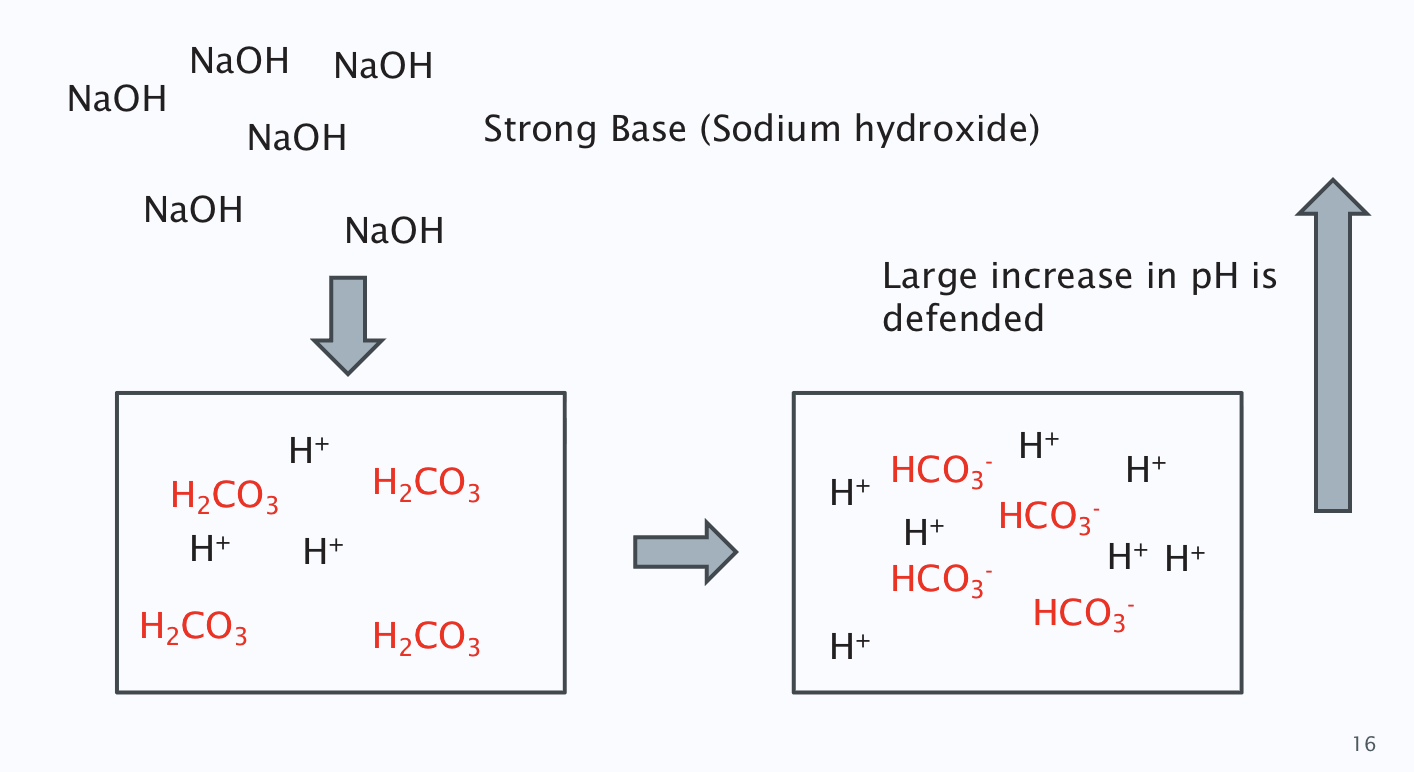
When a strong base is added to system
bicarbonate ions released
carbonic acid dissociates into H+ to bind to the H+ receptors
Buffer capacities
Carbonic acid buffer capacity = low
Protein buffer capacity = high due to where protein buffers are located
Protein buffer
• Intracellular proteins and plasma proteins form a large pool of protein molecules, which act as buffer molecules
• Provides ~3/4 of buffering capacity due to high concentration
• Haemoglobin important intracellular protein
Phosphate buffer solution
• Concentration of phosphate and phosphate containing molecules is low in the extracellular fluid compared to other buffers
• Important for inside cells
H₂PO₄⁻ = dihydrogen phosphate (acts as a weak acid)
HPO₄²⁻ = hydrogen phosphate (acts as a weak base)
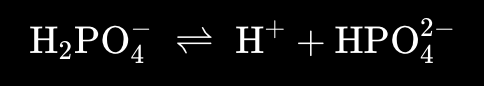
Regulation of acid-base balance
mechanisms of acid-base balance
buffer systems
respiratory system
renal system
changes to pH in body fluids
buffers respond instantly
respiratory system responds within a few mins
renal system responds more slowly (hours to days)
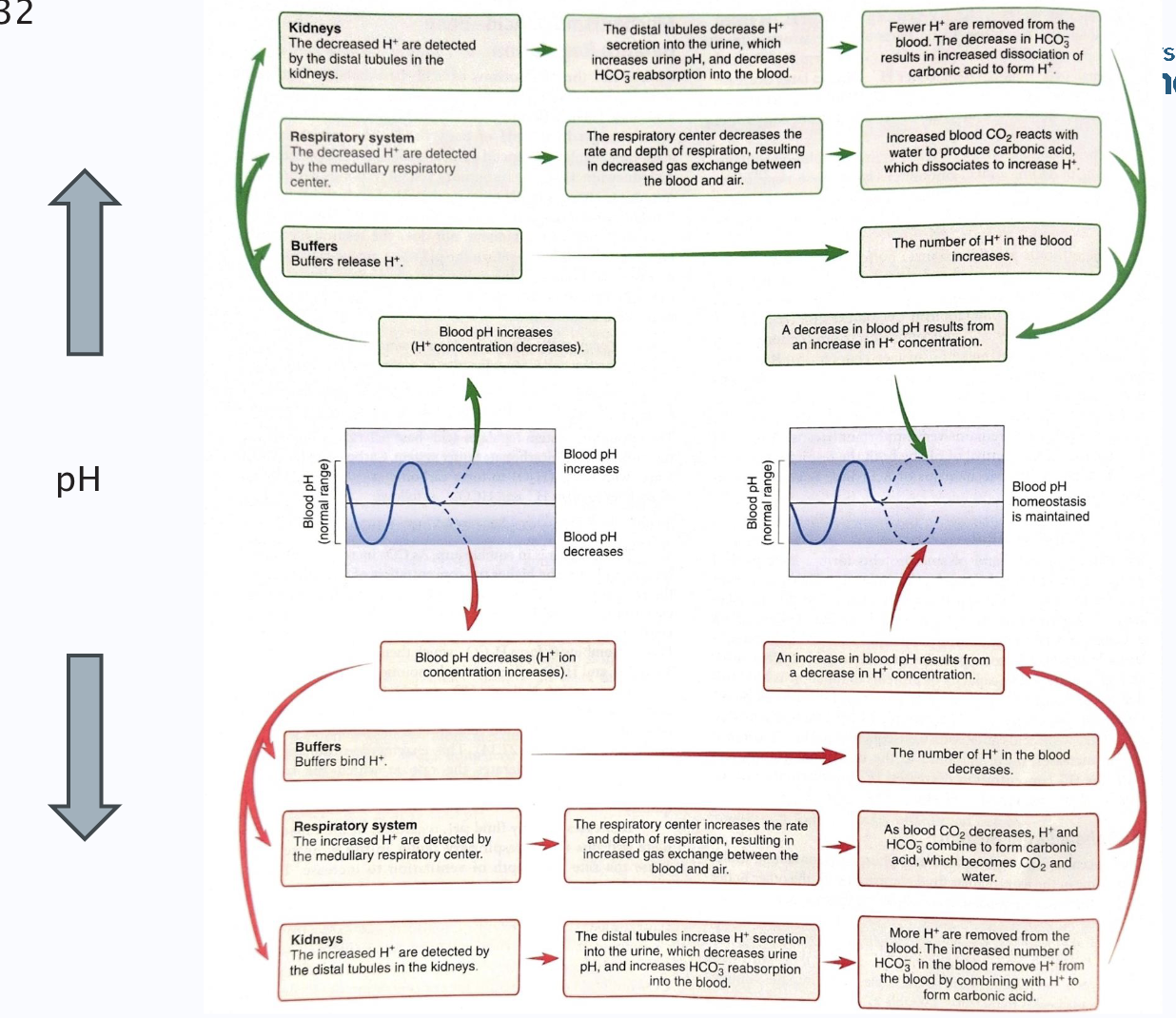
Mechanisms of homeostasis of blood pH
(both respiratory and renal systems)
Respiratory regulation of acid-base balance
Respiratory system influences the carbonic/bicarbonate acid
buffer system
CO2 reacts with H2O
– H2CO3 (carbonic acid)
• Dissociates to form
– H+ and HCO3-
H2O + CO2 H2CO3 H+ + HCO3-
Effect of hyperventilation on blood pH
Blood becomes more alkaline (due to more CO2 being breathed out)
Kidney regulation of acid-base balance
(dont need to learn the diagram, only that there are active transport molecules being used)
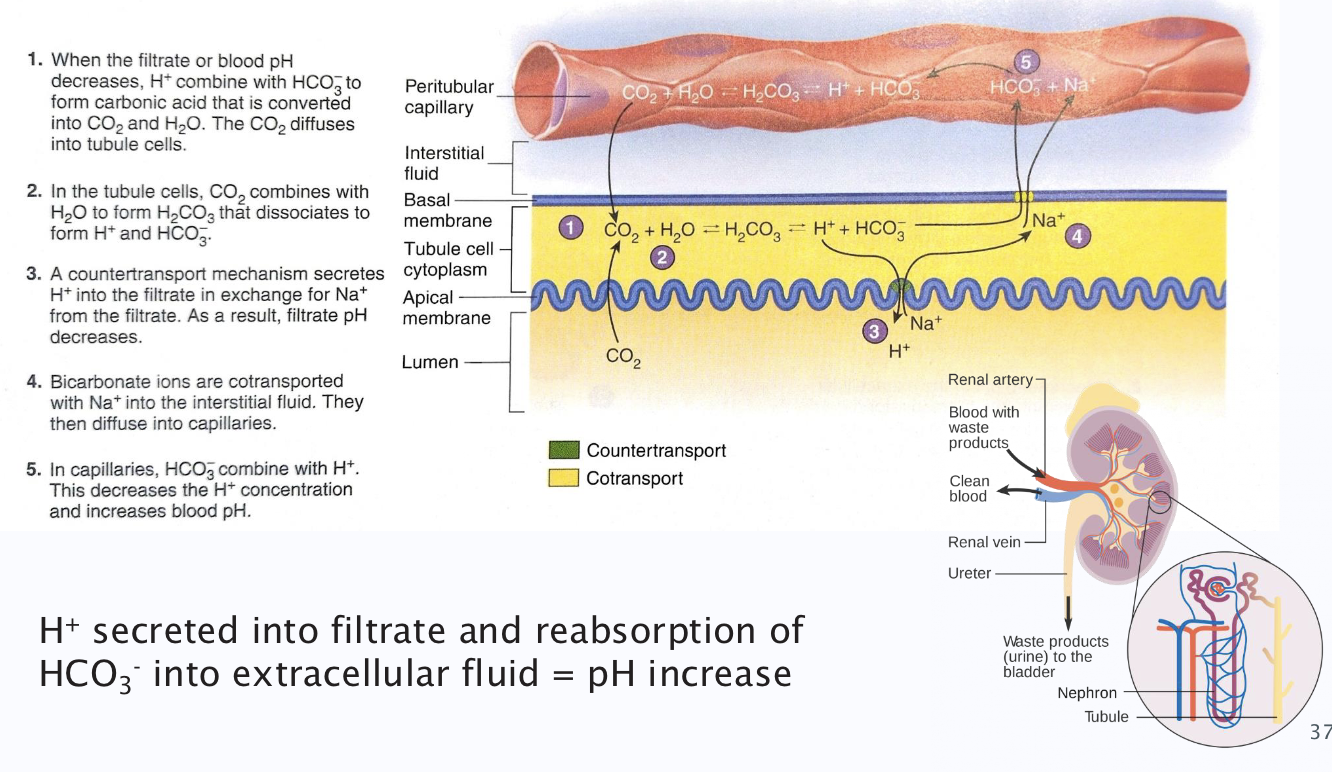
Normal pH value of body fluids
between 7.35 and 7.45
pH value of body fluids below 7.35
Acidosis
pH value of body fluids above 7.45
Alkalosis
Acidosis
Metabolism produces acidic products that lower the pH of body fluids
– CO2 is a by product of metabolism
– Combines with water to form carbonic acid
– Anaerobic metabolism produces lactic acid
– Failure to eliminate and regulate acidic substances causes drop
in pH = acidosis
– Excess elimination = alkalosis
Types of acidosis and alkalosis
respiratory acidosis or alkalosis
metabolic acidosis or alkalosis
Respiratory acidosis
– Reduced elimination of CO2 from body fluids
– Example conditions: COPD, asthma, asphyxia, hypoventilation
• Inadequate ventilation of the lungs causes respiratory acidosis
– Rate of CO2 removal is reduced
– Increases concentration of CO2 in body fluids
– Increased CO2 reacts with water to form carbonic acid (H2CO3_)
– Carbonic acid forms H+ and HCO3-
– Increased H+ causes decrease in pH
– Body fluid pH falls below 7.35 = respiratory acidosis
Major effect of respiratory acidosis on the body
depression in central nervous system (malfunctions, presents as individual becoming disorientated, dizzy, coma)
Respiratory alkalosis
– Reduced CO2 levels in the extracellular fluid
– Example conditions: hyperventilation, high altitude (decreased atmospheric pressure stimulates chemoreceptors to hyperventilate)
• Results from hyperventilation of the lungs
– Increased rate at which CO2 is removed from the body
– Decreased concentration of CO2 in the body fluids
– Increase in pH above 7.45 = respiratory alkalosis
Major effect of alkalosis on the body
hyper-excitability of the peripheral nervous system (causes nervous stimulation in muscles = spontaneous muscle spasms like respiratory muscles = death)
Metabolic acidosis
elimination of large amounts of bicarbonate (HCO3-)
example conditions: extreme vomiting/ diarrhoea, taking acidic drugs, production of lots of fatty acids/ metabolites
Metabolic alkalosis
elimination of large amounts of H+ and reabsorption of HCO3- in the stomach or kidney
examples of where this can occur: ingest excessive quantities of sodium bicarbonate