Inorganic Chemistry ACS Final Exam
1/105
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
106 Terms
Define this:
Energy required to remove the least tightly bound electron from a neutral atom in the gas phase
Ionization Energy
What is the periodic trend in ionization energy?
Lowest Energy → Bottom Left Coroner of the Table
Increases going up and to the right (same as effective nuclear charge)
Why are half-filled subshell so stable?
They maximize stabilizing interactions
Define this:
πe
It is a result of electrons pairing in degenerate orbitals with parallel spin.
Electron Interaction
Define this:
A destabilizing interaction, the Coulomb interaction, πC, and it is the energy of electron-electron repulsion in a filled orbital.
Pairing energy
What is harder to ionize a high energy electron or low energy electron?
A high energy electron is easier to ionize. It already contains more energy, so the input to remove it from the atom is less.
If a 3d series metal is ionized, what orbital loses the electron? What happens to the valence configuration?
If a d series metal has the configuration 4s23dn, the first electron to be ionized will come from the 4s orbital. The remaining s orbital will then enter a d orbital, giving the configuration of the +1 cation 4s03dn+1.
Define this:
F orbitals are poor at shielding, so any electrons added into the subshell following the f orbital experience a higher Zeff, shrinking their radius over what is to be expected from overall trends.
Lanthanide Contraction
Define this:
Formally, it is written as Zeff= Z – σ.
Z is the atomic number, and σ=Σsi
Slater’s Rules
From Slater’s Rules, for s and p valence electrons, (n-2) groups or lower contribute _ to si (weighting coefficient of 100%).?
1.00
From Slater’s Rules, (n-1) groups contribute to 0.85 to sl?
0.85
From Slater’s Rules, n(s,p) groups contribute?
0.35
From Slater’s Rules, the d and f valence electrons contribute:
Valence shell = 0.35
(n-1,2,etc.) = 1.00
Define this:
The reduction in charge attraction between the nucleus and electrons, due to electrons between the nucleus and the electron in question. An electron is ‘between’ the nucleus and the electron being considered if it has a lower energy.
Shielding
Define this:
When an electron of a higher atomic orbital is found within the shell of electrons of lower atomic number. That is to say that an electron of higher energy is found within an orbital of lower energy.
Penetration
Define this:
Atom’s ability to be distorted by an electric field. Basically, it means that regions of a molecule can take on partial positive, or partial negative charge.
Polarizability
Define this quantum number:
The principle quantum number. It defines the energy and size of the orbital.
Expected: 1, 2, 3, 4, etc
N
Define this quantum number:
Orbital angular momentum quantum number: it defines the magnitude of the orbital angular momentum, as well as the angular shape of the orbital.
Expected Values: 0 to n-1
L
Define this quantum number:
Magnetic Quantum number: Describes the orientation of the angular momentum.
Expected Values: 0 to ± L
M
Define this quantum number:
Spin magnetic quantum number: Defines the intrinsic angular momentum of an electron.
Expected Values: +1/2 or -1/2.
Ms
Define this:
When degenerate orbitals are available for occupation, electrons occupy separate orbitals with parallel spin.
Hund’s Rules
Define this:
No more than two electrons can occupy a single orbital, and to do so, their spins must be paired.
Pauli Exclusion Principle
Predict this molecular shapes based on VESPR:
Number of Bonded Electrons: 1
Number of Lone Pairs: 3
Total Electron Pairs: 4
Trigonal Pyramidal (107.3)
Predict this molecular shapes based on VESPR:
Number of Bonded Electrons: 2
Number of Lone Pairs: 2
Total Electron Pairs: 4
Bent (104.5)
Predict this molecular shapes based on VESPR:
Number of Bonded Electrons: 1
Number of Lone Pairs: 4
Total Electron Pairs: 5
See-saw (90, 120, 180)
Predict this molecular shapes based on VESPR:
Number of Bonded Electrons: 2
Number of Lone Pairs: 3
Total Electron Pairs: 5
T-Shape (90)
Predict this molecular shapes based on VESPR:
Number of Bonded Electrons: 1
Number of Lone Pairs: 5
Total Electron Pairs: 6
Square pyramidal (90)
Predict this molecular shapes based on VESPR:
Number of Bonded Electrons: 2
Number of Lone Pairs: 4
Total Electron Pairs: 6
Square Planar (90)
What is the effect of a lone pair on geometry?
A lone pair pushes strongly against all other substituents. It is the strongest steric force governing the shape of a molecule.
Define this:
It is an improvement over valence bond theory in that the bonding description extends to all atoms in a molecule, and handles polyatomic molecules easily. Atomic orbitals combine to form molecular orbitals, which are delocalized descriptions of electron distribution.
Thrives off of key assumptions:
Orbital Approximation
Linear Combinations of Atomic Orbitals
MO Theory
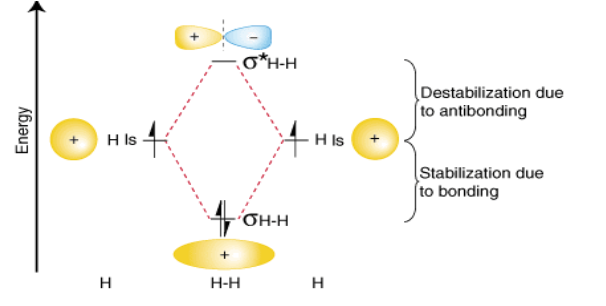
What type of MO is this?
Homonuclear Diatomic
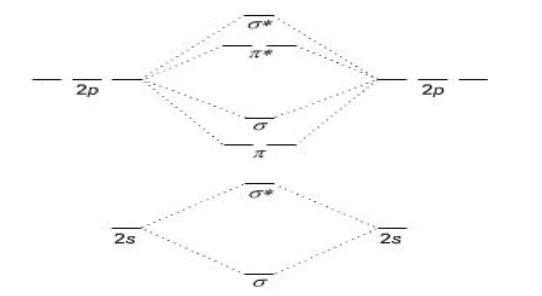
Which atoms use this orbital diagram?
Li2 - N2
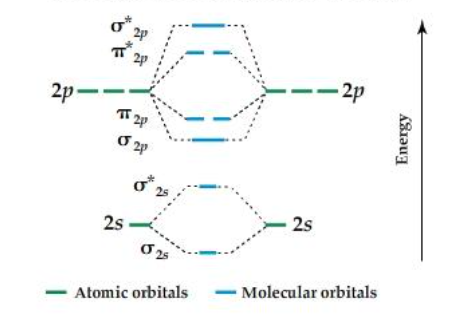
What atoms use this orbital diagram?
O2, F2,
For X-Y, heteroatomic diagram, why most we consider the electronegativity of the atom?
The more electronegative the atom, the lower in energy its orbitals will be.
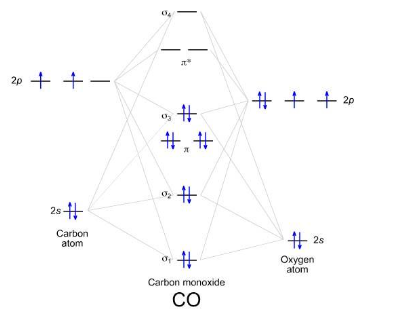
What is the formula for bond order?
B = ½ (n-n*)
Define this:
Highest occupied molecular orbital. It is where the highest energy electrons reside.
HOMO
Define this:
Lowest unoccupied molecular orbital. It is the energy level directly above the HOMO.
LUMO
Define this:
Two unpaired electron in MO diagram
Paramagnetism
Define this:
No unpaired electrons in MO diagram
Diamagnetism
Define this:
A 3D infinite array of (lattice) points which define the repeating structure of a crystal.
Lattice
Define this:
An imaginary, parallel sided region from which the entire crystal can be built, such that it fits perfectly together, giving rise to a crystal system. There are 7 types of crystal systems
Unit Cell
Define this:
ABCABC repeating layers of spheres. (ABAB results in hexagonally close packed).
Cubic closed-packed
Define this:
The ability of a metal to adopt different crystal structures based on the temperature and pressure.
Polymorphism
Define this:
A type of flow chart used to determine lattice enthalpies and other parameters governing how a solid behaves.
Born-Haber Cycle
What is the most stabilizing influence on a solid? Set up a proportion to show how it changes along with the values, and relate this to different types of systems.
Lattice enthalpy is the most stabilizing influence on a solid.
ΔHL0 ≈ q1q2/d,if the charge is high and distance small, lattice enthalpy is very strongly stabilizing. If the charge is small and the distance is great, the solid is not especially stable. ΔHL0 values are at their highest when the solid is tightly packed, and the ions are charge dense
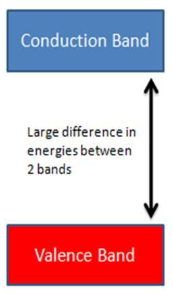
What type of (insulator, conductors, or semiconductors) is this?
Insulators

What type of (insulator, conductors, or semiconductors) is this?
Conductors

What type of (insulator, conductors, or semiconductors) is this?
Semi-conductors
Define this (semi-conductor):
The dopant supplies (Group V) additional electrons to the solid structure. These electrons lie above the valence band in energy, and close to the conduction band, so the gap is much easier to jump, thanks to the donor band.
N-type
Define this (semi-conductor):
The dopant material (Group III) has less valence electrons than the parent material. The holes it creates lie just above the valence band in energy, so electrons can jump to the acceptor band to allow current to flow.
P-type
Define this;
Electron Pair acceptor
Lewis Acid
Define this:
Electron Pair Donor
Lewis Base
Soft acids are typically………?
Larger and more polarizable
What is the relationship between a strong acid and its conjugate base?
A strong acid seeks to remain deprotonated, while a strong base seeks to remain protonated.
How is the Bronsted definition of acids and bases extended by the solvent system definition?
The Bronsted definition is only concerned with proton transfer.
Acid as any solute that increases the concentration of the cation generated by auto ionization of the solvent.
Base as anything that increases the concentration of the anion generated by the auto ionization of the solvent.
How do you charge balance a redox reaction?
MnO4-(aq) + H2SO3(aq) -> Mn2+(aq) + HSO4-(aq)
→ First, separate it into half reactions
MnO4-(aq) + 5e- + 8H+ -> Mn2+(aq) + 4H2O(l) Eo= +1.51V
*This half reaction was obtained from a Latimer diagram. The Latimer diagram only shows the species which are present, the oxidation state, and the standard cell potential. You have to be able to figure out the number of electrons to add, and because the problem specified that it is in acid, how many H+ to add.
HSO4-(aq) + 2e- + 3H+(aq) -> H2SO3(aq) + H2O(l) Eo= +0.158V
*Same as for the other half reaction, make sure everything adds up
We have to get the number of electrons to equate. They will be on opposite sides of the reaction when it is written out completely.
Multiply the top by 2, and the bottom by 5
Because HSO4- is the product, we need to reverse the half reaction containing that species.
2MnO4-(aq) + 10e- + 16H+ + 4H2SO4 + 5H2O -> 2Mn2+ + 8H2O + 5HSO4 + 10e- + 15H+
1H+ 3H2O
Final balanced equation: 2MnO4-(aq) + H+(aq) + H2SO3(aq) -> 2Mn2+(aq)+3H2O(l)+5HSO4-(aq)
Under what conditions might a reaction be thermodynamically favorable but kinetically slow?
If Eo>0, it is spontaneous, but if it requires the movement of multiple electrons simultaneously, it will be slow.
Discuss the pH dependence of redox reactions?
As the pH increases, E decreases. As the pH decreases, E increases.
Define this molecular symmetry:
Rotation by 360o/n.
If n=2, there is only one rotation operation involved. If n=3, there are two operations associated with it’. There will always be n-1 rotational operations involved, only considering that axis.
Cn
Define this molecular symmetry:
The identity operator. It is always there.
E
Define this molecular symmetry:
A mirror plane
σ
Define this molecular symmetry:
Operator consists of a rotation through angle followed by a reflection in a mirror plane perpendicular to the rotation
Improper Rotation
Helpful Card for assigning point group
Is it linear? If yes, there are only two options. If it has a center of inversion it is D∞v, if it does not, it is C∞v.
If it is not linear, then we first consult the rotational symmetry elements.
If it has two or more unique Cn axes with n>2, given the axes are not collinear, it is Td, Oh, or Ih. If it does not have a center of inversion, it is Td. If it does have a center of inversion and a C5 axis it is Ih. If it does not have a C5 axis it is Oh.
If it does not have two unique C axes that are not colinear, does it have rotational symmetry? If yes, then…
Is the principal axis perpendicular to the C2 axes? If it is, it is Dnh, Dnd, or Dn. If it has σh symmetry, it is Dnh. If no, consult, σd. If it has σd symmetry, it is Dnd, if not, it is Dn.
If the C axes are not perpendicular, it is either Cn, S2n, Cnv, or Cnh. To determine the point group, consult the σh symmetry element. If it does have a horizontal mirror plane, it is Cnh. If there is no horizontal mirror plane, if it has a vertical mirror plane it is Cnv. If it has a screw axis it is S2n. If it is does not, it is Cn.
If there is no rotational symmetry, it is Cs, Ci, or C1. Does it have a mirror plane? If yes, it is Cs if no, does it have a center of inversion? If yes, it is Ci, if no, it is C1.
What type of groups these?
D∞v, C∞v, Ih, Oh, Td
High symmetry
What type of groups these?
Cs, Ci, C1
Low Symmetry
What is the formula for vibrational nodes of non-linear molecules?
3N - 6
What is the formula for vibrational nodes of linear molecules?
3N - 5
In nomenclature for coordination compounds, what is written first?
Cation, Anion and then ligands (alaphabetically)
Define this:
The term for a complex which forms when a polydentate ligand binds to a metal with more than one of its donor atoms. They tend to bind at adjacent sites in a cis configuration.
Chelate
What group on the periodic table is known to form 2 coordinate complexes?
Group 11
What type of isomer is this?
Define this:
Depict the energies of electronic states of complexes as a function of the ligand field strength. More specifically, it shows terms splitting into different microstates.
Tanabe-Sugano Diagram
Define this:
Used for 3d metals, and is the process of combining electron angular momenta by first summing spins, then orbital momenta, and then combining the procedure
Russel-Sanders Coupling
What is the formula for total spin in a term symbol?
2S + 1
What orbital does L = 0 represent?
S → A1g
What orbital does L = 1 represent?
P → T1g
What orbital does L = 2 represent?
D → T2g and eg
What reduces what?
The ligand reduces the metal ion.
What characteristics of the metal atom will give a strong CT band?
Metal Ion in a high oxidation state
What characteristic of the complex makes a MLCT likely to occur?
Low Lying Pi antibonding orbitals, especially with aromatics
What spin is allowed to transitions from singlet to triplet?
Cannot undergo transitions, forbidden
Define this:
Transitions from g to u terms are allowed, but those from g to g, and u to u are forbidden.
Forbidden transitions are s-s, p-p, d-d, f-f, s-d, p-f, while the allowed transitions are s-p, p-d, d-f.
Laporte Selection Rules
At what energy of transition is both spin and Laporte allowed?
E < 50,000
What increases Δo?
Increasing oxidation state and going down in a group
Pi acceptor ligands
Define this spin:
When there are more electrons unpaired than in a free ion
High Spin
Define this spin:
When there are more electrons paired than otherwise
Low Spin
What orbital configuration has this?
Highest in energy is the x2-y2, followed by xy, then yz and zx at an equal level, and then z2 lying lowest in energy.
Favored for d8 complexes
Square Planar
What d-n will not experience the JT effect?
d3, d5, d8, d10
Define this:
Compressed along the z-axis
Causing the z-component orbital to raise due to greater overlap
JT compression
Define this:
Pulls the z-orbitals down
Causing greater overlap in dxy and dx^2-y^2 orbitals
JT Elongation
What effect does JT distortion have on spectra?
Absorbance peaks will asymmetric
Define this:
Specific pathway for electron transfer between molecules involving metal complexes
Requires a bridging ligand
Inner Sphere Mechanism
Define this:
Specific pathway for electron transfer between molecules where the electron transfer occurs without direct bonding or significant interactions
Outer Sphere Mechanism
Define this crystal system:
All equivalent (a = b = c)
Highest symmetry
Cubic
Define this crystal system:
All angles equal 90
a = b ≠ c
Tetragonal
Define this crystal system:
All angles equal 90
a ≠ b ≠ c
Orthorhombic
Define this crystal system:
Distorted more
Lowest symmetry
Nothing is equal
Triclinic
What is the relationship between bond length and charge?
A negative charge indicates a shorter bond length than neutral
What is the formula for magnetic moment?
sqr(n(n+2) )