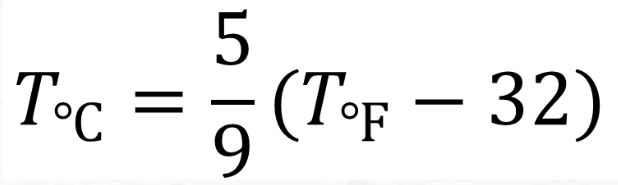Gases KMT: Boyle's Law and Charle's Law
1/18
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
KMT?
Kinetic Molecular Theory
Properties of Gas:
? or more gases form a homogenous mixture
Two or more gases form a homogenous mixture
No definite ? or ?
Gas expands ? to fill its container
No definite volume or shape
Gas expands spontaneously to fill its container
? compressible
Gas molecules can be pushed closer together because the gas particles are far apart.
Highly compressible
Gas molecules can be pushed closer together because the gas particles are far apart.
Exerts ? on the walls of the container
Exerts pressure on the walls of the container
Explain the states of matter
Describes the behavior of an ideal gas
The foundation for the gas laws
Kinetic-Molecular Theory
What shape particles are the gases?
spherical particles that are far apart
They move in ? rapid motion in ? directions
in constant rapid motion in random directions.
? forces of attraction or repulsion between gas particles
NO forces of attraction or repulsion between gas particles
Collisions between molecules and container are perfectly ? collisions
Collisions between molecules and container are perfectly elastic collisions
The average kinetic energy of gas particles is ? upon the temperature
The average kinetic energy of gas particles is dependent upon the temperature
At a constant temperature, the change in pressure is ? proportional to the change in volume.
At a constant temperature, the change in pressure is inversely proportional to the change in volume.
Formula for Boyle’s Law
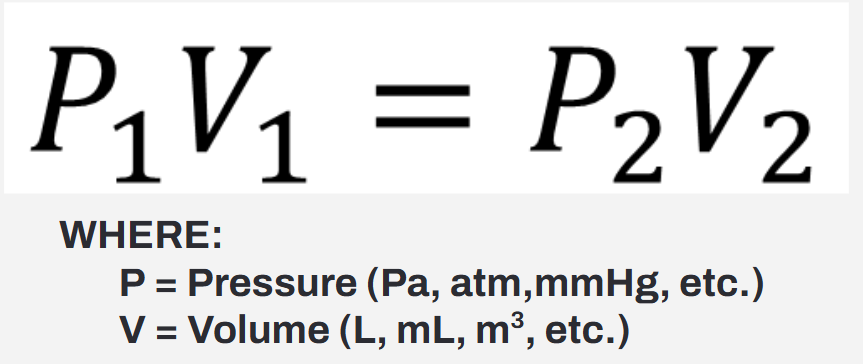
Standard Pressure (STP) = ?
Standard Pressure (STP) = 1.0 atm
At a constant pressure, the change in volume is ? proportional to the change in absolute temperature.
At a constant pressure, the change in volume is directly proportional to the change in absolute temperature.
What is the Formula for Charles’s Law

Standard Temperature = ?
Standard Temperature = 273.15 K
Formula for Celsius to Kelvin
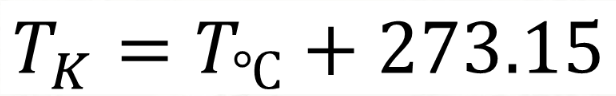
Formula for Fahrenheit to Celsius