Instrumental Analysis Unit 3
1/53
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
54 Terms
Mas spec interferences
Isobaric: When species with similar mass/charge ratio overlap. Fixed by using a diff isotope
Polyatomic: such as formation of O2+, can also cause overlap. Oxides/hydroxide formation
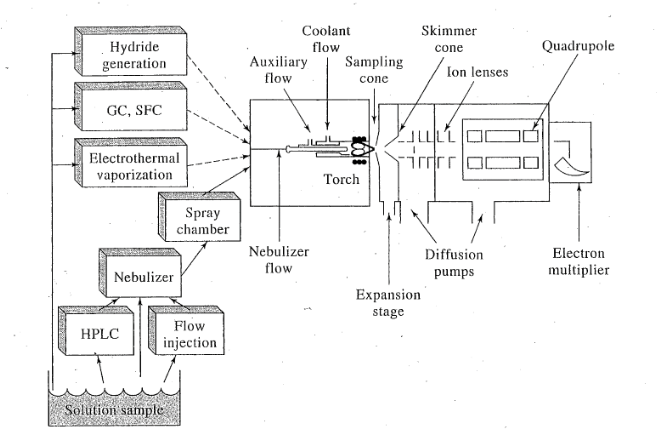
ICPMS
Inductivly coupled plasma mass spectroscopy
Dynamic range of 6 orders of magnitude
can detect .1-10 ppb
good for analyzing solids
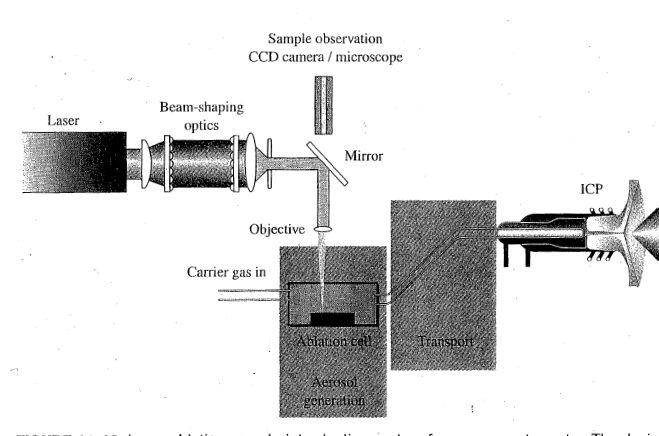
Laser Ablation ICPMS
A way to introduce a sample to ICPMS
How can ICPMS be used?
Qualitative/semiqualitative applications
Qualitative anaylsis
Isotope ratio measurments
X-ray wavelength
.1-25 A for tranditional x-ray spec
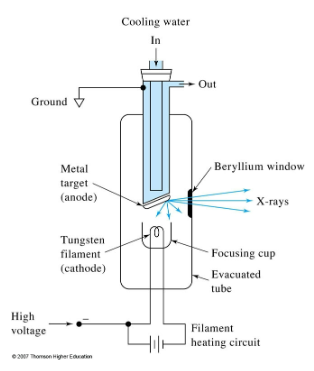
How are X-rays generated for spec?
Bambardemnt of a metal target with high energy electrons
Exposure to a X-ray beam to generate X-ray flourescense
A continuum spectra from radioactive subtances
X-ray tube
Continuum signal from a tungsten anode. electrons are shed from anode after stimulation with a high voltage. Produces white radiation.
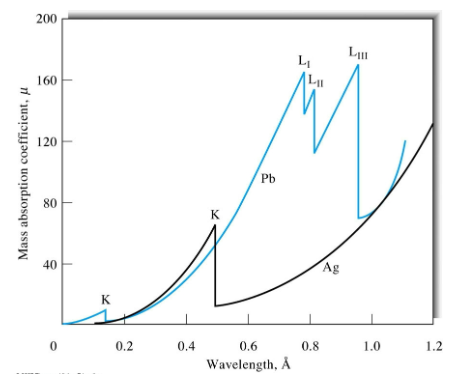
X-ray spectra characteristics
well defined, short wavelength limit. The minimum excitation voltage increases with atomic number.
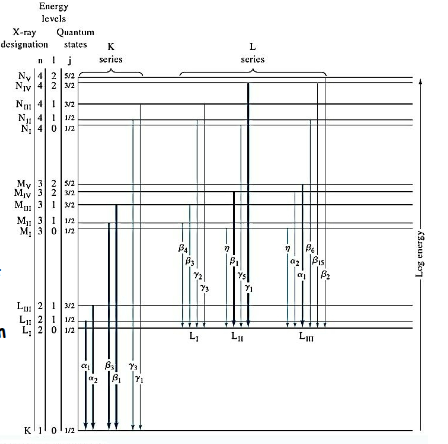
K series
X ray spec, emissions where an electron falls to innermost K shell (n=1). Higher energy
Ka: n=2 →1
KB: n=3 → 1
L series
x ray spec- when an electron falls to n=2. more complex than k series (with subsets of alphas and beta transitions), with lower energy than k.
K vs L energy diagrams
what produces monochromatic x-rays
alpha and beta decay, electron capture (when an electron is absorbed into nucleus to turn a proton into a neutron).
x-ray Absorption spectra example
X flourescense instrument sources
x-ray tube
radioisotope
secondary fluorescence
Needs a filter/monochromoter
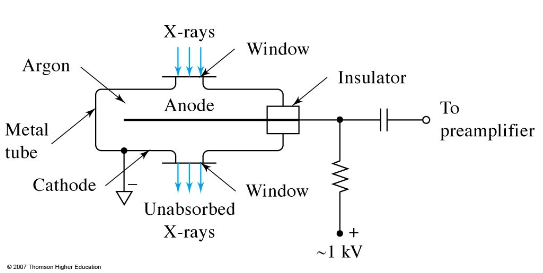
gas filled x ray detector
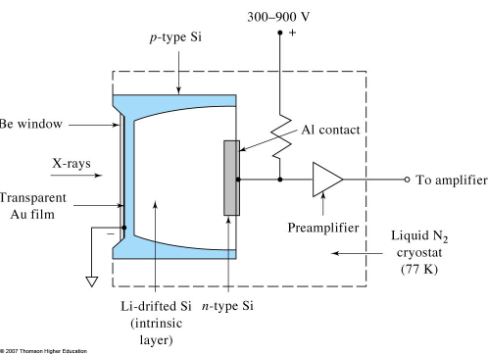
Si(Li) detector
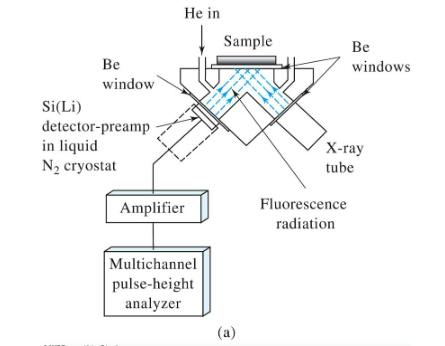
What is X-ray florescence used for?
rapid, quantitative determinations
works on Na-U elements
quantitative analysis
qualitative analysis
Energy-dispersive X-ray fluorescence spectrometer
Radiation losses in UV-Vis cell
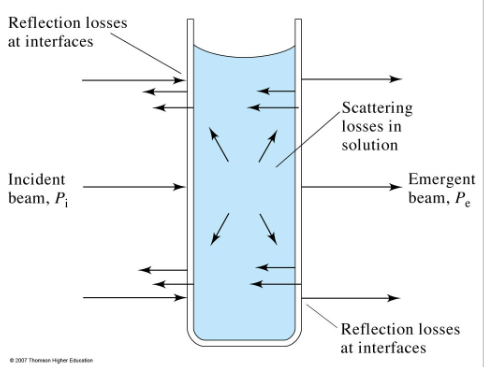
Uv-Vis limitations
not linear above 1 absorbance
High concentration can cause scattering and index of refraction changes (which can effect molar extinction)
reactions in the cell, such as a equilibria
needs monochromatic radiation (a wavelength selectors to .1 peak width of Absorption band)

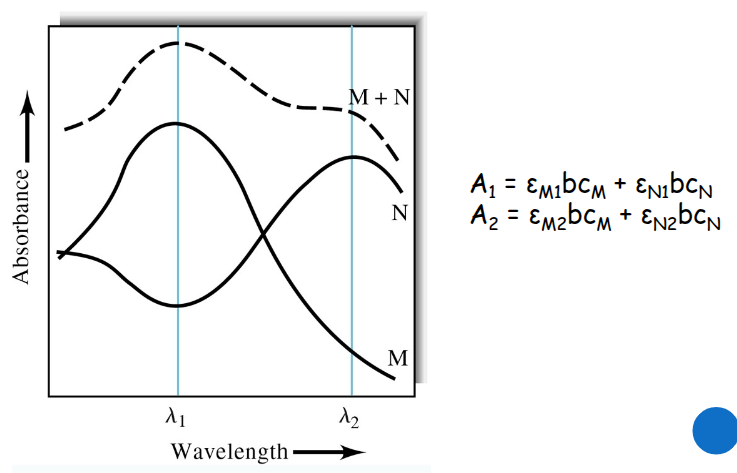
Beer’s law for mixtures
sum the absorbance values for Atot, where 1 refers to wavelength 1
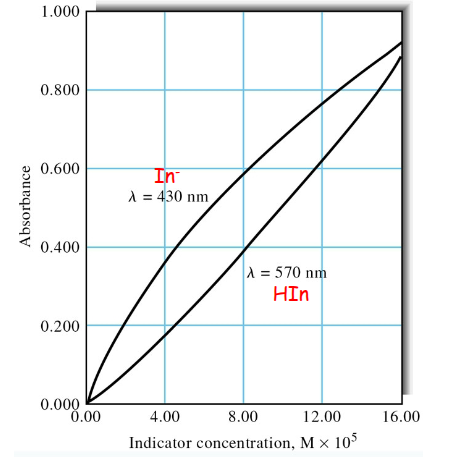
Absorbance deviations for a weak acid
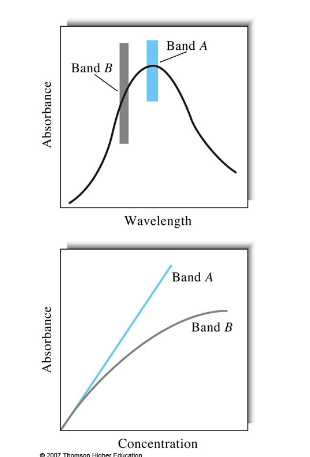
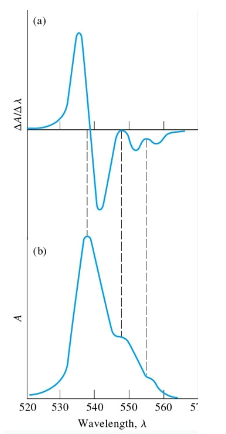
Why is lambda max important
absorbance values taken anywhere else will not be linear and can have varying molar extinction coefficients. The sharper lambda max is, the more sensitive the instrument
Stray radiation correction equation
for radiation that doesn’t pass through the cell
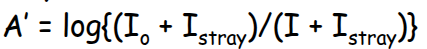
How does slit width effect absorbance?
Increases absorbance and peak resolution- however can increase noise by reducing signal.
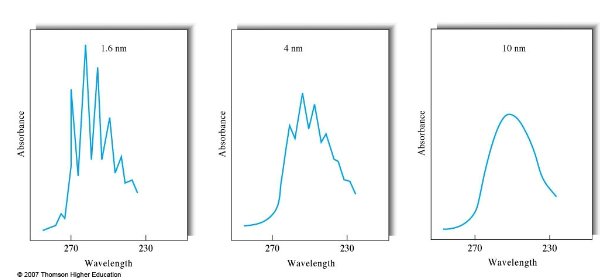
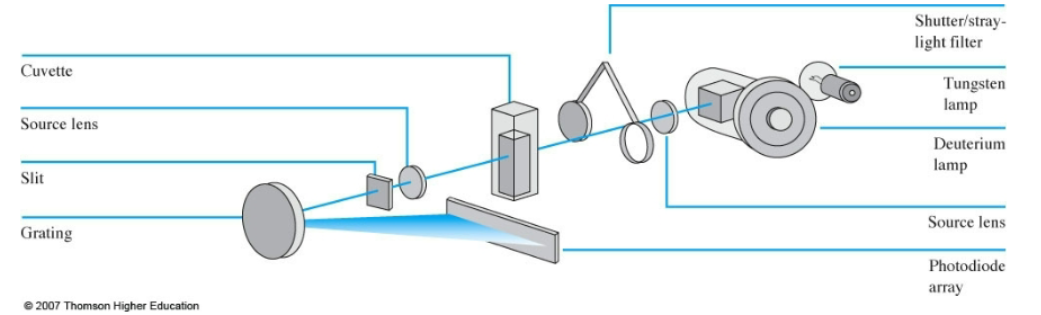
UV-Vis components
sources
sample continers
Instrument diagrams

single beam UV-Vis design
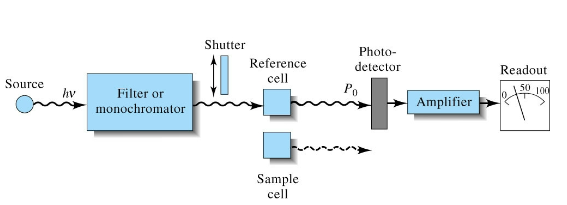
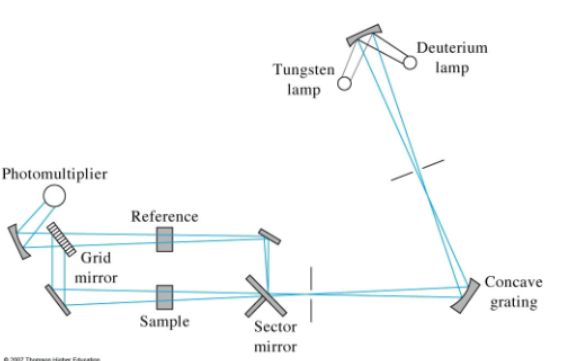
Double Beam UV-Vis design
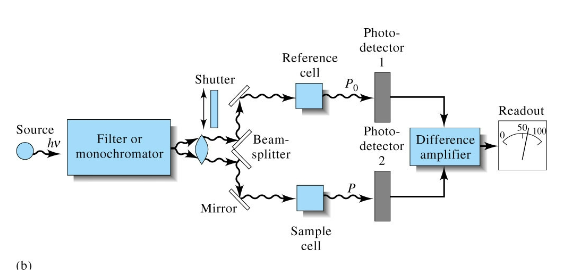
Double beam in time UV-Vis
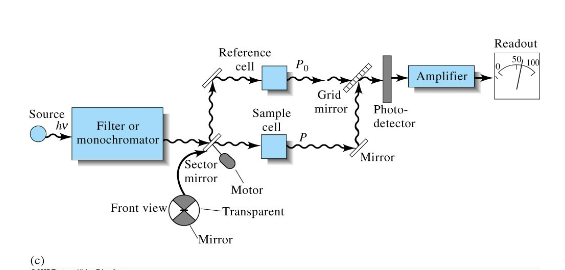
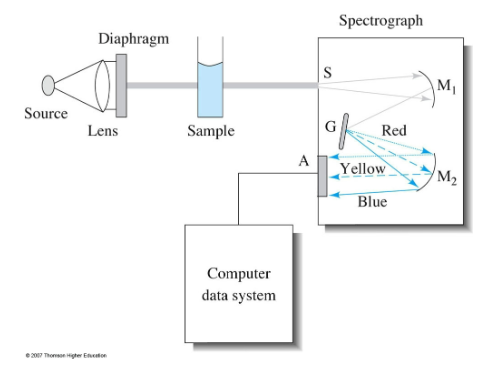
Multichannel UV-Vis design
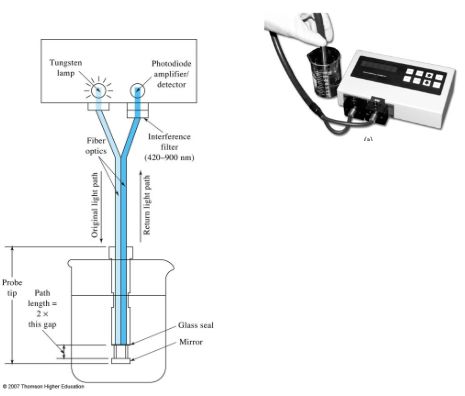
Probe type photometer
Manual Double-Beam Spectrometer
Aligent Diode Array UV-Vis
What can UV-Vis be used for?
determination of chromophore functional groups since lambda max is distinct (samples must be dilute for qualitative analysis.)
Inorganic and organic compounds can be quantitatively analyized for concentration, sensitive to 1E-5 M with good accuracy
Used in: Clinical tests and water analysis
what affects the molar extinction
Probability of a energy transition (between 0 and 1)
cross sectional area of the molecule
How do organic molecules enter the excited state?
through molecular orbitals
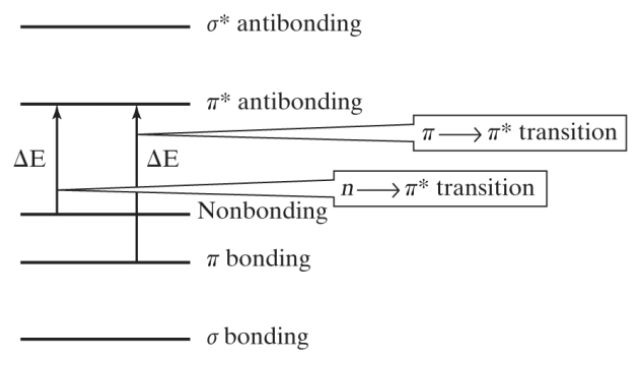
types of molecular transitions
hysochromatic shift: to a shorter wavelength (red shift)
bathochromatic shift: to a lower wavelength (blue shift)

chromophore
a portion of a molecule capable of absorbing light in the UV-Vis range
Formula for molar extinction coefficient
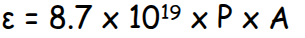
Auxochromes
functional groups that does not absorb themselves but they affect the chromophores by shifting their peak to a longer wavelength as well as increasing the intensity.
inorganic anions absorbtion
since ions can have a different Absorption to a protonated molecule, must select a buffer carefully
Lanthenides and actinides absorbtion
f electrons have sharper bands because of the inner electrons
uneffected out species on the outer complex
charge transfer complex absorbtion
an electron donor group bonded to an electron acceptor absorbed radiation, where an electrons from the donor will move to the orbital of a acceptor.
rare earth absorbtion
Similar to f electrons. The electrons responsible for absorption by these elements are shielded from external influence by electrons that occupy orbital with larger principal quantum numbers - Spectra are narrow and unaffected by species bonded to outer electrons.
how does slit width effect absorbtion
smaller slit means more absorbance
Why would the deriative of a UV-Vis plot be useful?
lamda max will be when the plot is 0, makes it easier to visualize.
Photometric titrations graph examples
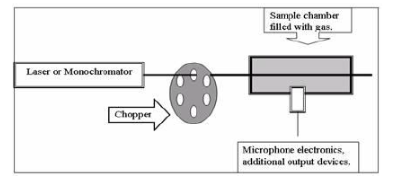
photoaccoustic spectroscopy
radiation is chopped through a gaseous, nonreactice substance to the sample, as the sample undergos excitation heat is generated which changes the pressure. the sound produced by that is tracked.
can be used on solids
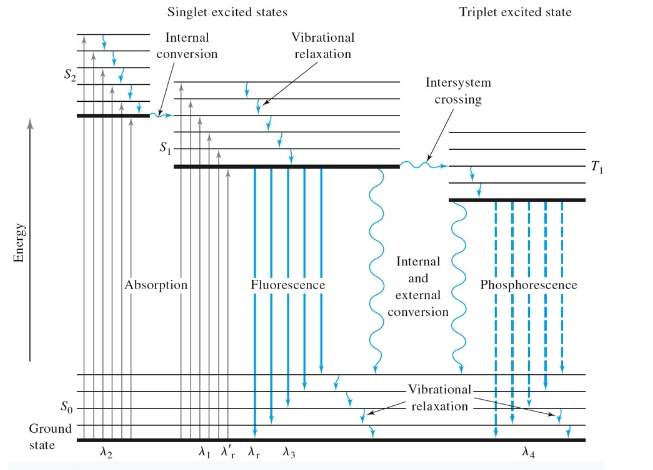
Jablonski diagram- look up a YouTube vid or smth, this is dumb af
vibrational relaxation: non radiative, gives off heat
intersystem crossing: movement from lowest vibrational level of 1 state to a higher level of another state. causes a spin change. phosphorescense
Quantum yield
A measure of how much floresence is produced.

How to triple bonds effect flouresence
enhance, because a molecule with a lot of pi character will floures better.
How do electron donating grouped effect flouresence
will stabilize the molecule and enhance fluorescence, electron withdrawing groups will hinder fluorescence (Halogens, nitros, and carboxylic acids)
What are the best transitions for florescence
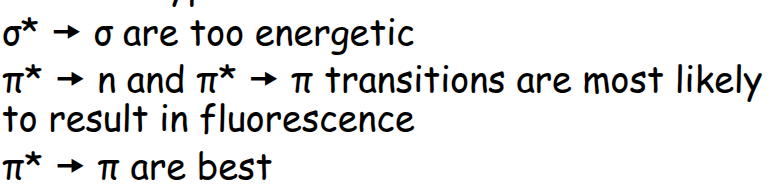
How do complexes effect flouresense
will increase when organic molecules form a complex with metals