Food Class test
1/245
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
246 Terms
what is a starch?
an energy source which is used across a range of moisture contents
starch benefits/properties in food processing
provides interesting textures at a range of moisture contents
can be stored dry
is a starting material for other chemicals
can be modified to give an even greater range of properties
common storage carb in plants
where can starch be found naturally
seeds
roots
tubers
stems
native starch is always…
amylopectin (waxy starch)
amylose
high amylose starch benefits
structure functions health benefits - helices can encapsulate small enough molecules for targeted delivery and can form starch-lipid complexes which can impact quality in baking
starch macromolecules organization
semi crystalline
amorphous amylose region
crystalline amylopectin region
importance of water for starch
starch is biosynthesized in an aqueous enviro.
drying starch can cause shrinkage and cracks
most water goes to amorphous phase or to surface of crystallites
water absorption is reversible
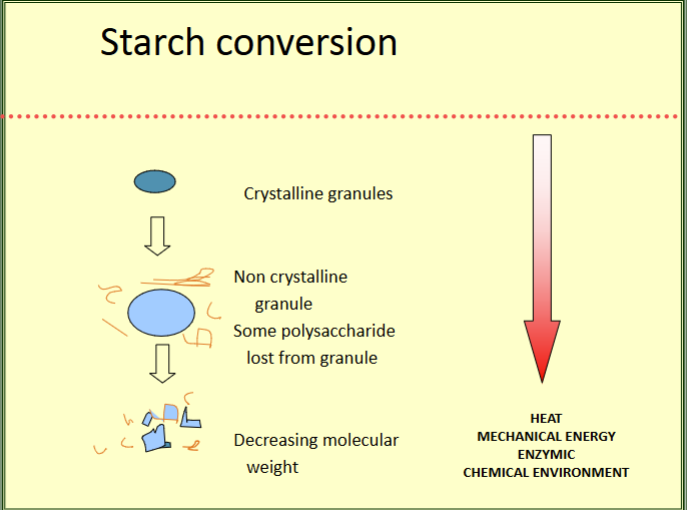
starch gelatinisation
disruption of molecular orderliness within the starch granule due to water absorption
water in - granule swells - loss of some polymer
starch pasting
continuing starch transformations at temperatures exceeding gelatinisation temperature
conditions required for starch gelatinisation and pasting
native starch granules in excess water
temperatures above cooperative disassociation of the crystallites
heating starches in limited water
loss of helical order and crystalline state
starch conversion
wheat flour components
protein (gluten) - forms support for final structure
carbohydrate (starch) - sets the loaf in the oven
damaged starch (water holding)
enzymes (water releasing)
lipids (fats)
cell wall material
different flours and their purposes
stronger - premium white products
higher extraction where colour is less important
weaker for standard and value white and brown bread
wholemeal - includes endosperm, bran and germ
gluten purposes
supplements a weak flour - improves proof height
supports recipes that contain ingredients that damage the gluten or bubble structure e.g. vinegar, fruit, milk
enables excess expansion
for long fermentation processes to support dough slackened by amylases and yeast
requires extra water and work input
water purpose in bread making
essential for dough formation
‘optimum’ level is essential for good dough development, dividing, texture and colour, slice strength
yeast role in bread
eat sugar and produce gas creating bubbles
killed by too much salt, sugar, vinegar, preservatives, flour and heat
need to be stored cold because they become active in warm storage
salt role in bread
enhances flavour
slows down yeast activity
strengthens gluten structure by improving proof stability and giving a finer crumb texture
increases oven spring by increasing setting temperature and delaying gas release
slows mould growth and staling
gives brown crust colour
sugar role in bread
feeds yeast so saves fermentation time
increases dough setting temperature - can lead to underbaking
often not included commercially as yeast and amylase create their own
what are polysaccharides
natural polymers formed of repeating subunits (10+)
generic uses of polysaccharides
thickener
mimic full fat food textures
create food gels
emulsifier
control ice crystallisation
lipids uses
flavour
heat transfer
sensory qualities
emulsification
staling inhibition
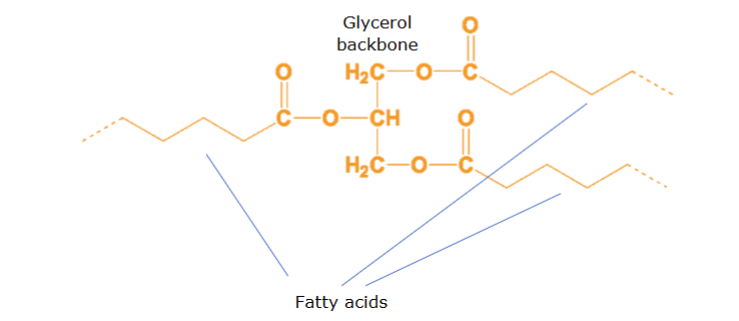
transglycerols (TAGs)
esters derived from glycerol and 3 fatty acids making up over 90% of dietary fats
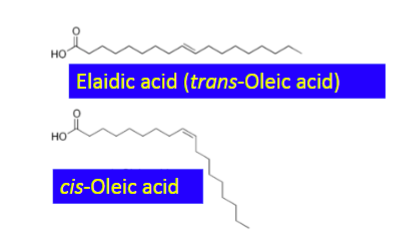
unsaturated fatty acids
straight or kinked structure
structure affects packing thus melting the T of TAGs
factors affecting solid fat content (SFC)
crystal morphology
type of vegetable oil/fat
temperature
factors affected by solid fat content
rheology
melting range
heat capacity
density
how to modulate solid fat content
hydrogenation
chemical interesterification
fractionation
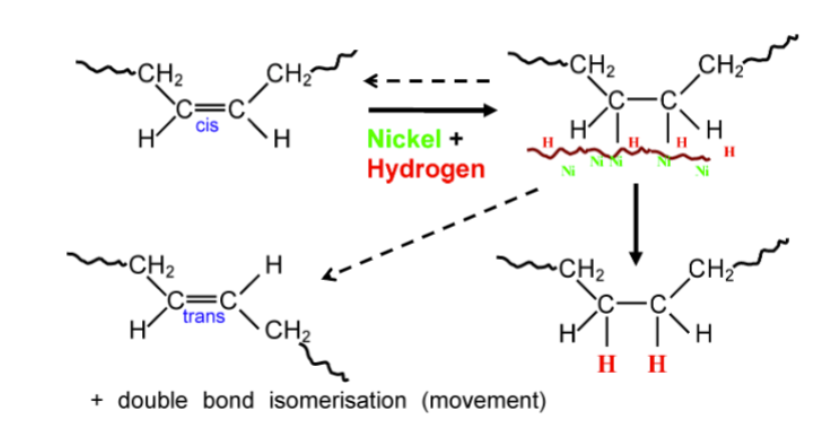
hydrogenation to modulate solid fat content
improves oxidative stability by eliminating unsaturated fatty acids
increases solids to make more functional and useful (i.e. hardening)
easily controlled to give a wide range of products from liquid to 100% solid
associated with soybean and rapeseed oils
produces trans-fatty acids therefore not a preferred apporach (bad for heart health)
interesterification to modulate solid fat content
rearranges fatty acids on the glycerol moiety to create new TAGs
traditional chemical process produces a random distribution of fatty acids
enzymic process can be more controlled
associated with palm oil, palm kernel oil and other solid fats
does not affect fatty acid composition so cannot produce trans fatty acids
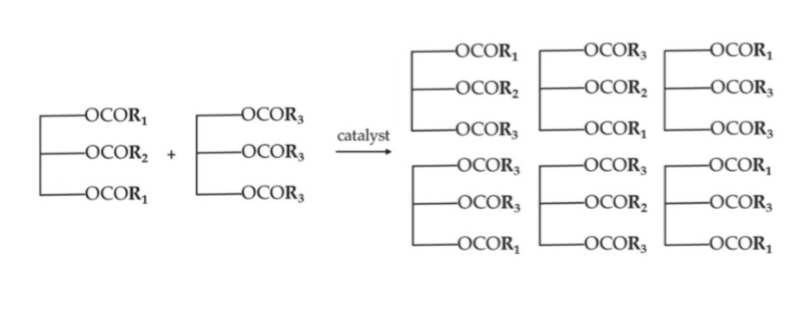
fractionation to modulate solid fat content
fractional crystallisation
melt oil completely
cool under controlled conditions until crystal nuclei form
allow crystals to grow and agglomerate
separate crystals from liquid
long process - up to 24 hours
environmentally friendly
does not change fatty acids from natural state
no health issues known
relatively cheap
assoicated with palm oil, palm kernel oil, milk fats
proteins
natural polymers made up of 20 different amino acids linked by peptide bonds
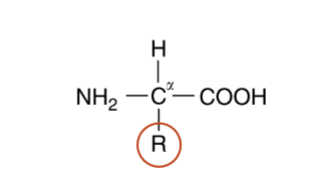
amino acid structure
α-carbon atom linked to a hydrogen atom, an amino group, a carboxyl group, and a side chain
high quality protein meaning
proteins containing all essential amino acids at levels above FAO/WHO/UNU reference levels
primary protein structure
amino acids linearly linked by peptide bonds to form a chain
secondary protein structure
peptide chain twists arise from near-neighbour or short-range noncovalent interactions between amino acid side chains
main structures: α-helix, β-sheet
tertiary protein structure
folding pattern of peptide chains into a 3D form due to intrachain interactions
relocation of most of the hydrophobic amino acid residues into the interior of the protein structure away from the water environment
relocation of most of the hydrophilic amino acid residues, especially charged residues, to the protein surface
quaternary protein structure
protein containing more than one polypeptide chain
complete protein is an oligomer
stability of the protein structure
native protein structure is stabilised by
intramolecular interactions emanating from forces intrinsic to protein molecule e.g. van der waals
intramolecular interactions affected by surrounding solvent e.g. hydrogen bonding
intramolecular disulfide bonds
intermolecular repulsive interactions between protein molecules in the same solvent
protein denaturation
changes in environment such as pH, ionic strength, temperature and solvent composition affect forces that stabilise protein structure.
level of denaturation affects protein structure
types of protein
globular
fibrous
flexible
globular proteins
polypetide chains folded into tightly packed structures
emulsifiers or foaming agents - non-polar patches at protein surface - adsoprtion to oil-water or air-water interfaces
gelling agents - unfolding during heat exposes hydrophobic and sulfhydryl groups promoting bonds formation between proteins and trapping solvent
fibrous proteins
rigid molecules
contribute to textural attributes of foods
mainly of animal origin
plant e.g.: glutenin
flexible proteins
fairly disordered structures: random coils
mainly of animal origin
important role in emulsification
solubility in proteins
insoluble proteins have v limited uses
functional properties of proteins are often affected by their solubility
solubility of a protein is the thermodynamic manifestation of the equilibrium between protein-protein and protein-solvent interactions
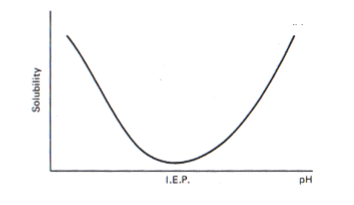
solubility vs pH
amino acids have different ionisation states depending on p\h of the solution due to their acidic carboxyl group and basic amino group
solubility curve - electrostatic repulsion and hydration of charged residues promote solubilisation of the protein
minimum solubility occurs at about the isoelectric pH of proteins; lack of electrostatic repulsion promotes aggregation and precipitation
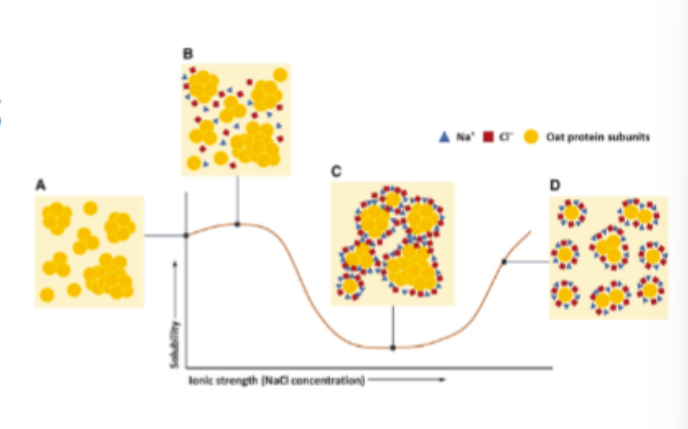
solubility vs ionic strength at pH values well above or below the IEP
very low salt
proteins repel each other due to strong neg. or pos. charge of proteins
low ionic strength
ions neutralise charges at the surface of proteins and solubility decreases due to aggregation
high salt
anionic counter ions preferentially attach to cationic patches on the protein surfaces which increase neg. charge on the proteins and thus electrostatic repulsion and solubility increases
solubility vs temperature
at constant pH and ionic strength, solubility increases with temp. between 0 and 40C
above 40C, protein denaturation takes place, causing a solubility decrease
protein emulsification process
proteins form protective layer at oil-water interface, lowering interfacial tension and providing a barrier to prevent accumulation of oil droplets
protein molecules migrate to oil-water interface and adsorb on the oil droplets, forming a viscoelastic film that prevents destabilization
adsorbed protein layer lowers interfacial tension and free energy, providing electrostatic repulsion and steric barrier to prevent oil droplet accumulation
this is essential for food products such as milk, salad dressings, ice cream, butter and cakes
foaming proteins
foaming agents such as egg whites, gelatins, casein, soy and gluten influence stability and texture of food products.
foaming properties of proteins are influenced by their ability to absorb at interface and reduce surface tension
protein gelation methods
addition of salt or acid
enzymatic reactions
fermentation
shear
high pressure
temperature change
heat induced gelation
native protein structure denatures under heating
protein aggregates
network may undergo restructuring and reinforcement upon cooling
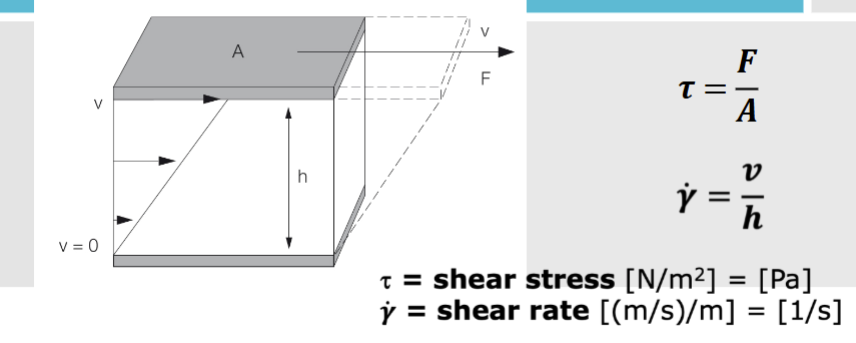
shear stress and shear rate
types of liquid foods rates
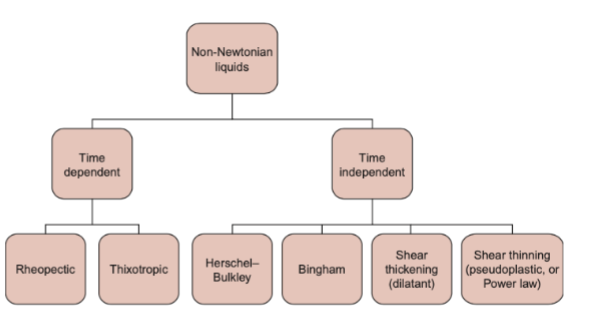
newtonian
shear-thickening
shear-thinning
viscosity
constant of proportionality between shear stress and shear stress and shear rate
temperature dependent

non-newtonian liquid foods
shear thinning
viscosity decreases with increasing shear rate
how does shear thinning occur in protein microstructures
polymer chains disentangle and stretch
emulsion droplets reorganise and deform
elongated particles align with flow
aggregated structures break down to primary particles
shear thickening
viscosity increases with increasing shear rat
yield behaviour role in foods
affects texture and ‘thickness’, important when considering flow of coatings, balance of emulsions, texture of sauces
yield stress
minimum shear stress that needs to be overcome before onset of shear flow
most simple yield stress behaviour is bingham plastic liquid
viscoelastic behaviour
tack and stringiness
stirring process - rod climbing effect - poor mixing result
extrusion - extrudate swelling
shear modulus
measure of a materials ability to resist deformation when subjected to shear stress
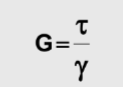
viscoelastic moduli

oscillatory shear testing
characterizes viscoelastic properties of fluids and soft materials
applies sinusoidal deformation to a material and measuring its mechanical response over time
small amplitude oscillatory shear tests
probes linear viscoelastic properties of materials
applied strain is small enough that response remains linear, allowing for determination of storage (elastic portion) and loss (viscous portion) moduli
commonly used for characterizing polymers, emulsions and other complex fluids
large amplitude oscillatory shear tests
investigates nonlinear viscoelastic behaviour of materials
as strain amplitude increases, material response becomes nonlinear, which is essential for understanding how materials behave in real-world applications where they may expereince large deformations
useful in quality control for rubber and other materials, can reveal interactions between fillers and polymers
temperature dependence of shear
follows arrhenius type relationship
types of emulsions
oil in water - e.g. mayo
water in oil - e.g. butter
water in oil in water - reduced fat and can deliver water soluble actives
emulsifier structure
prevents coallescence
amiphilic

emulsion microstructure size
10-9 —> 10-6 m colloidal systems
emulsion instability
most stable state is when oil and water are in separate layers so droplets in emulsion can coallesce to form larger droplets
why add surfactants to emulsions?
reduces interfacial tension which facilitates droplet break-up in top-down processes
rapid in process adsorption at interface stabilises small droplets which in turn benefits gravitational stabilisation
measuring surface/interfacial tension
force tensiometry - Du Nouy ring and Wilhelmy plate methods
optical methods - pendant drop technique
capillary rise method
maximum bubble pressure method
relationship between surface tension vs surfactant concentration
as surfactant conc. increases, surface tension decreases
de nuoy ring method
pull ring through liquid surface to cause a non-equilibrium state that affects measured surface tension
preferred for high-viscosity liquids
wilhelmy plate method
stationary
measures force required to detach a plate from the liquid surface
preferred for measuring surface tension variations over time
pendant drop technique
drop of liquid suspended from needle and image of droplet is captured using camera
software analyzes image to determine shape of droplet, which is used to calculate surface tension using young-laplace equation
high precision, so useful for development of coatings and paints
common food surfactans
mono and diglycerides of fatty acids
lecithin
polysorbates
citric acid esters
these may also be applied to process and stabilise foams or control particle aggregation and flow bheaviour
surfactant addition in bread
can enhance volume, soften crumb structure, increase shelf-life
different surfactants can strengthen or soften dough
emulsifiers in ice cream
provide smoother texture and ensure ice-cream does not melt rapidly after serving
improve freeze-thaw stability
emulsifiers in margarine
provide required stability, texture and taste
ensure water droplets are finely dispersed in oil phase and prevent splattering
polymeric emulsifiers use
better foam stabilisers than small molecular surfactants as they give stronger films
food emulsifier selection
hydrophilic lipophilic balance
predicts behaviour of emulsifiers related to their solubility
HLB=1-20 scale from hydrophobic to hydrophilic
bancroft rule
hydrophobic emulsifiers stabilise water-in-oil emulsions
hydrophilic emulsifiers stabilise oil-in-water emulsions
particulate emulsifiers
solid particles such as starch, chitin, cellulose that stabilize emulsions
offer enhanced resistance to coalescence, high stability, good repeatability, simple scalability, enhanced biocompatibility
advantages of using food powders
ease of use, processing, transport
maintain quality and stability
states of food powders
crystalline
amorphous
crystalline powders
non hygroscopic, stable, easy flowing e.g. salts, sugars, organic acids
amorphous powders
obtained by rapid supercooling or rapid removal of solvent
particle morphology affects/effects
affected by raw materials and process conditions
impact
bulk density
flowability
ease of handling
mixing
compressibility
surface activity
particle processes for liquids
spray
freeze
drum
belt
crystallisation
particle processes for solids
size reduction
granulation
mixing
water activity role in stability
water activity quantifies amount of free/bound water in a compound, so signifies availability of water for microbial processes i.e. spoilage
microorganisms require water to grow, and each has a minimum water activity threshold below which it cannot multiply
no microbial growth possible below 0.6 water activity
drying processes
freeze
spray
roller and drum
fluidised bed
roller and drum drying process
liquid applied in thin layer to rotating steam heated drum
dried film scraped off after ¾ of a complete rotation
economical for small volumes
applied to milk, oats, whey, corn and instant mash processes
freeze drying process
dehydration by sublimation of a frozen product
primary drying - rapid sublimation occurs by controlling vacuum level in dryer and careful heat input - heat supplied by combination of conduction and radiation - ice leaves as porous dry cake
secondary drying - warmer temp. to remove bound water, slower drying rate, moisture loss occurs only by diffusion
slow freezing leads to formation of large ice crystals while rapid freezing promotes formation of small ice crystals
cake resistance affects product temperature and speed of primary drying
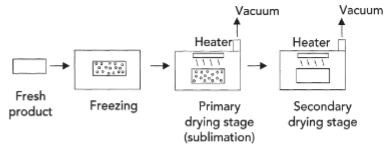
main components of freeze dryer
product chamber
refrigeration system
condenser
vacuum system
control system
pros and cons of freeze drying
can yield high quality products
high operational cost and long processing time restrict its usage just to high-value products
spray drying process
preparation of dry stable additives, instant food powders, functional ingredients and flavours
co-current, counter-current and mixed mode types
convective heat and mass transfer induced by movement of air
high pressure nozzles or centrifugal atomizers used
rapid drying due to small size of droplets
a range of particle morphologies can be obtained
co-current spray drying
food droplets at room temp, air at high temp
water removed from droplets provides an evaporative cooling effect
when product reaches its direst state, the air has also cooled
powder separation methods
cyclone - centrifugal force causes particles to segregate from air. air flows back out the top, while particles are removed from bottom
textile filter - powdered air passes through fabric filter before being exhausted into atmosphere. fine particles are trapped by filter