Thermodynamics, Free Energy, and Electrochemistry
1/32
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
33 Terms
Thermodynamically favorable Reaction
A reaction that occurs under specific conditions
always move a reaction mixture toward equilibrium.
Often known as Exergonic
Thermodynamically Unfavorable reaction
A reaction that does not occur under specific conditions
Often known as Endergonic
Entropy
Amount of molecular disorder in a system
Chaos
Methods of Changing:
Altering temperature
increased movement → increase reacting
Moles created/used
If products have more moles → more chaos
Increase Gas volume
more space → more methods of configuration → increase
Atom size
Larger atoms → more chaos (Usually)
Molecule complexity
More complex → more states of formation/reaction → increased chaos
Forming mixture
Pure substance to mixture → increased randomness → increased chaos
Second Law of Thermodynaics
Reactions proceed in the direction that increases the entropy of the Universe.
Meaning:
High Chaos/entropy → Favorable
Low Chaps/entropy → Unfavorable
Third Law of Thermodyanmics
Entropy of a perfectly ordered crystalline substance at 0 K is zero - Absolute Entropy!
Standard Molar Entropy (S°)
Absolute entropy of one mole of a pure substance at 1 atm & specified Temperature
General Rules:
Sgas>Sliquid>Ssolid
SLarge Molecule>Ssmall molecule
Standard Molar Entropy of Reaction (ΔS°rxn)
the difference in entropy of the products and the reactants

Gibbs Free Energy (ΔG)
Looks at enthalpy and entropy of the system to determine if overall reaction is favorable.
Represents energy left/ Excess energy available to make reaction favored
Meanings of Signs:
if ΔG is -, Forward reaction favored & reaction is favorable
if ΔG is +, reverse reaction favored & reaction is unfavorable
if ΔG is 0, both reactions equal & reaction is at equilibrium
ΔG if ΔS>0 and ΔH<0
ΔG is -
ΔG if ΔS<0 and ΔH<0
Temperature Dependent
if High enough T, ΔG is +
If Low T, ΔG is -
ΔG if ΔS >0 and ΔH>0
Temperature dependent
When T is high enough ΔG is -
When T is low ΔG is +
ΔG when ΔS<0 and ΔH>0
ΔG is +
Standard Gibbs Free Energy (ΔG°rxn)
ΔG of products -reactants
Free Energy for a reaction under standard conditions]
1 atm
1 M concentration
Specified temp (usually 25 Celcius)
Kinetic vs Thermodynamic control
Kinetic Control favors lower activation energy
Thermodynamic control favors most negative ΔG value
How to interpret ΔG° graphs
Where line comes from is starting point, have line straight from there to end
Like a ball on a curve
As the curve goes down, ball can easily roll so favorable.
Can’t go back up on its own so not-favorable
Direction doesn’t matter
Equilibrium is where ball would eventually stop, at the complete bottom
ΔG is distance from line of initial to the point on curve
On X axis is formation of reactants and products
Left side is more reactants, right side more products
How to find Normal ΔG (Not on Reference Sheet MUST MEMORIZE)

Thermodynamic Coupling
Method to overcome lack of favorability
Use external source of energy in reaction to overcome positive ΔG
Main methods:
Addition of energy
Couple non-favorable reaction with favorable
Hess’s Law
For ΔG it is plus minus and multiplication, not the K ones
Ensures overall reaction is favorable
Importance of Electrons
Are reasons for Bond formation
Transferred in ionic
Shared in Covalent
Atoms use Intramolecular forces
Redox Reactions
Often known as Oxidation- Reduction Reactions
Process in which one or more electrons are transferred between reaction partners.
Both Oxidation and Reduction MUST occur together
Oxidation Number
Number telling us whether atom is neutral or electron rich or lacking
Compare old and new number to determine if atom has gained or lost electrons
NOT ALWAYS EQUAL TO CHARGE ON ION
How to Assign Oxidation Numbers
Atom in elemental state → Oxidation number of 0
Atom in monatomic ion state → Oxidation number = to charge
Atom in polyatomic ion or molecular compound → same number if monoatomic ion
Sum of oxidation numbers in neutral compound =0
Sum of oxidation numbers in charged polyatomic = charge
When one atom loses an e- another must gain an e-
IMPORTANT MEMORIZES FOR OXIDATION NUMBERS
Hydrogen:
Is +1 when bound to a non-metal
Is -1 when bound to a metal
Oxygen:
Usually has oxidation number of -2
In peroxides (O-O bonds) Oxygen has -1
Halogen:
Usually oxidation number of -1
When with Oxygen, Cl, Br, and I, have positive oxidation numbers
LEO the Lion goes GER
LEO
Lose Electrons Oxidation
GER
Gain Electrons Reduction
Reduction Agent
a substance that loses electrons
is oxidized
Increases oxidation number
Oxidizing agent
Substance that gains electrons
Is reduced
Oxidation number decreases
Half Reactions
Method of breaking apart a redox reaction
Creates 1 oxidation reaction and 1 reduction reaction
Balancing Redox Reaction
Write balanced net ionic equation
Split it into ½ reactions
Add e- to each half reaction
Multiple by whole numbers to get equal e-s in both equations
Add together reactions
Electrical Potential
The strength of the movement of electrons (push and pull) within a redox reaction
Higher Potential → increased work capability
Oxidation reactions cause “push” of electrons
Reduction reactions cause → pull of electrons
E°
Overall, just Standard Potential
Both Standard Half-cell potential and cell potential
standard cell potential (E°cell)
Sum of standard half reaction potentials for oxidation & reduction half reactions
Do not have to multiply by coefficient of your equation, just what’s on the sheet
Must be positive for favorable reaction

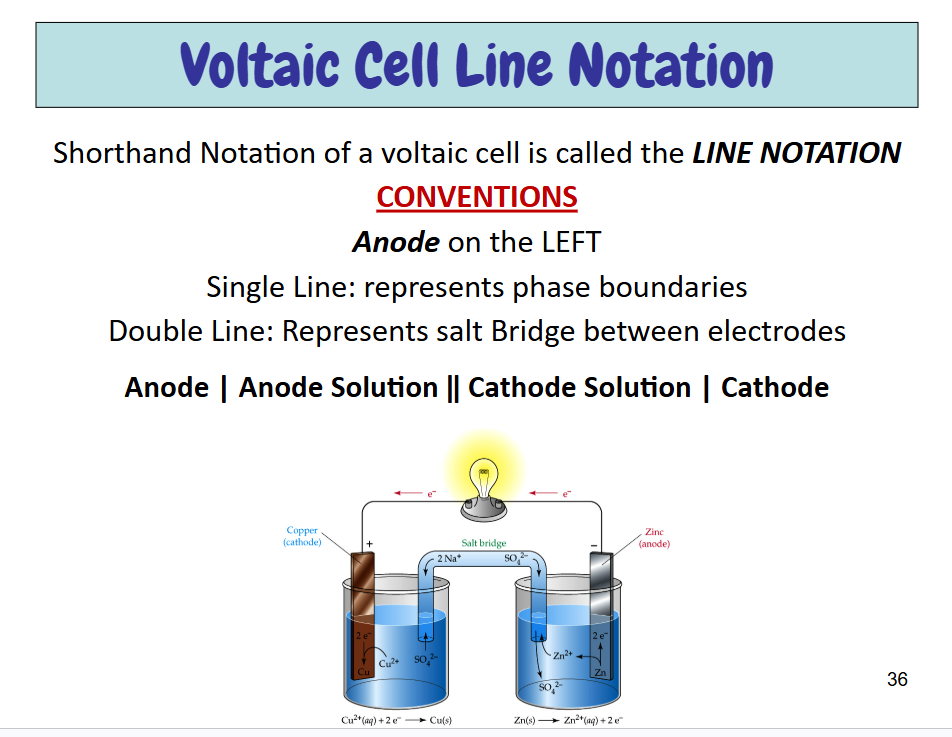
Voltaic Cells
Also known as Galvanic Cells
a favorable chemical reaction generates an electric current that can do work
Parts of Voltaic Cell
Electrode — metal strips/wires connected by an electrically conducting wire
Anode — Where oxidation occurs, anions migrate towards,-
Cathode — Where reduction occurs, cations migrate towards, +
Salt Bridge — Tube with a ionic salt. Ions don’t react with each other. Neutralizes growing charge, necessary for current, anions towards anode, cations towards cathode
Load — part of circuit which utilizes the flow of electrons to perform a function
Shorthand notation of Voltaic Cell