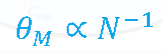Thermodynamics
1/50
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
51 Terms
Work done at a constant Pressure
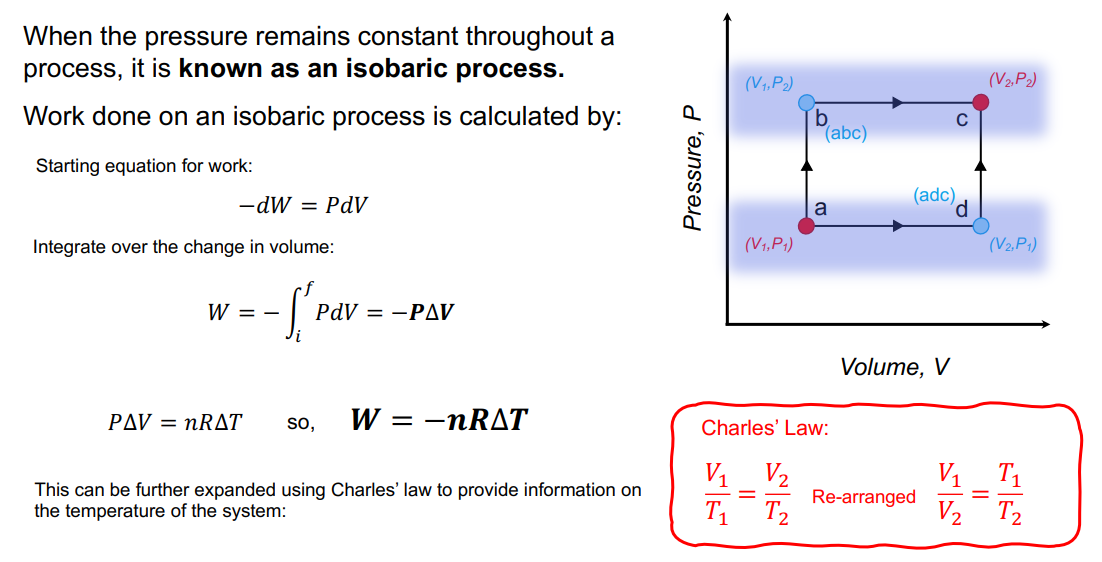
Work done at constant temperature
what is intensive and extensive properties
Intensive - Measurable property of systems which is indapendant of mass or size of systems
Extensive - Dependant on mass/size of system
Heat capacity relationship
Cp= Cv +R
Work done in an adiabatic process
W = cp(p2v2-p1v1)/R
Mass and energy balance for open system
Mass balance - dm/dt = min- mout
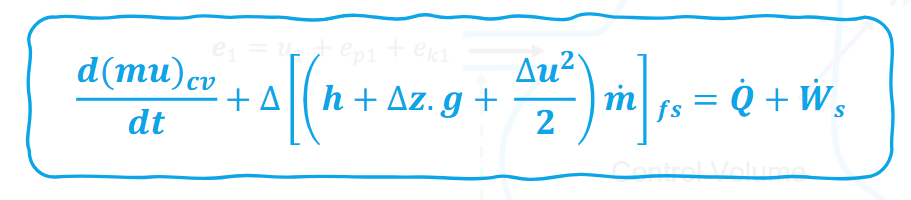
Entropy Change for ideal gas in isochoric process
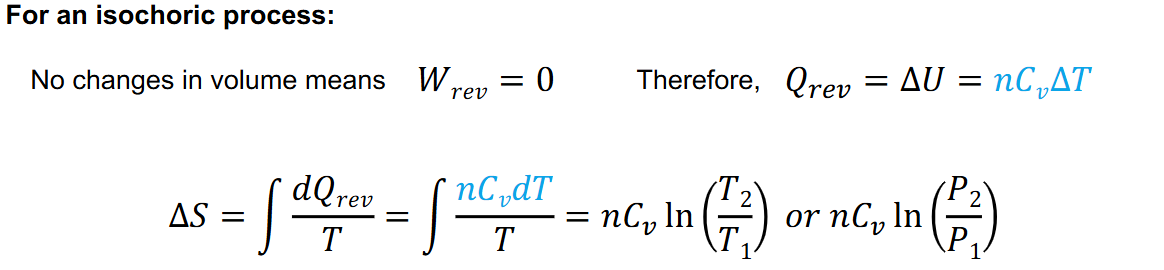
Entropy Change for ideal gas in isobaric process

Entropy Change for ideal gas in isothermal process
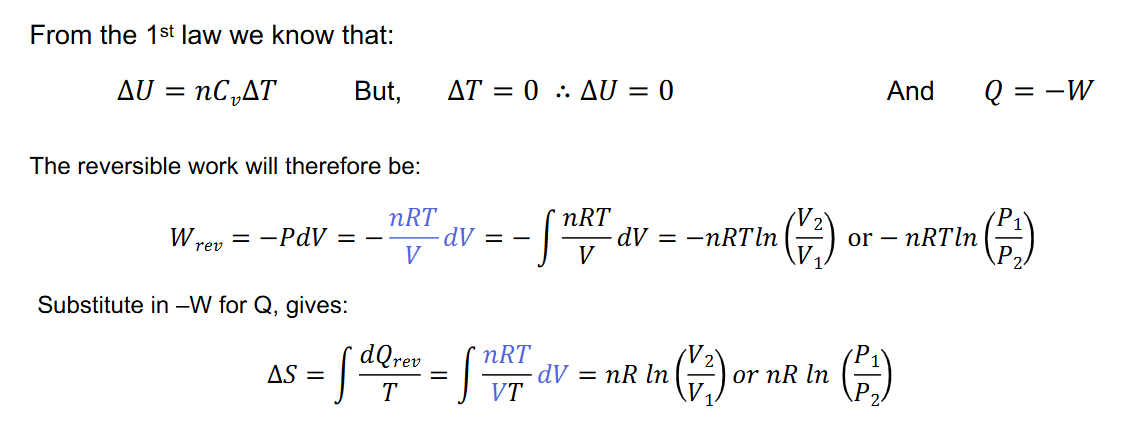
What are the two equations for the max efficiency
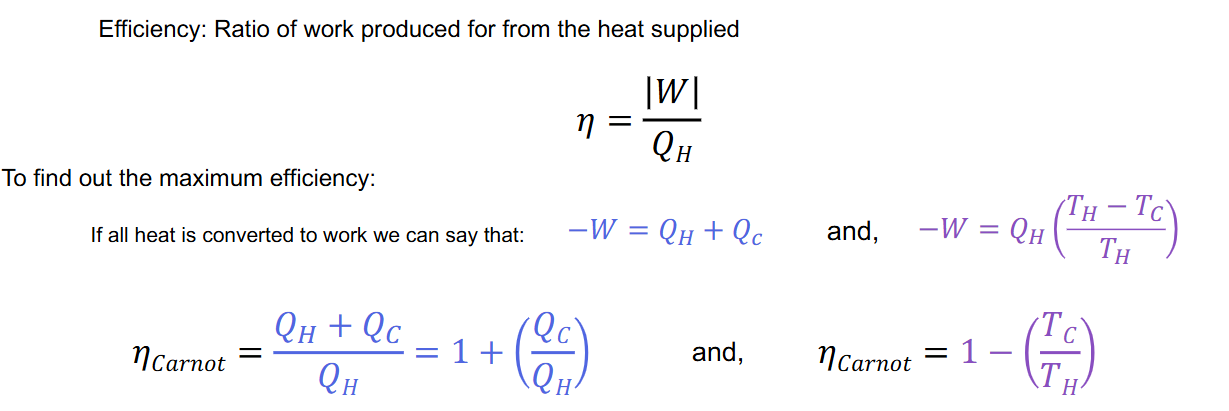
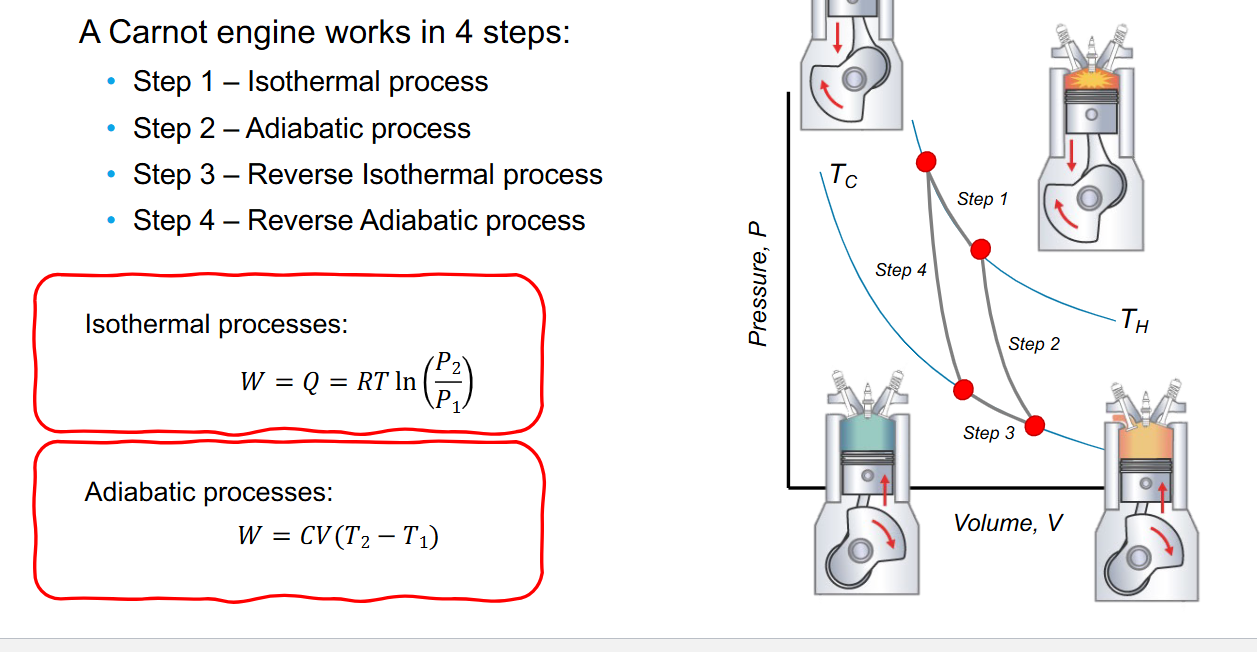
4 cycles of carnot cycle
Refrigeration efficiency and max efficiency
n = Qc/W
Nmax = Tc/(Th-Tc)
Entropy for open system
dst>0, entropy is not conserved
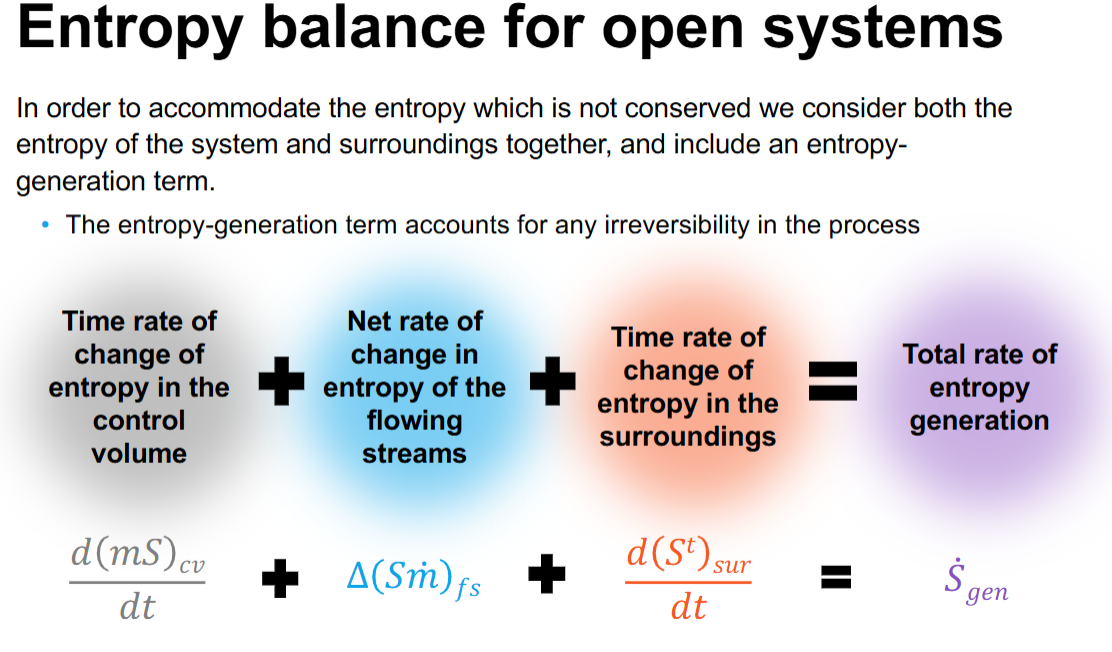
What do we need to consider for dU and dH for physical change
What are the equations for L-G transition and S-L transition for u and h
Need to overcome the bonds between the molecules and the need to expand against the atmosphere
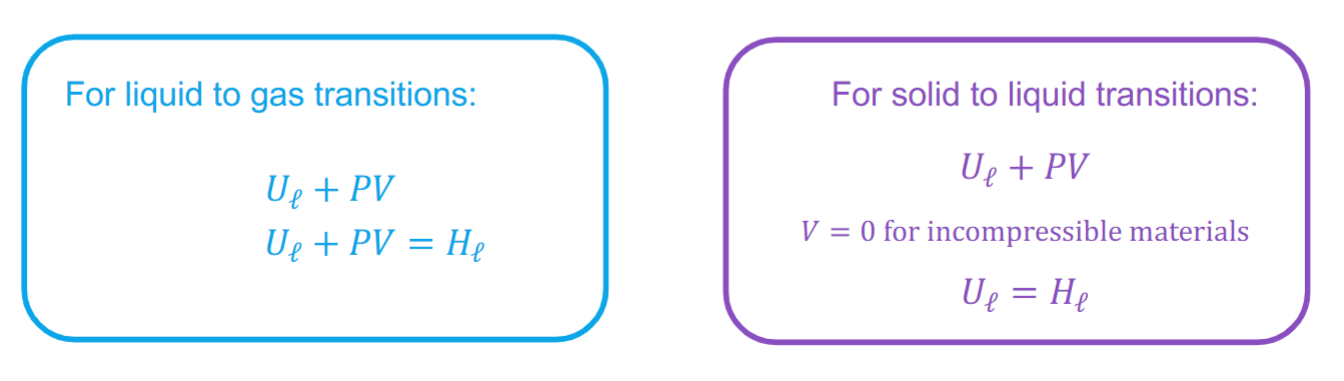
Thermodynamic diagram for P and V
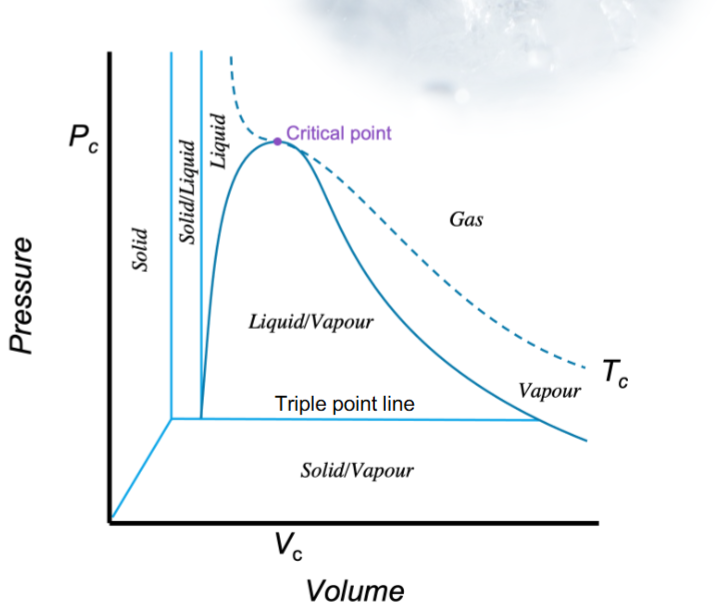
How to use relations cheat to derive fundamental property relation
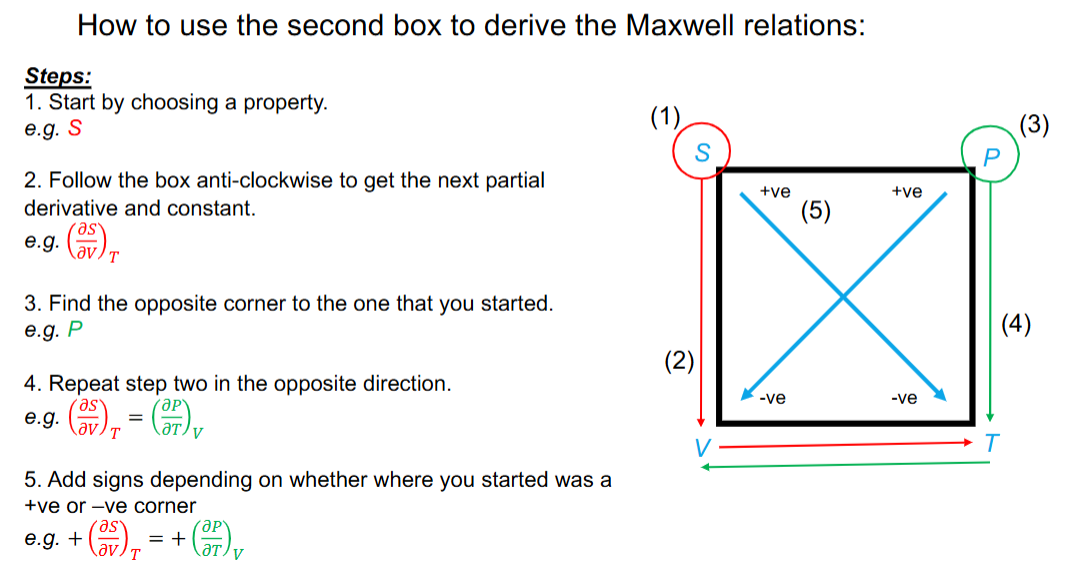
How to use the relations cheat to derive the maxwell relations
Derive the equation for the change in volume and enthalpy in a mixing process

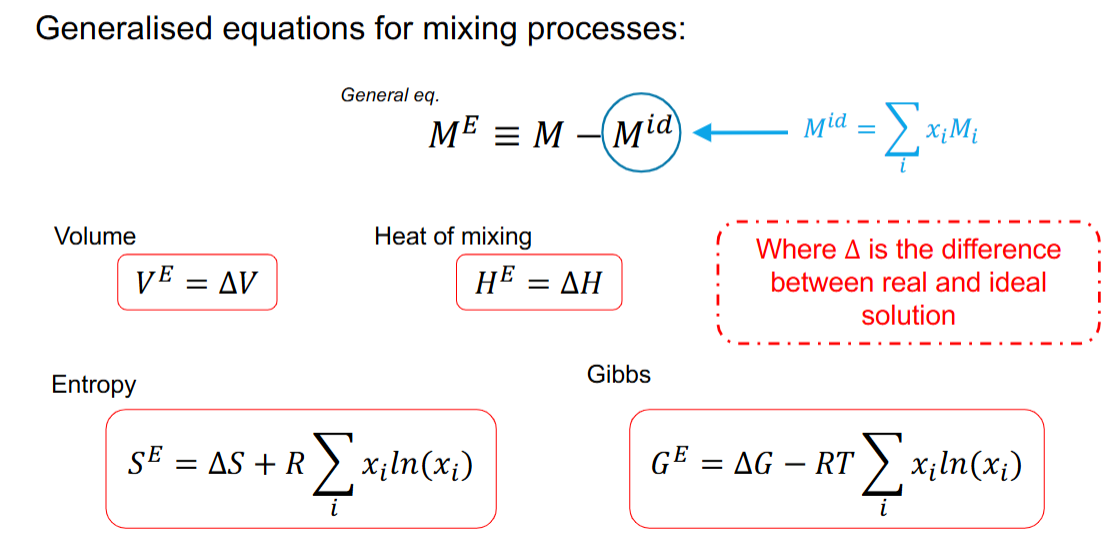
What are excess properties, and what is the equation for it

What are the excess properties equations for volume, enthalpy, entropy, and gibbs
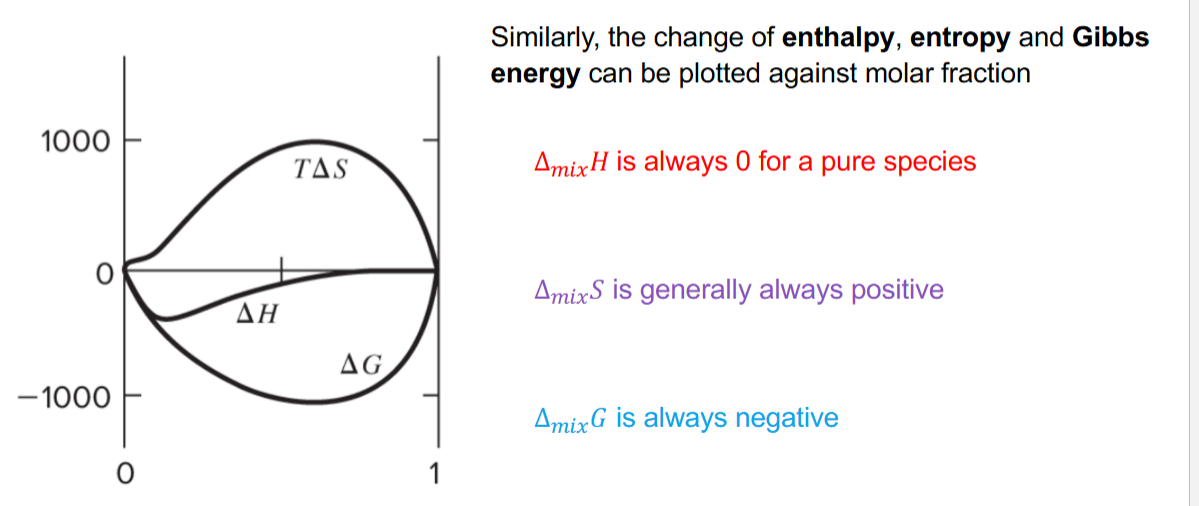
Show the excess entropy, enthalpy, and gibbs against mole fraction
How can enthalpy of a binary system be calculated

For a binary, adiabatic system, what is the straight line equation which connects all the original solutions

What is the equation for the heat of solution

Partial Properties for a single system
• 𝑀𝑖 – is the property for each individual specie
• 𝑀𝑖 – is the partial molar property of a component in mixture
• 𝑀 – represents of all the individual components together

Partial Properties for a binary system

Equation for extensive properties in a mixture

Derive the equation for internal energy for a binary system

Co-axial cylinder mixing equation
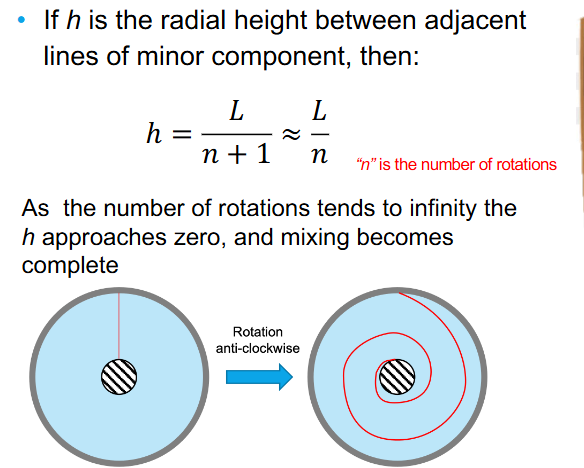
Laminar shear mixing for a co-axial cylinder mixing

laminar shear mixing for 1d laminar flow between parallel plate
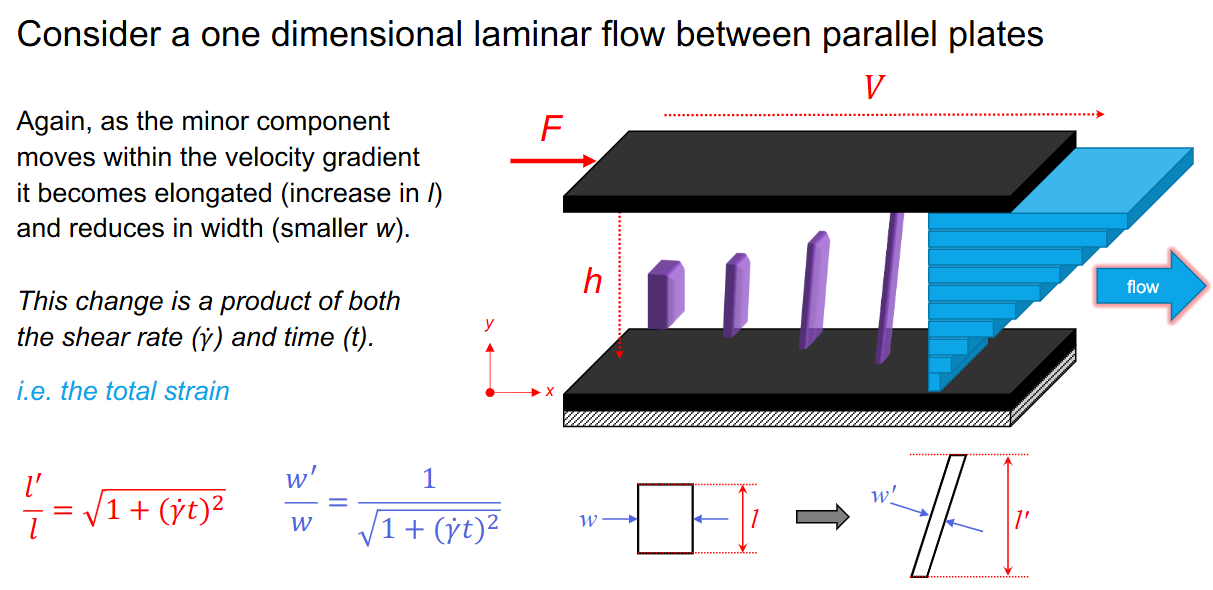
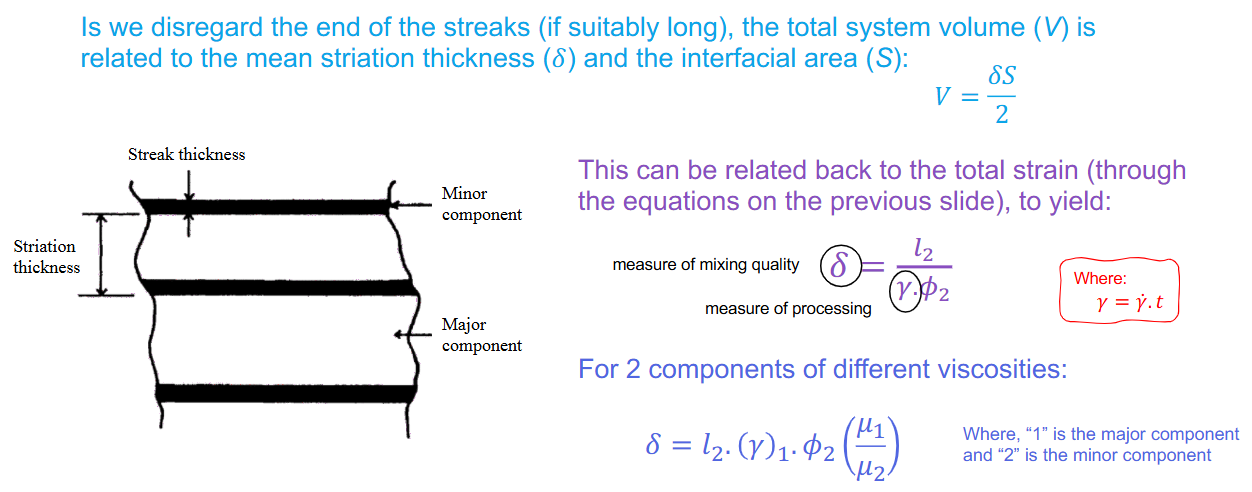
Laminar shear mixing for striated system
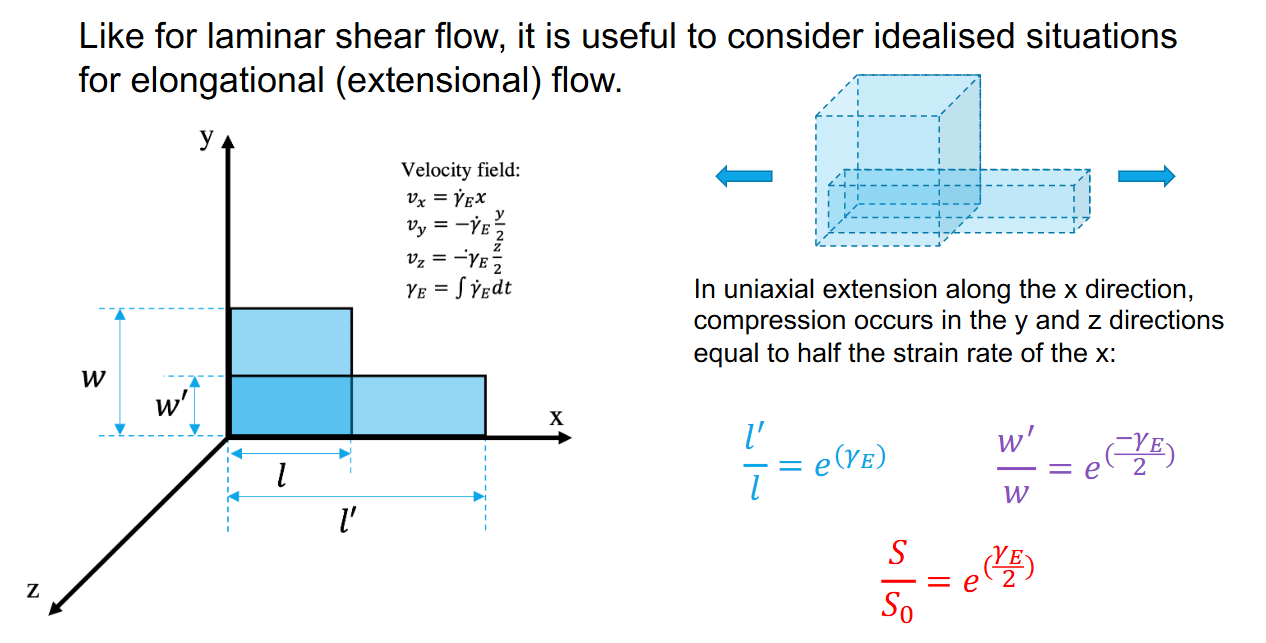
Extensional Laminar Flow- uniaxial extension
Extensional Laminar Flow- Planar extension
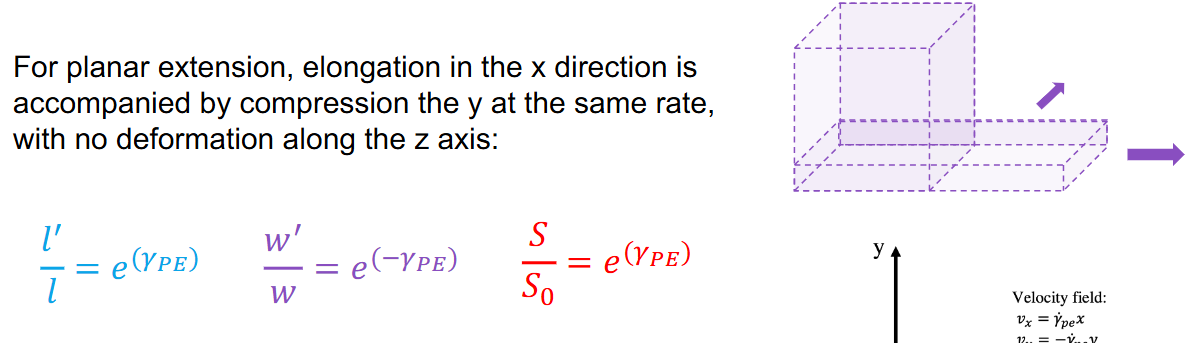
definition of distributive and dispersive mixing
Distributive: Looks at blending a material through spatial redistribution
Dispersive: Is the breaking down of a material into smaller and smaller pieces
What is an in-line static mixture, and what equation relates striation thickness, number of elements it contains, and its diameter
Mixer which uses stretching and folding or slicing and rotating to do mixing.

What are the equations used to measure dispersion
Variance
Complete segregation
Randomised Mixing

What is the Lacey mixing index and what is its equation
Ratio of how far the system is from being completely mixed and its initial state.
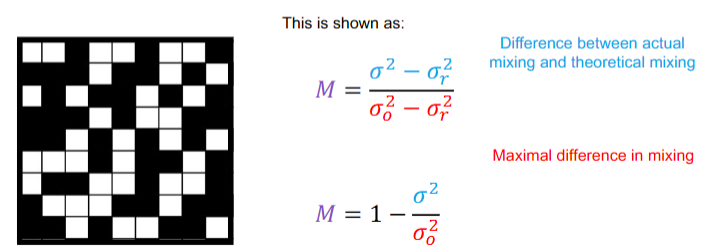
What is the coefficient of variance equation

How can pressure drop characteristics of the process be described
ratio of the static mixer pressure drop to the pressure drop of
the empty pipe
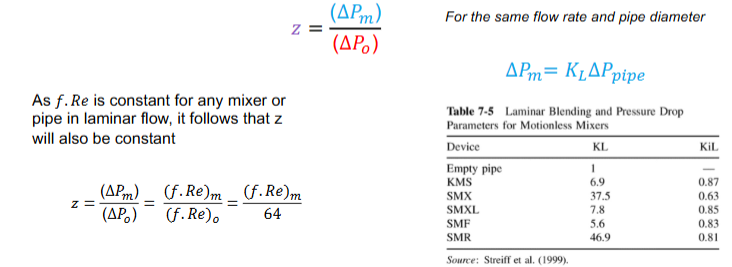
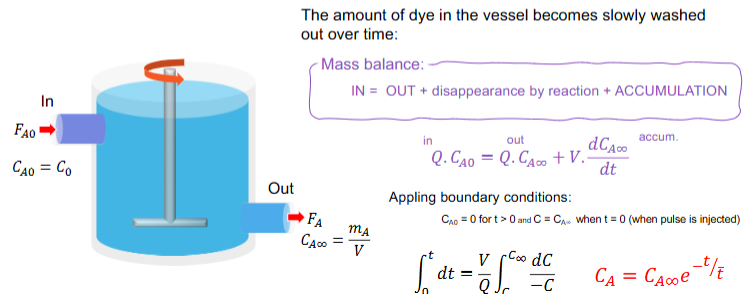
What is the mass balance equation for a perfectly backmixed CSTR
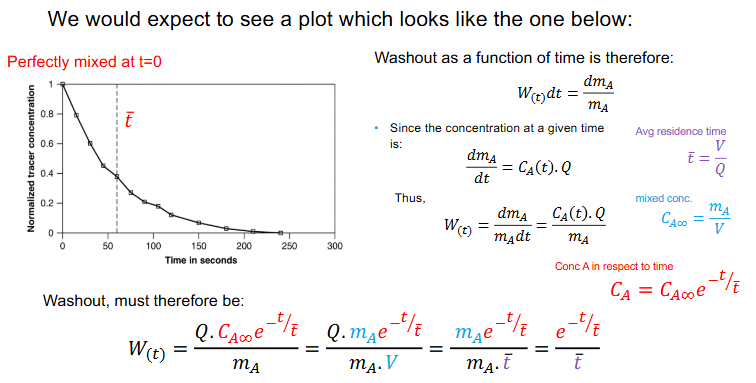
What is the washout equation as a function of time for CSTR.
Plot the graph as well
What is the concentration at any time for a plug in a PFR, give the concentration against time graph aswell.

For a fluid in a real pipe
Give the fluid velocity profile equation
and give the equation for the minimum time needed for a particle to travel through the pipe
Give the particle distribution graph against time
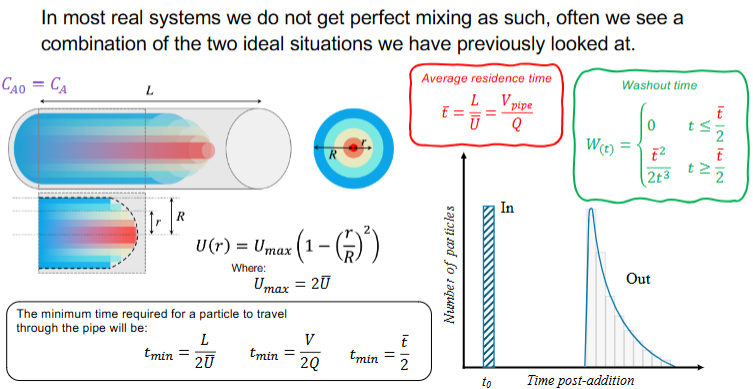
Derive the washout time for a fluid in a real pipe

What is the equation which relates the ratio of mixing times

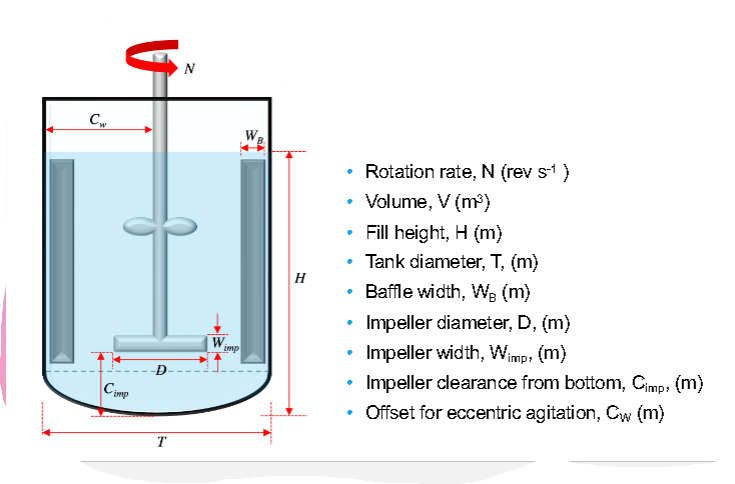
Draw a basic stirred tank
Equation for power number, Froude number, and Reynolds number
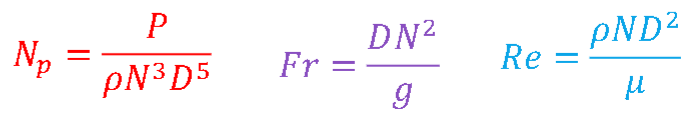
Equation for specific power
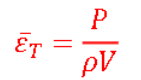
What is the importance of the Damkoeler number and what is its equation
Allows us to understand how rates of reactions are affected by transport phenomena
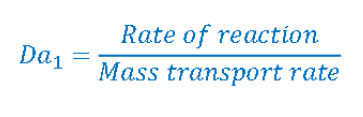
What is the relationship between mixing time and stirrer speed