Citric Acid Cycle
1/34
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
35 Terms
What are the three main stages of cellular respiration? What is cellular respiration?
Cellular Respiration: wen pyruvate from glycolysis is further oxidized to H2O and CO2.
1) Acetyl-Co A production
2) Acetyl-CoA oxidation via Citric Acid Cycle
3) Electron transfer and oxidative phosphorylation
What is coenzyme A and what is its main function?
Coenzyme A is an acyl carrier that convalently links to thiol to form a thiolester — thus wants to donate their acyl group.
→ will transfer its acyl group to pyruvate so that it can enter the citric acid cycle as Acetyl-CoA.
How is Acetyl-CoA produced?
Primarily produced via pyruvate!
→ it is oxidized by pyruvate dehydrogenase complex into Acetyl-CoA
→ uses five cofactors
→ oxidization carboxylation: irreverisble, where carboxyl group is removed from pyruvate as CO2 and 2 carbonsd become acetyl group
this is under aerobic conditions
what are the fates of pyruvate?
pyruvate has two main fates after it diffuses or is carried into mitochondria:
1) Oxidized via TCA
2) Acetyl-CoA is used in fatty acid synthesis, etc.
What are the five cofactors used by Pyruvate Dehydrogenase Complex? Why are these important — function. Also know mechanism.
TPP
Lipate
FAD
NAD
CoA-SH
What are the three enzymes used by pyruvate dehydrogenase complex? Why are these important — function. Also know the mechanism.
Pyruvate dehydrogenase — the rate limiting step! This is where pyruvate is attached to TPP as hydroxyethyl group and releases CO2
dihydrolpoyl transacetylase — here hydroxyethyl group is oxidized to acetate removing 2e- reducing S-S bond to S-H bond (thiol)
— acetyl moeity is esterified to lipoyl-SH followed by transesterification to CoA to acetyl-coA (high energy thioester)
dihydriolipoyl dehydrogenase — energy transfers to FAD and NAD (from hydroxyethyl group) to regenerate disulfides of lipoyl groups
this is under aerobic conditions
NADH can be used for ATP synthesis via oxidative phosphorylation (later)!!
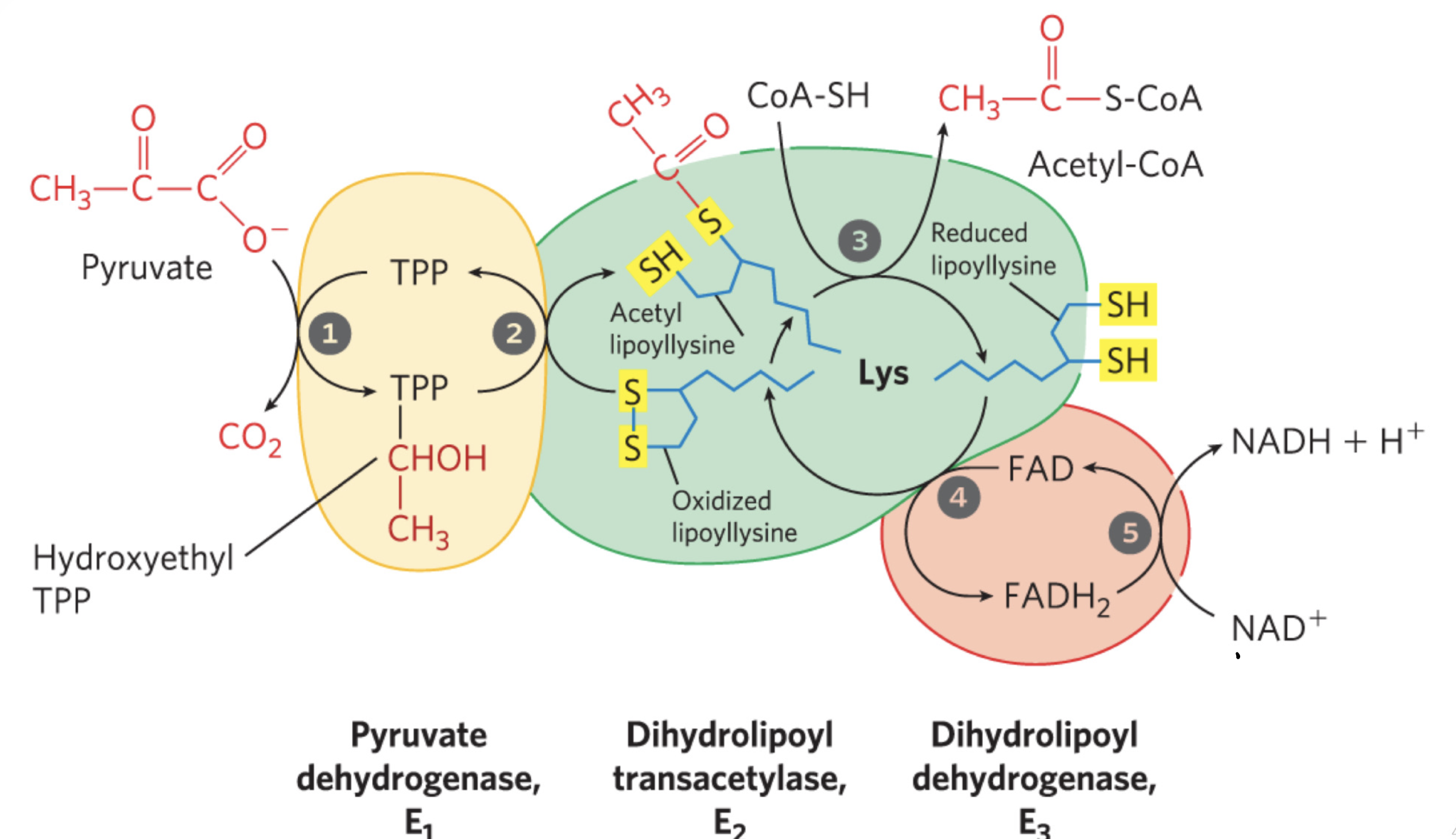
Take this time to practice the Pyruvate Dehydrogenase Complex mechanism!
Why can’t you directly oxidize acetate or acetyl-coA?
You can’t because this would primarily yield CO2 and CH4 (methane)!
→ most animals cant break down methane so its “unreactive”
→ methelyne groups however are readily metabolized so we have to make our carbon groups more reactive like succinate oxidized to oxaloacetate
What is the citric acid cycle? What does it do and what does it make? Where does it take place?
This is where acetyl-CoA from the PDC reacts with oxaloacetate to generate 3 NADH, 1 FADH2 and 1 ATP.
→ this occurs in the mitochondria of _____
What is the first step of the citric acid cycle and what enzyme catalyzes this?
1: Formation of Citrate by Citrate synthase
Acetyl-CoA reacts with oxaloacetate to form citrate
→ methyl needs to attacj oxaloacetate to be reacation (now methelyne)
→ can undergo oxidation
→ large and negative free energy.
Why does the first step of the citric acid cycle have a large, negative delta G?
→ due tt break down of high energy thioester bond via hydrolysis
→ stop reaction from going backwards, helps oxaloacetate in low contrations and acetyl-coA to commit to cycle.
explains why its irreversible!!
Take this time to practice the Citrate Synthase mechanism.
What is the second step in the citric acid and what enzyme catalyzes this?
2: Formation of isocitrate via enzyme aconitase (two steps)
Step 1) Formation of cis-aconitate
does not leave active site
Step 2) Formation of isocitrate
through reversibly addtion of water can generate isocitrate or citrate
→ Postive delta G
→ aconitase has iron-sulfur cluster
Why does aconitase reaction move froward if it has a postive delta G?
since delta G is postive — subtrates are favored (citrate)
→ however reaction proceeds forward because irrevsible consumption of isocitrate (isocitrate is quickly used up by dehydorgenase - pushing the reaction forward)
How is it possible that citrate which is non-chiral makes only one form of isocitrate?
What is the third step in the citric acid and what enzyme catalyzes this?
3: Formation of alpha-ketogluterate by isocitrate dehydrogenase
→ oxidative decarboxylation: carbonyl removed and released as CO2.
→ requires NAD —> formation of NADH
→ Mn2+ stabilizes enolate intermediates
Take this time to draw the mechanism for isocitrate dehydrogenase.
What is the fourth step in the citric acid and what enzyme catalyzes this? What is this similar to?
4: Oxidation of alpha-ketogluterate to succinyl-CoA by alpha-KG dehydrogenase complex
REVIEW!!
What is the fifth step in the citric acid and what enzyme catalyzes this?
5: Converstion of succinyl-coA to succinate
→ substrate level phosphorylation that produces GTP by substate acyl-phosphate
→ hydrolysis of thioester — so negative delta G
What type of reaction of GTP production?
This is part bi-bi and part ping pong
What is the sixth step in the citric acid and what enzyme catalyzes this?
6: Oxidation of succinate to fumerate
What is the seventh step in the citric acid and what enzyme catalyzes this?
7: Hydration of fumurate to malate by fumerate hydratase
highly stereoselective and sterospecific
only generates L-malate will not dehydrate D-malate
What is the eight step in the citric acid and what enzyme catalyzes this?
8: oxidation of malate to oxaloacetate by malate dehydrogenase
→ high delta G (FAVORS substrate - malate) but proceeds forward because oxaloacetate consumption by citrate syntehase
→ low concentration of oxaloacetate pushes product formation
Why is it important to have low concentrations of oxaloacetate in CAC?
Low oxaloacetate levels help drive the citric acid cycle forward by pulling unfavorable reactions and preventing buildup that would slow the cycle.
Why does oxidative metabolism (PDHC + citric acid cycle make more ATP compared to fermentation?
Oxidative metabolism makes more ATP because it fully oxidizes glucose and uses NADH/FADH₂ in the electron transport chain to drive oxidative phosphorylation, while fermentation only produces ATP through glycolysis.
Oxidation
Pyruvate → acetyl-CoA via pyruvate dehydrogenase (PDH)
Acetyl-CoA enters the citric acid cycle
Produces lots of NADH and FADH₂
These feed into the electron transport chain (ETC)
Oxygen is the final electron acceptor
Result: ~30–32 ATP per glucose while glycolysis only produces 2!!
Can you convert acetyl-CoA back to glucose?
No!
When acetyl-CoA enters the citric acid cycle → its two carbons are fully oxidized and released as 2x CO₂, meaning there is no net gain of carbon skeletons that could be used to form glucose.
CAC —> ATP synthases and not glucose
Oxaloacetate can’t be used for glycolysis because no net production!
What is an anplerotic rxn?
This rxn replenishes intermediates when use in biosythensis lowers the concentration of intermediates enough to SLOW DOWN cycle.
Is TCA an amphiolic pathway?
Yes, it does both catabolism and anabolism
catabolism: breaking down to power the cell
makes ATP
oxidizes
energy release
anabolism: buiidlign up to use power
uses ATP
reduces
growth or storage
Why is TCA important for muscle contraction?
→ Muscle contraction = ATP consumption
→ TCA cycle = ATP supply chain (indirectly)
The TCA cycle doesn’t make muscles contract—it keeps the ATP production system running that allows contraction to continue.
The TCA cycle is central to energy production because it generates NADH and FADH₂ that drive ATP synthesis, and that ATP is what muscles use for contraction.
Do CAC intermediates only stay in CAC or do they get shunted off?
TCA = feeder pathway, where intermediates get shunted off to other pathways.
Citrate → Fatty Acids, sterols
alpha- KG → Glutamate
Succinyl-CoA → Porphyrins, heme
Oxaloacetate → aspartate and Asparagine → prymidines
Oxaloacetate → PEP
PEP → Glucose or Serine, Glycine, Cysteine, Phenylalanine, Tyrosine, Tryptophan
What are four anaplerotic reactions that replenish CAC intermediates?
1) Formation of oxaloacetate from pyruvate by pruvate carbocylase
at liver, kidney
2) Formation of oxaloacetate from PEP by PEP carboxykinase
at heart, skeletal muscle
3) Formation of oxaloacetate from PEP by PEP carboylase
plants, yeast
4) Formation of malate from pyruvate by malic enzyme
bacteria
Why is pyruvate carboxylase one of the most imporant anaplerotic reactions?
Formation of oxaoacetate by reversible carboxylation of pyruvate (keeps CAC running)
requires energy by ATP and needs Biotin
inactivated when low concentration of acetyl coA (positive allosteric modulator)
How is PDHC regulated by PDHC kinase?
PDHC is regulated by products of PDH: ATP, acetyl-coA and NADH
→ when there are high PDH products → PDHC kinase will phosphorylate PDHC at ser residue to inactive via ATP making ADP and stop feeding into CAC
How is PDHC kinase regulated?
PDHC kinase is regulated by PDH substrates: ADP, pyruvate, NAD+
This will cause phophatase to remove phosphate group and activate PDHC so it can make acetyl-coA and enter CAC
What are the three irreversible steps in CAC? How are they regulated?
1) citrate synthase:
→ activated by low energy: ADP
→ inactivated by high energy and products: NADH, citrate and ATP
and succinyl-CoA = signals downstream accumulation in the TCA cycle - so that more acetyl-coA doesn’t enter
2) isocitrate dehydrogenase
→ activated by low energy: Ca2+ and ATP
Ca²⁺ activates enzymes of CAC to increase ATP production in response to increased cellular energy demand, during muscle contraction.
→ inactivated by high energy: ATP
3) alpha-KG dehydrogenase complex
→ activated by low energy: Ca2+
→ inactivated by high energy: succinyl-coA, NADH