3.3.1.0 the basics
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
13 Terms
what is organic chemistry?
the chemistry/study of the millions of covalent compounds of the element carbon
why does carbon form a vast number of compounds?
because it can form strong covalent bonds with itself
what does the fact that carbon can form covalent bonds with itself result in?
it enables it to form long chains (up to 5000 in length) of carbon atoms + hence an almost infinite variety of carbon compounds are known
the physical + chemical properties of organic compounds depend on what two factors?
the number + arrangement of carbon atoms in the molecule
the functional groups in the molecule
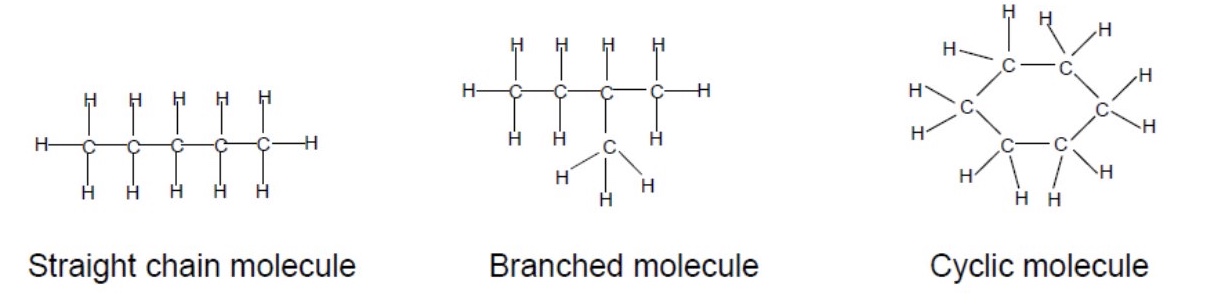
give the 3 ways carbon atoms can arrange themselves
straight chain molecule
branched molecule
cyclic molecule
what is a functional group?
a specific atom or group of atoms which give certain physical + chemical properties to the molecule
what is a homologous series?
organic compounds with the same functional group but a different number of carbon atoms are said to be part of the same homologous series
what are the 4 main characteristics/features of a homologous series?
they have the same general formula
they have similar chemical properties
there is a trend in their physical properties
one differs from the next by CH₂
give the first 4 alkanes
methane
ethane
propane
butane
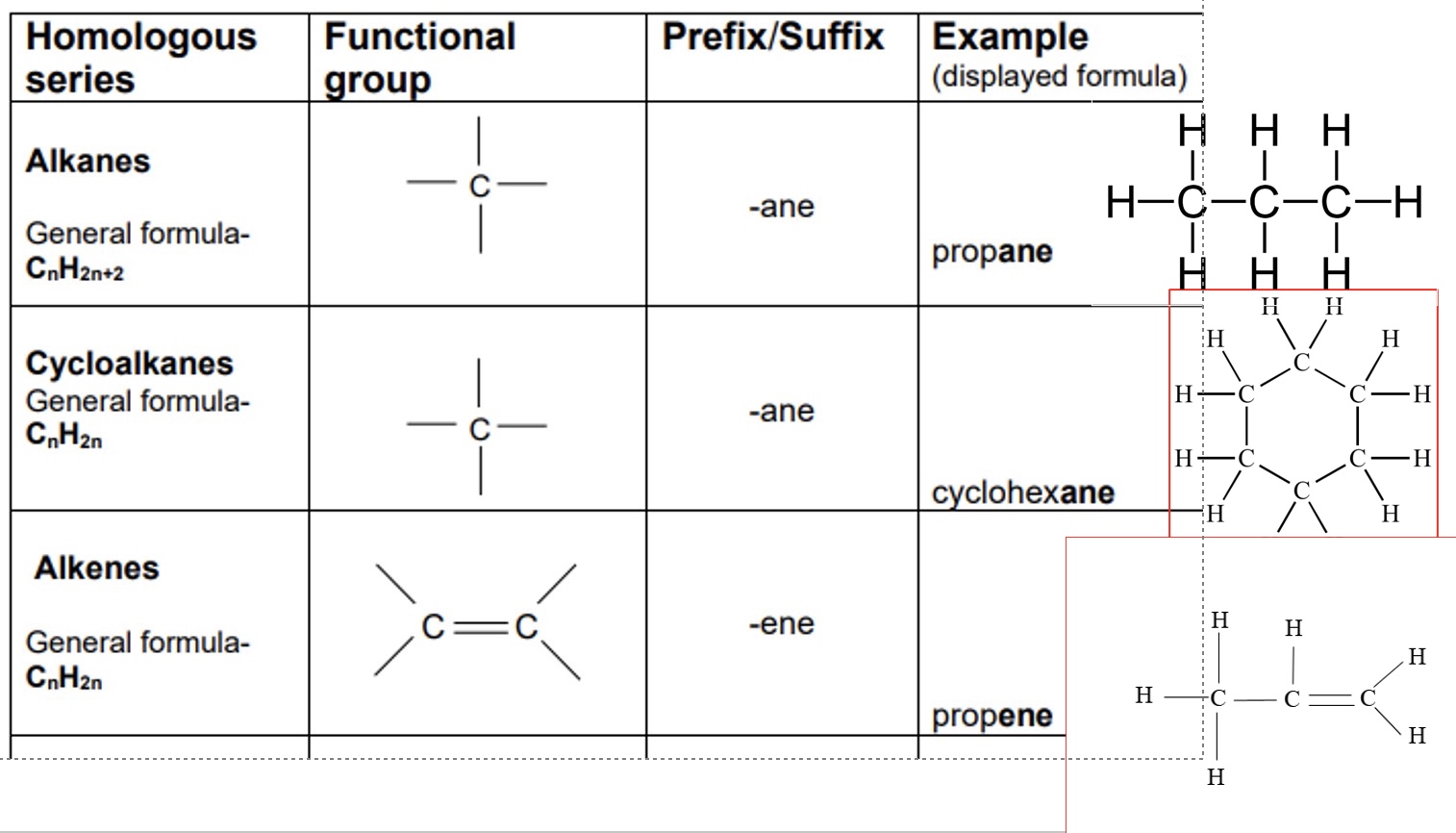
give the general formula, functional group, prefix/suffix + an example of:
a. alkanes
b. cycloalkanes
c. alkenes
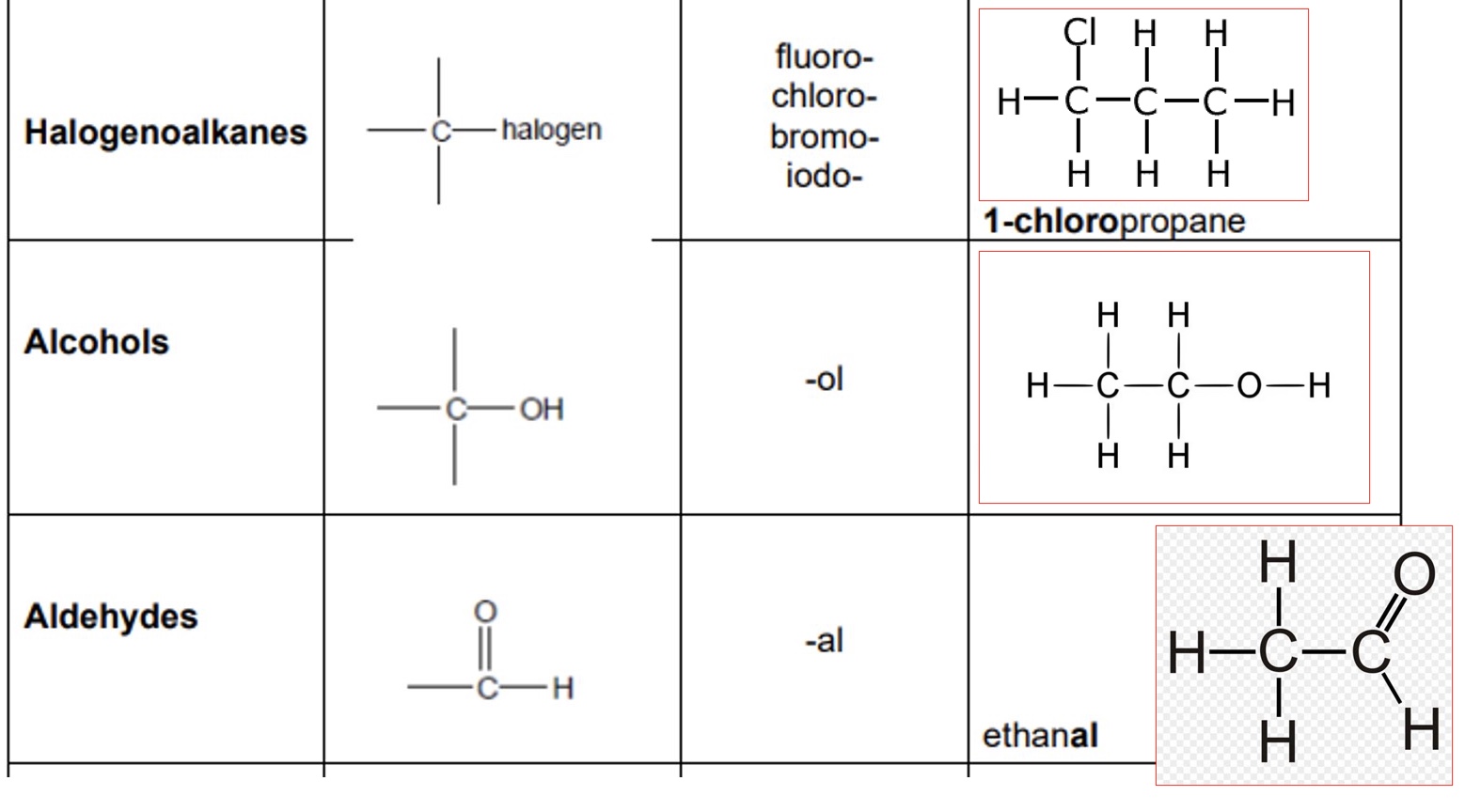
give the functional group, prefix/suffix + an example of:
a. halogenoalkanes
b. alcohols
c. aldehydes
give the functional group, prefix/suffix + an example of:
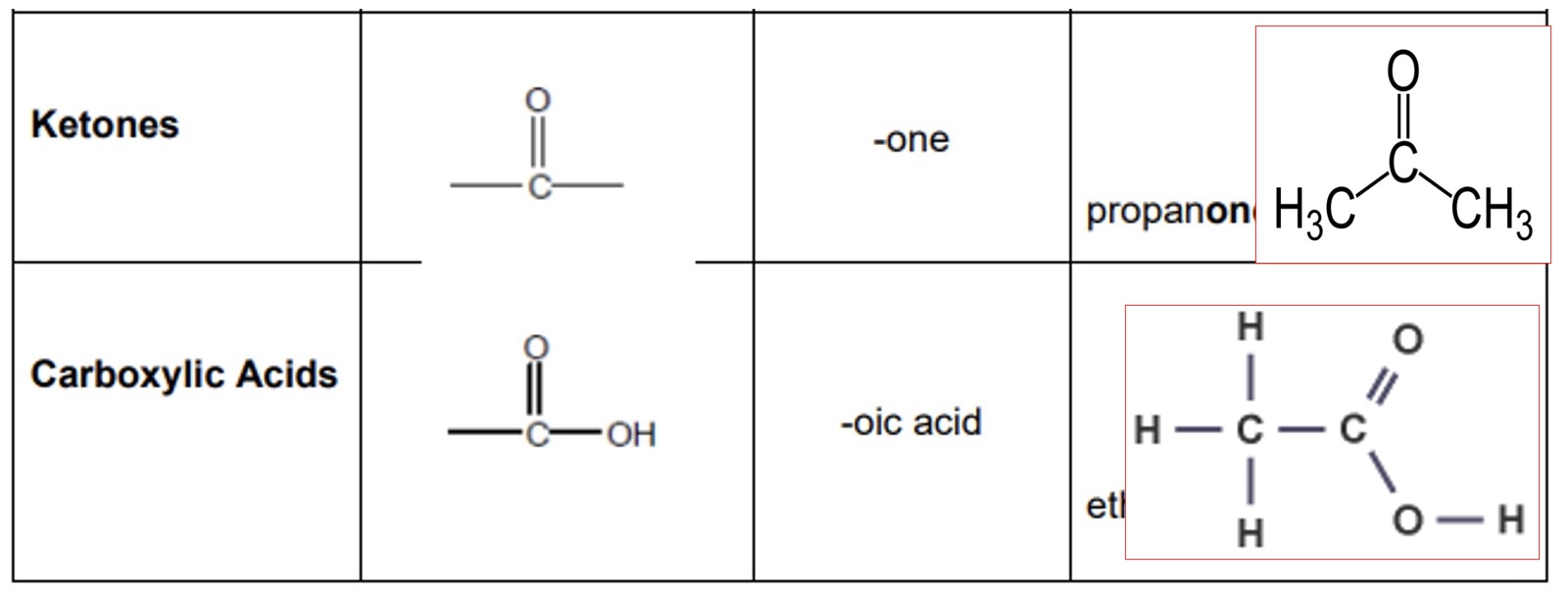
a. ketones
b. carboxylic acids
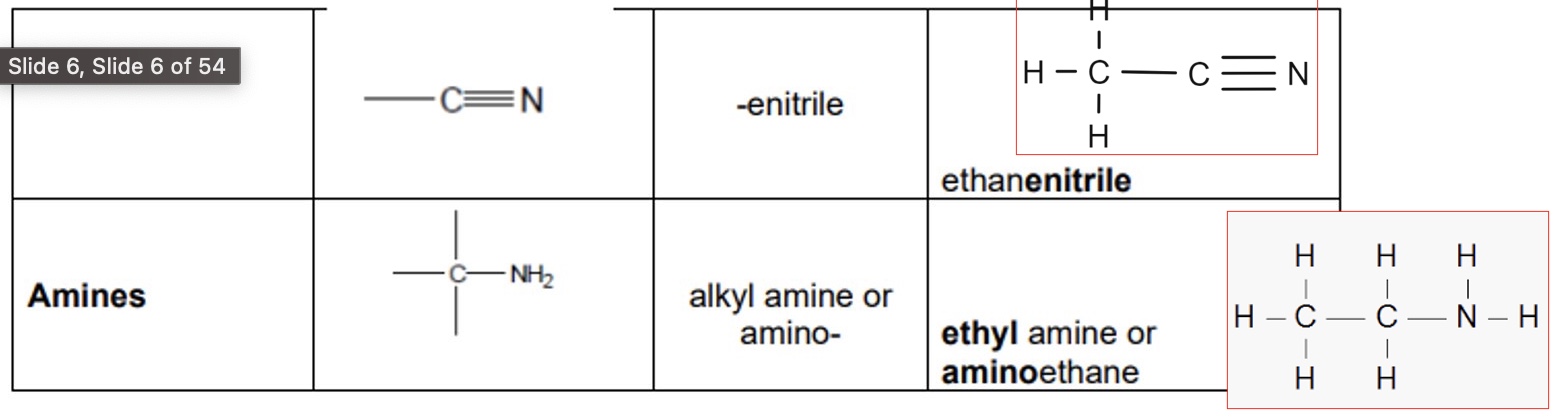
give the functional group, prefix/suffix + an example of:
a. nitriles
b. amines