Energetics
1/33
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
34 Terms
What is the enthalpy change of a reaction?
The heat energy change of a reaction at a constant pressure.

What does this symbol represent?
Standard conditions and standard states.
What are standard conditions for:
a) temperature?
b) pressure?
c) concentration?
a) 298K
b) 100kPa
c) 1 moldm-3
Draw the symbol for enthalpy change.

What are the units for enthalpy change?
kJmol-1
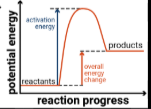
What is an endothermic reaction?
A reaction that absorbs energy from it’s surroundings.
Which has higher energy in an endothermic reaction, the reactants or the products?
The products.
Is a reaction with a positive enthalpy change exothermic or endothermic?
Endothermic.
Sketch an energy profile or an endothermic reaction.
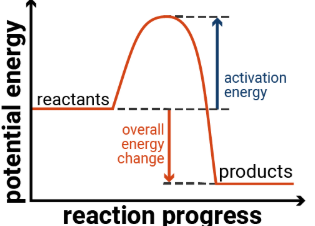
What are exothermic reactions?
Reactions that release energy into their surroundings.
Which has a higher energy in an exothermic reaction, the reactants or the products?
The reactants.
Is a reaction with a negative enthalpy change exothermic or endothermic?
Exothermic
Sketch an energy profile for an exothermic reaction.
Is bond breaking exothermic or endothermic, why?
Endothermic. Bonds need to absorb energy from the surroundings to be broken.
Is bond making exothermic or endothermic, why?
Exothermic. Energy is released into the surroundings when bonds are broken.
If more energy is required to break the bonds in the reactants than released when forming bonds for the products, will the reaction be exothermic or endothermic? Why?
Endothermic. More energy will be taken in from the surroundings overall meaning the enthalpy change will be positive meaning the reaction is endothermic.
If more energy is released when the bonds are formed than energy absorbed when bonds are broken, will the reaction be exothermic or endothermic? Why?
Exothermic. The overall enthalpy change of the reaction will be negative meaning the reaction is exothermic.
What is the mean bond enthalpy?
The mean amount of energy stored in a specific bond.
True or false? Bonds of the same type don’t all have the same amount of energy.
True.
Give the equation for calculating enthalpy change with bond enthalpies.
Enthalpy change = total energy to break bonds (in reactants) - total energy released forming bonds (in products)
What is calorimetry used for?
Working out enthalpy of combustion.
Talk me through and explain the general process of calorimetry.
1) Weigh the fuel.
2) Wind shield placed around flame so the flame doesn’t move.
3) Burn the fuel below the water, heating up the water.
4) Reweigh the fuel to see how much has been burned.
Give a thing in calorimetry that can lead to inaccuracies in results.
Some energy from burning the fuel is not transferred to the water but lost to the surroundings.
Give an equation to calculate the amount of energy transferred to the water in calorimetry.
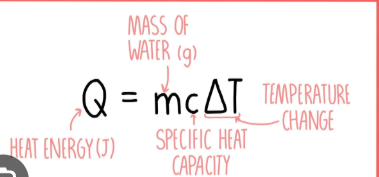
How do you convert from J to kJ?
Divide by 1000.
Give an equation for calculating enthalpy change once you have worked out heat energy.
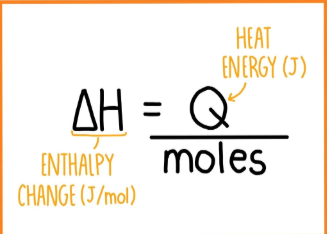
(MOLES OF FUEL/LIQUID THAT YOU WANT TO WORK OUT ENTHALPY OF NEUTRALISATION OF).
What is an assumption (that leads to inaccuracies) when calculating (using calorimetry):
a) enthalpy of combustion?
b) enthalpy of neutralisation?
a) All solutions have the specific heat capacity of water.
b) All solutions have the same density as water (1 g dm-3).
True or false? When calculating enthalpy of neutralisation once you’ve worked out heat energy, you divide heat energy by the moles of all the substances added to the substance being neutralised.
False. YOU ONLY DIVIDE HEAT ENERGY BY THE MOLES OF THE SUBSTANCE BEING NEUTRALISED.
State Hess’s law.
The total enthalpy change of a reaction is independent of the route taken.
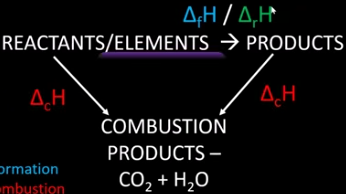
Define standard enthalpy of combustion.
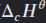
The enthalpy change when one mole of a substance is combusted completely in oxygen, in standard states and under standard conditions.
Define standard enthalpy of formation.
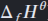
The enthalpy change when one mole of a compound is formed from it’s constituent elements in their standard states and under standard conditions.
Draw and label the general Hess cycle for working out the enthalpy of a reaction when you have values for the enthalpy of formation.
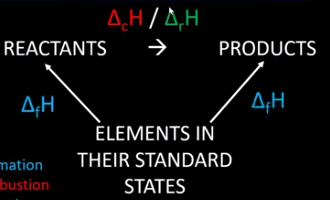
True or false? Go with the arrow, keep the sign the same. Go against the arrow, change the sign.
True.
Draw and label a general Hess cycle for combustion.