Chemistry IGCSE - States of matter
1/13
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
14 Terms
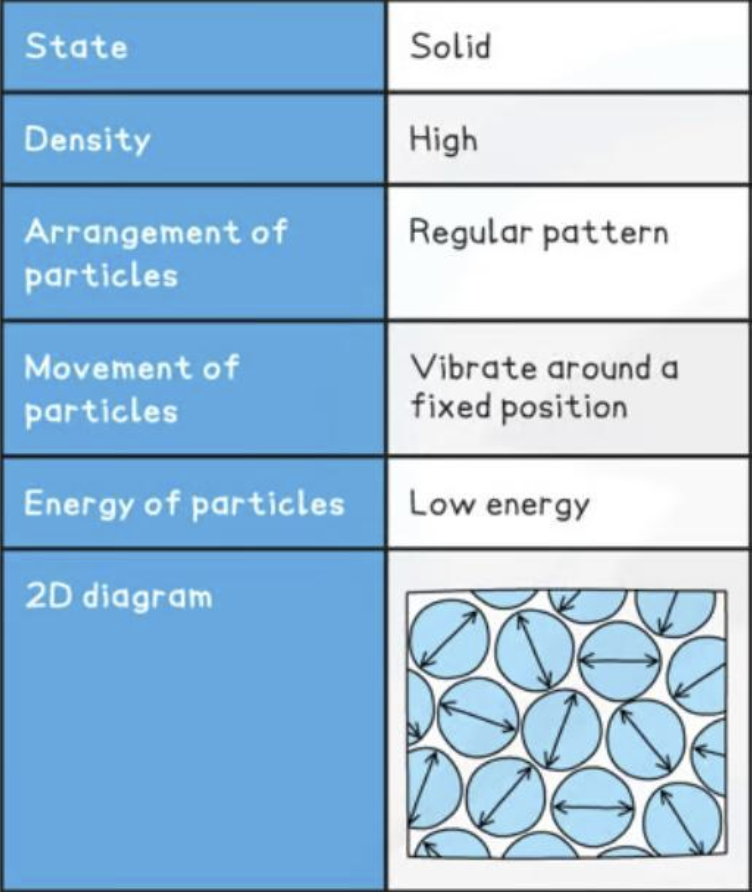
Solid
definite volume and shape
incompressible
Liquid
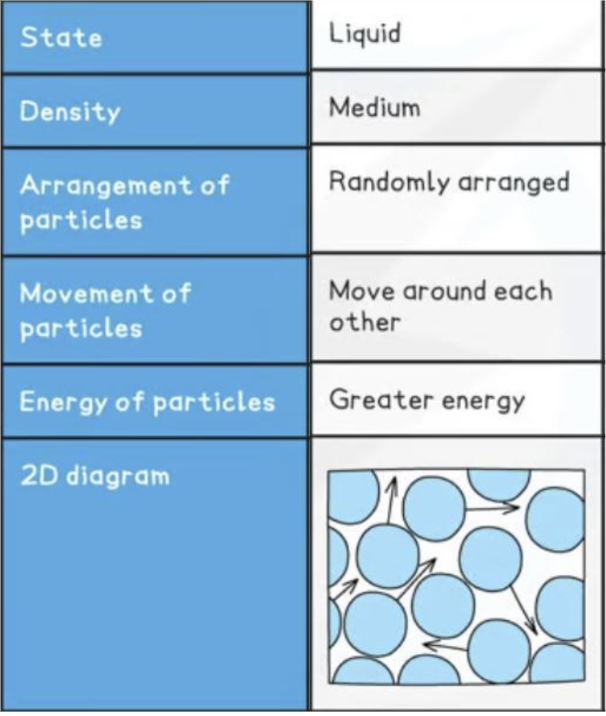
definite volume
no definite shape
slightly compressible
Gas
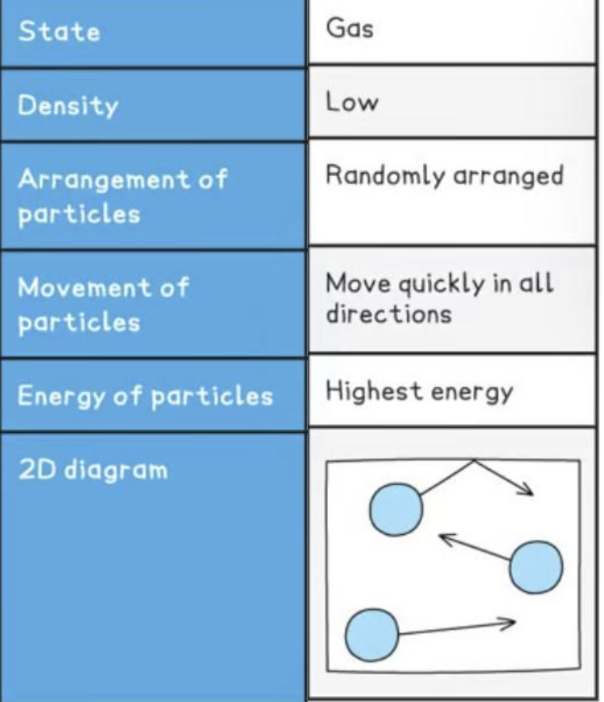
no definite shape nor volume
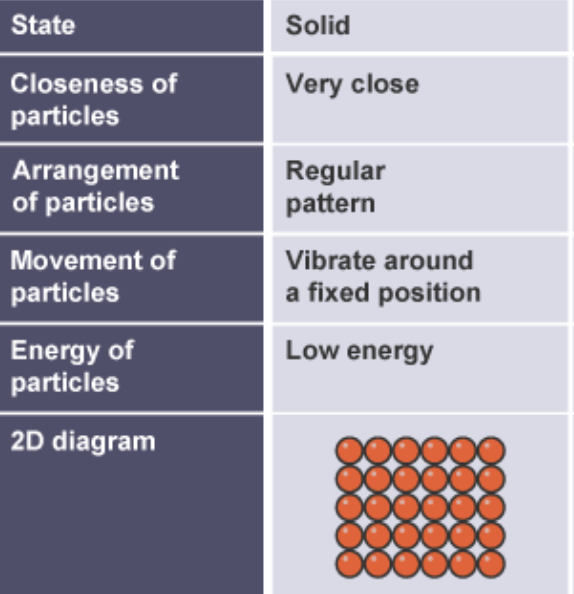
structure of solids in terms of particle separation, arrangement and motion
packed tightly together
regular arrangement
vibrate in fixed position
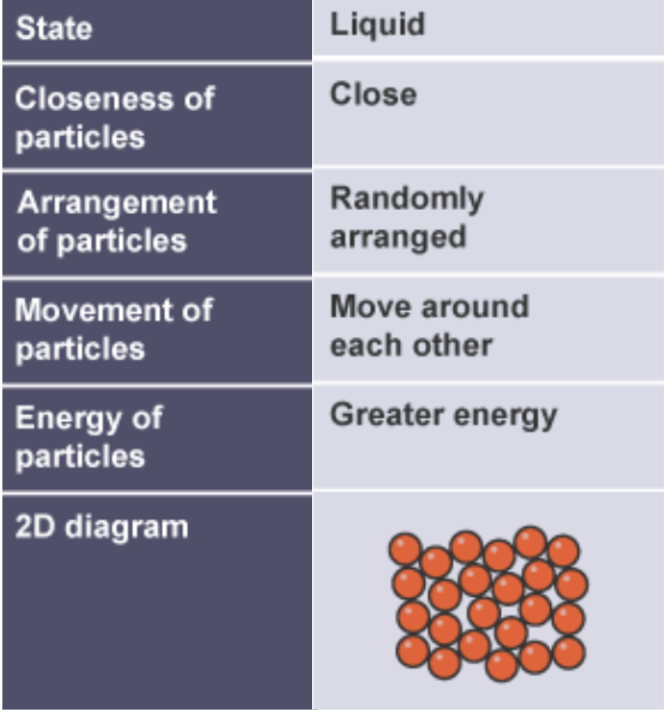
structure of liquids in terms of particle separation, arrangement and motion
arranged randomly
close together, touching close particles
random vibration
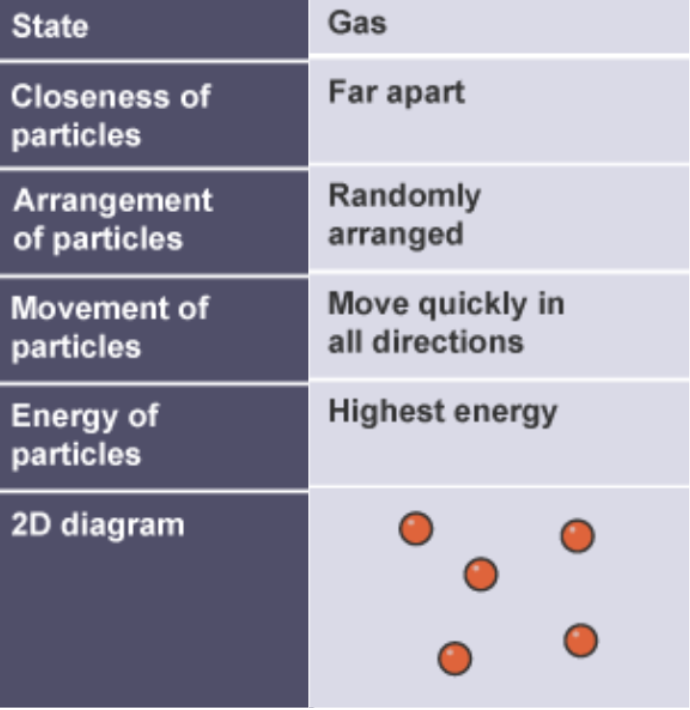
structure of gases in terms of particle separation, arrangement and motion
far apart with no regular arrangement
separate far apart
move randomly quickly
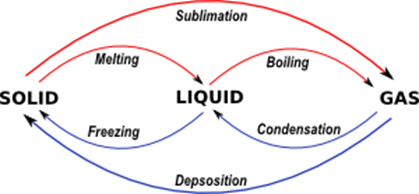
Changes of state in terms of melting, boiling, evaporating, freezing and condensing
Melting: solid → liquid
Evaporating: liquid → gas
Freezing: solid → gas
Condensing: liquid → solid
Describe the effects of temperature and pressure on the volume of a gas
increase in temperature → increase in kinetic energy of particles → more forceful collisions against container wall → increase in pressure → the walls can expand → gas volume increases
kinetic particle theory
all matter is composed of tiny particles which are in random movements through space that exists between them
Explain changes of state in terms of kinetic particle theory
decrease in kinetic energy → movements decrease → goes from liquid to solid
increase in kinetic energy → movements of particles increase → goes from solid to liquid
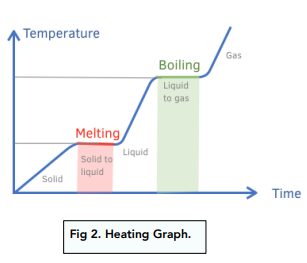
Heating curves
temperature of substance increase with kinetic energy → solid to gas
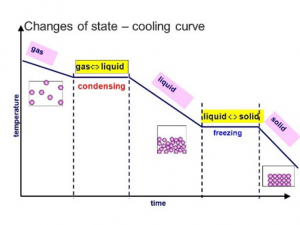
Cooling curves
temperature of substance decreases with kinetic energy → gas to solid
Describe and explain diffusion in terms of kinetic particle theory
diffusion → random movement of particles from high to low concentration
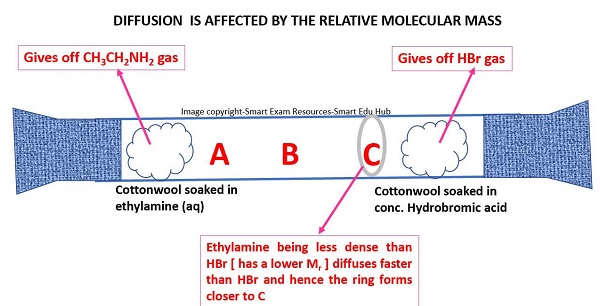
Describe and explain the effect of relative molecular mass on the rate of diffusion of gases
Molecules with lighter masses → diffuses faster
Molecules with heavier masses → diffuses slower