CH302 - Unit 4: Kinetics (Tuley) (Master Edition)
1/106
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
107 Terms
What are isotopes?
Like elements that has the same number of protons but different number of neutrons.
How many isotopes are known?
3000
How many isotopes are predicted to exist?
Up to 7000
How many isotopes are stable and where do they lie on the periodic table?
250 isotopes and they consists of elements below Pb.
Which element has the heaviest stable isotopes?
Pb
Small elements are stable with a ___ ratio of _______ and _______?
1:1 ratio, neutrons and protons
What do large nuclei need to be stable?
Increasing numbers of neutrons
Fission
The splitting of large unstable isotopes. This is the basis for the first nuclear bombs as well as nuclear power plants.
Fusion
The combining of small particles in a highly exothermic process. It occurs within the sun and is potentially the ideal source of clean nuclear power to solve to solve the world’s energy crisis. Technological challenges have slowed progress on the development of fusion reactors.
Radioactive Decay
The process by which the approximately 7000 isotopes decay to the 250 stable isotopes on Earth. There are many types of radioactivity that involves small particles like photons, protons, electrons, positrons, alpha particles, etc., to be emitted with a half life that can be extremely fast or millions of years.
Alpha Decay (α)
Loss of 42 He
ex. 23290 Th → 22888 Ra + 42 He
Beta Decay (β)
Neutron converted to proton ejecting e-.
ex. 31 H → 32 He + 0-1 e-
Gamma Decay (γ)
Ejection of photon
ex. 15266 Dy → 15266 Dy + hv
Positron/Beta Plus Decay (β+)
Proton converted to neutron ejecting positron and electron neutrino.
ex. 85 B → 84 Be + 01 e+ + ve
What does the number 6 (top number) represent in the carbon element, 612 C?
Atomic Number
What does the number 12 (bottom number) represent in the carbon element, 612 C?
Atomic Mass
What is the rate determining step and how does it affect the reaction?
The r.d.s. is the slowest step and controls the overall speed of the reaction.
Each bump/hill on a reaction coordinate diagram is equal to what?
One transition state
At transition states, bonds are _____ formed and _____ be isolated.
half formed and can’t be isolated.
at transition states, old bonds are beginning to break while the new bonds are just starting to form. The system is also fleeting.
At intermediates, bonds are _____ formed and _____ be isolated.
fully formed and can be isolated
Activation energy is always going to be _________.
Positive. Ea will always be positive as it represents the minimum energy/energy barrier required to break chemical bonds and initiate a reaction (which must be supplied to the reactants to reach the transition state).
How does a higher Ea affect the reaction?
A higher activation energy means fewer molecules can succeed/overcome the barrier, thus the reaction will process slowly.
How does a lower Ea affect the reaction?
A lower activation energy means more molecules can react/succeed and will ultimately lead to a faster rate.
What do catalyst do to the speed of a reaction? Do they change the energy or get consumed?
Catalyst speed up reactions by providing an alternative reaction pathway with a lower Ea. They do not change the overall energy of the reactants and products, nor do they get consumed. They just make it easier for reactant molecules to reach the transition state.
How does temperature affect rate?
Proportionally. Temperature can affect how many molecules have enough kinetic energy. At higher temperatures, molecules move faster and they collide with greater energy. Thus, higher temps → more collisions → higher rate (collisions happen more frequently and with the proper orientation).
Molecules that start as a product then turn into a reactant in the following steps classify as an….?
Intermediate
Molecules that start as a reactant then turn into a product in the following steps classify as an….?
Catalyst
What is the unit for a rate constant of the 0th order?
Msec-1 or M/s
What is the unit for a rate constant of the 1th order?
sec-1 or 1/s
For 1st Order, the rate of reaction decreases ______________ with time.
Exponentially
What is the unit for a rate constant of the 2th order?
M-1sec-1
What are the reactants and products in ozone layer decomposition?
Reactants: O3
Products: O2
What is the catalyst used in ozone layer decomposition?
Homogenous chlorine radical (Cl•)
How does ozone layer decomposition affect the environment?
The hole in the ozone layer is an environmental issue that was addressed in the 1980s and 1990s with the elimination of chlorofluorocarbons.
What are the reactants and products in a catalytic converter?
Reactants: NO(x), CO, CH(x)
Products: N2, CO2, H2O
What happens to smog/nitrogen oxides in a catalytic converter reaction?
Smog/nitrogen oxides (NO, NO2, NO3) undergo reduction to from nitrogen gas (N2).
What happens to carbon monoxide in a catalytic converter reaction?
Carbon monoxides and hydrocarbons (CHx) undergo oxidation to from carbon dioxide (CO2).
What is the catalyst used in a catalytic converter?
Heterogenous Pt/Pd metal catalyst
What is a benefit of using catalytic converters?
Catalytic converters significantly improve air quality.
What are the reactants and products of the haber process?
Reactants: N2, H2
Products:NH3 (Ammonia, used in agriculture)
What is the catalyst used in the haber process?
Heterogenous slightly oxidized iron powder.
Is the haber process endo or exothermic and how does that affect the reaction?
The Haber Process is exothermic and will shift towards the left when the temperature increases. An increase in temperature also speeds up ammonia formation.
What is the name for a molecule with a total number of 1 carbon atom?
Methane
What is the name for a molecule with a total number of 2 carbon atom?
Ethane
What is the name for a molecule with a total number of 3 carbon atom?
Propane
What is the name for a molecule with a total number of 4 carbon atom?
Butane
What is the name for a molecule with a total number of 5 carbon atom?
Pentane
What is the name for a molecule with a total number of 6 carbon atom?
Hexane
What is the name for a molecule with a total number of 7 carbon atom?
Heptane
What is the name for a molecule with a total number of 8 carbon atom?
Octane
What is the name for a molecule with a total number of 9 carbon atom?
Nonane
What is the name for a molecule with a total number of 10 carbon atom?
Decane
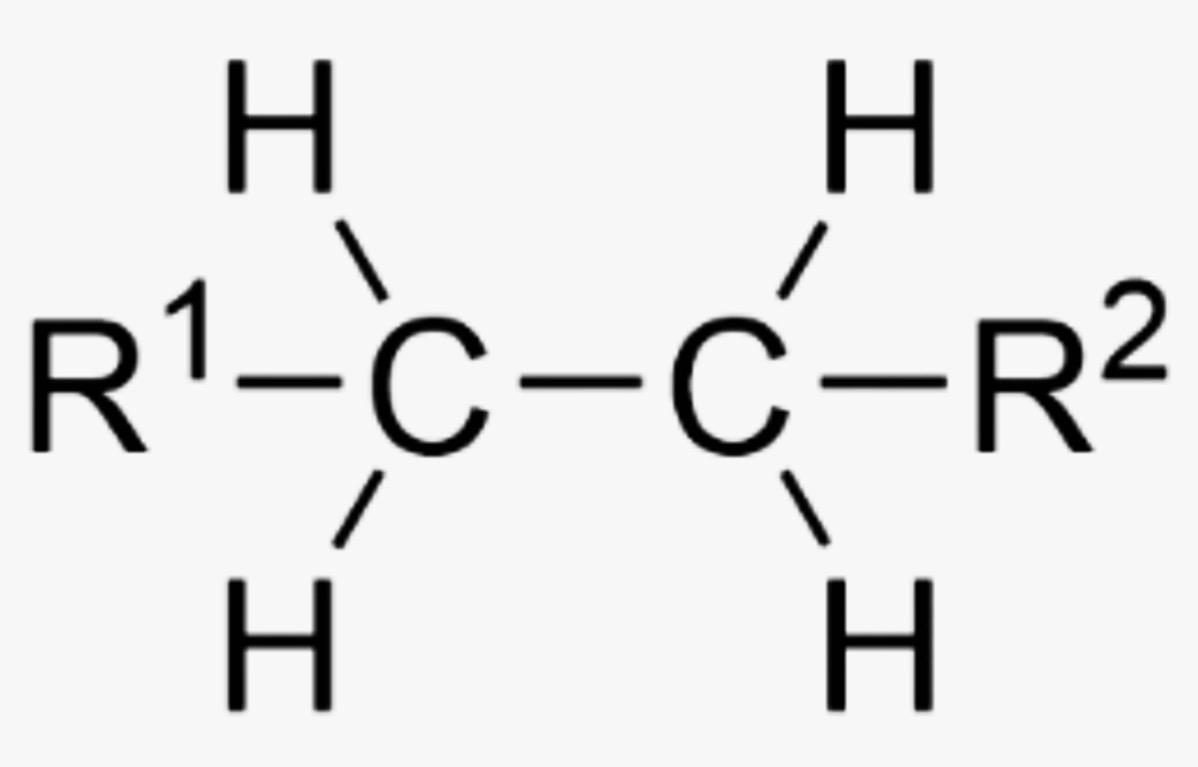
What is the name of this organic functional group?
Alkane
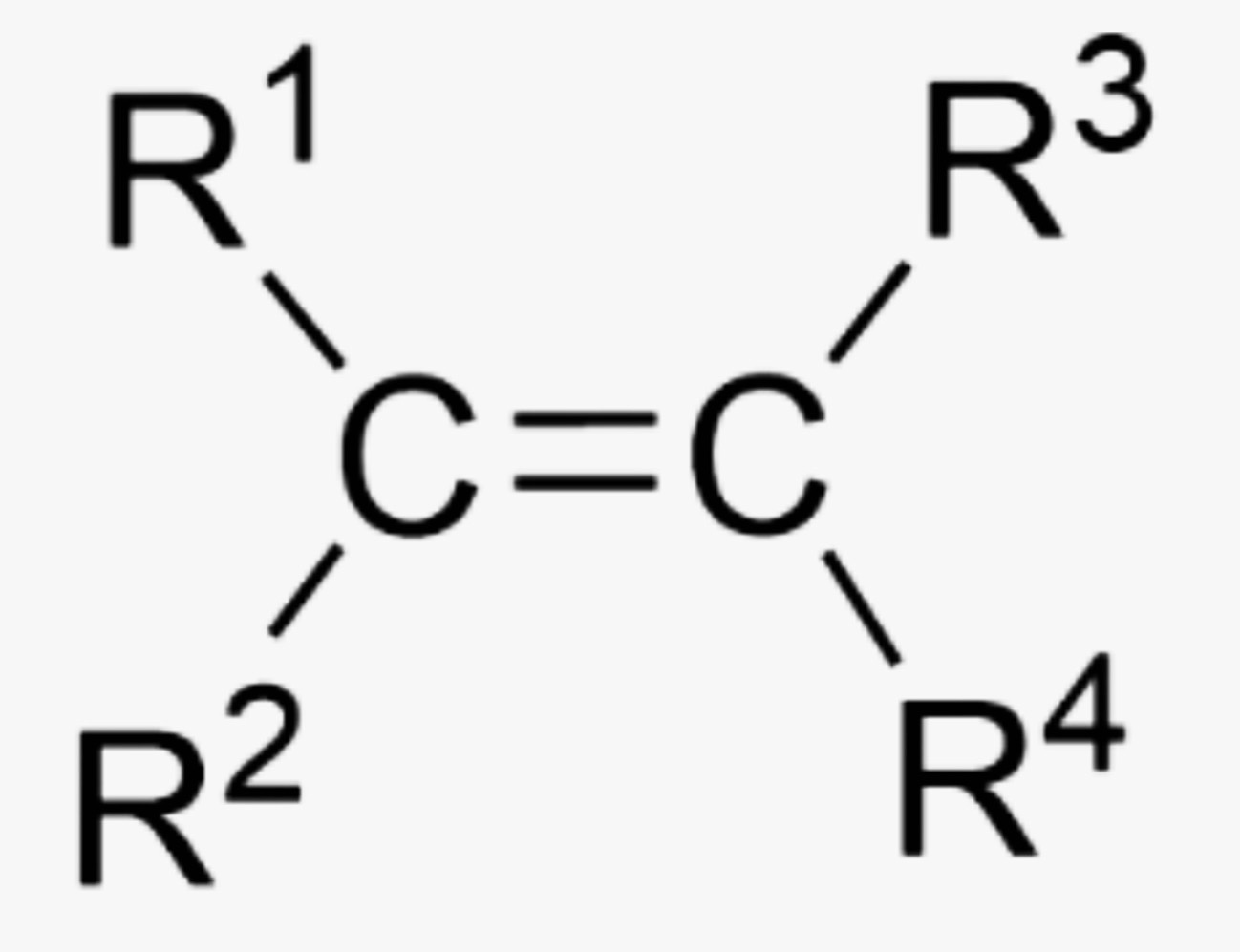
What is the name of this organic functional group?
Alkene
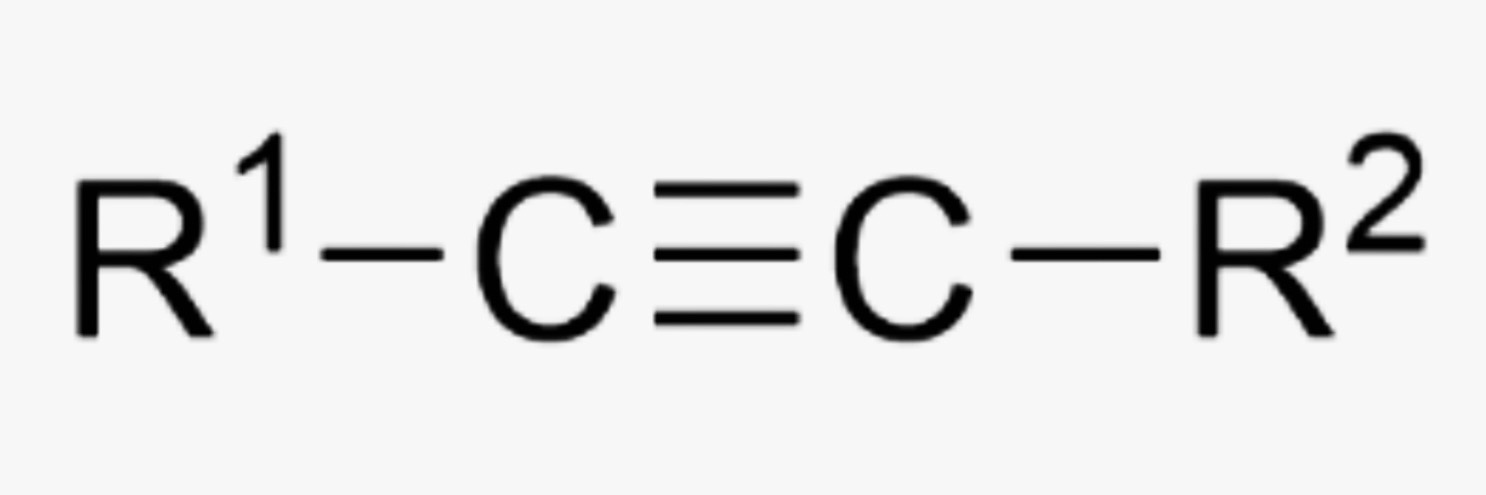
What is the name of this organic functional group?
Alkyne
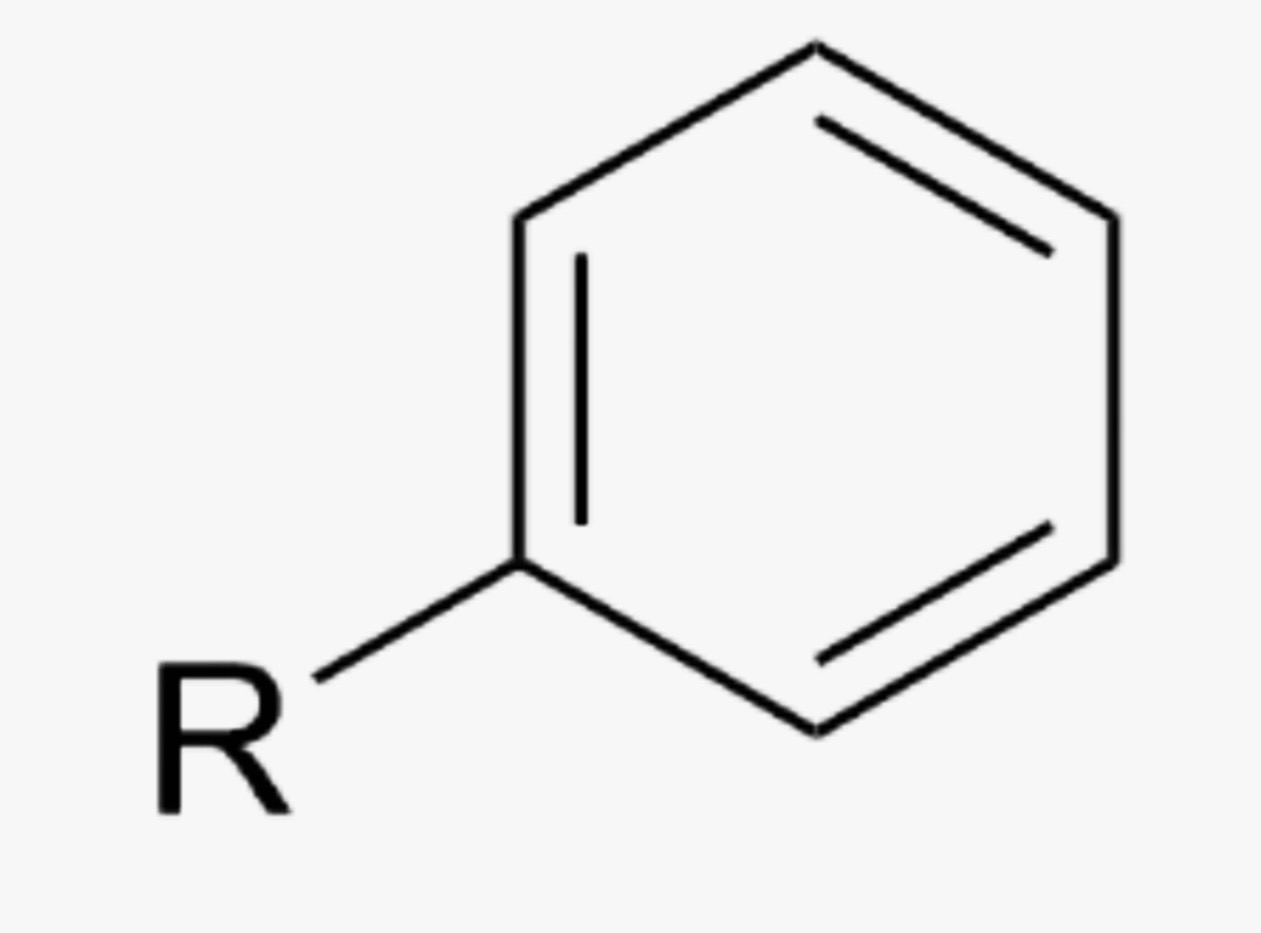
What is the name of this organic functional group?
Arene
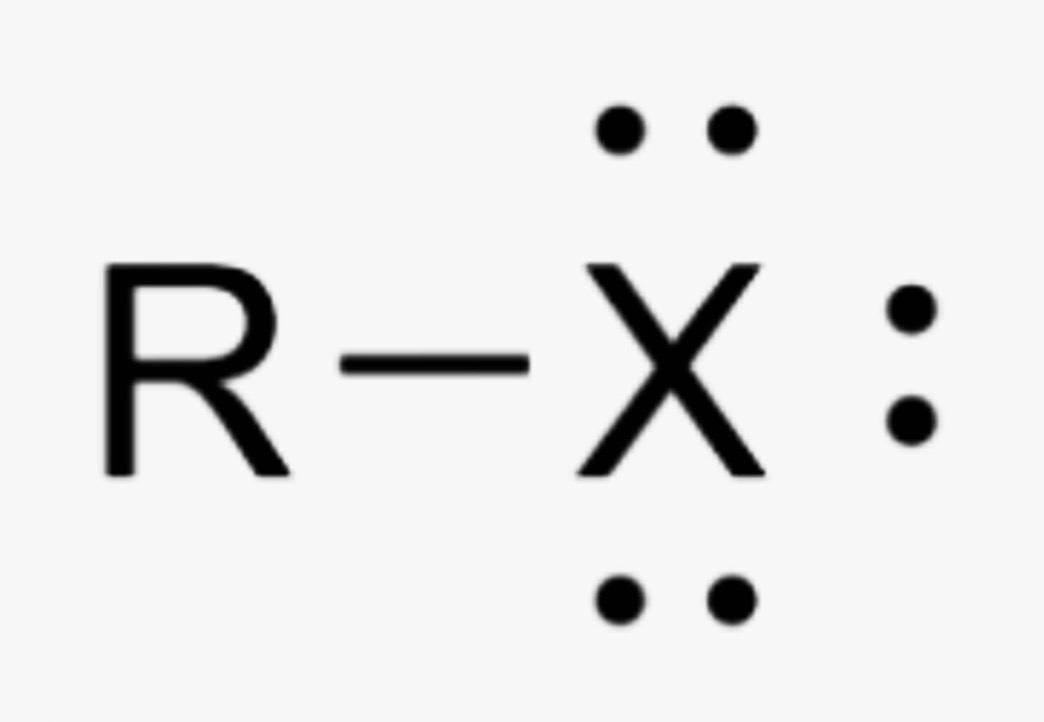
What is the name of this organic functional group?
Alkyl Halide
What elements can be X in an alkyl halide functional group?
F, Cl, Br, I

What is the name of this organic functional group?
Amine
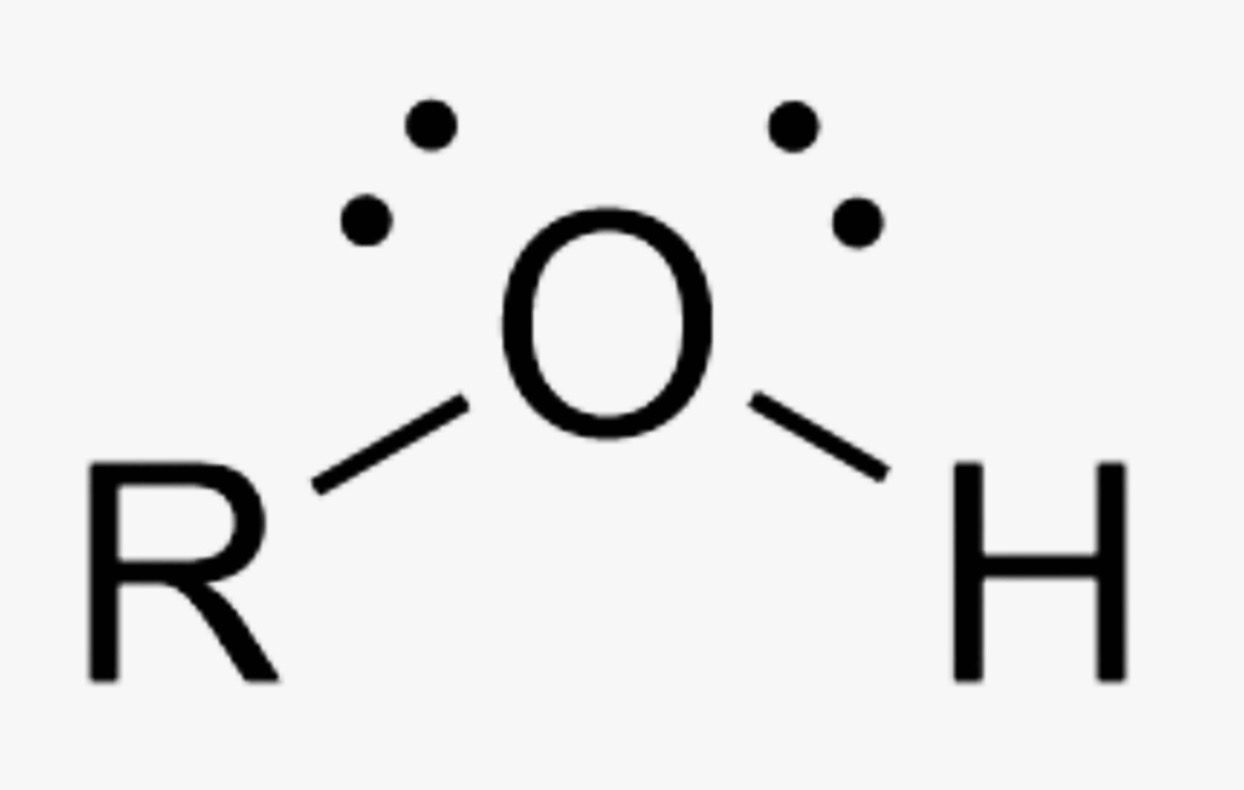
What is the name of this organic functional group?
Alcohol
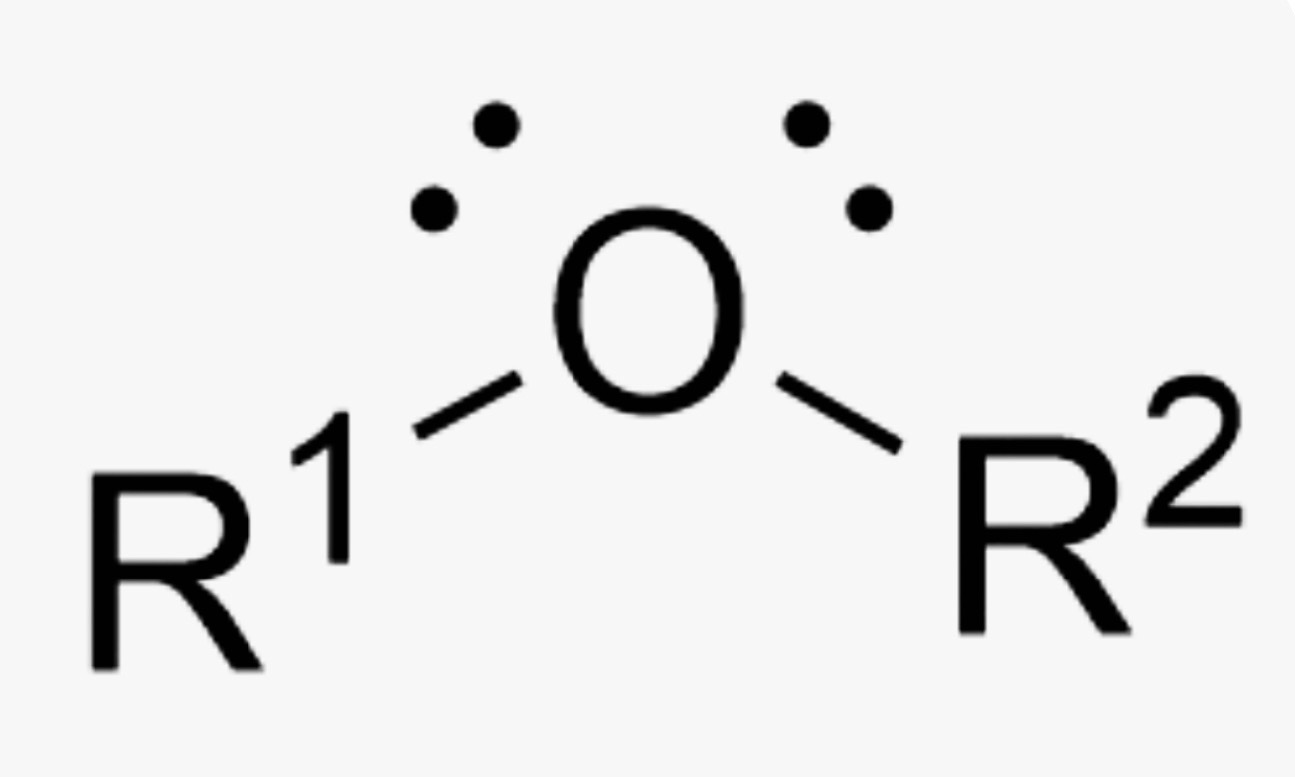
What is the name of this organic functional group?
Ether
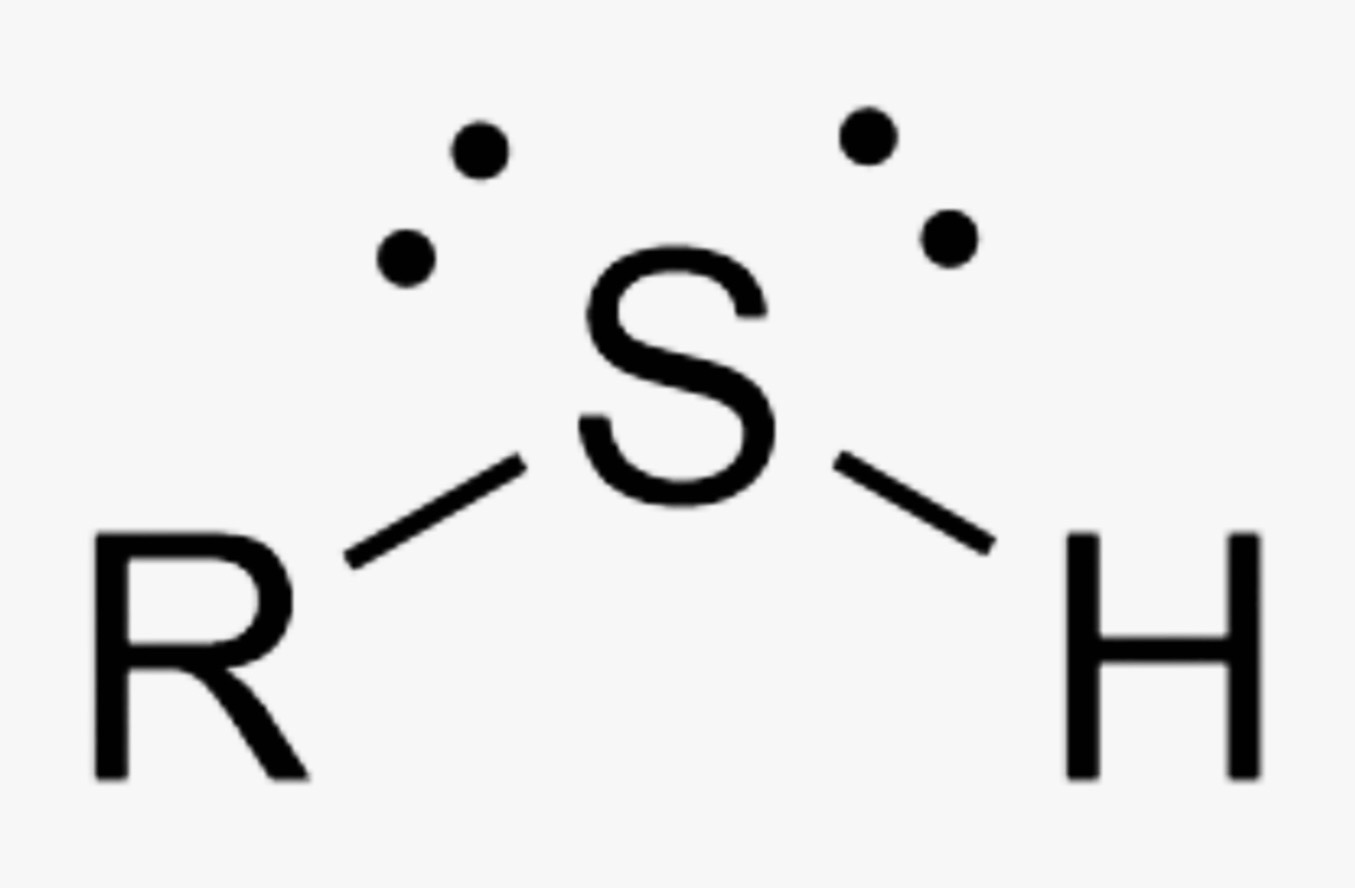
What is the name of this organic functional group?
Thiol
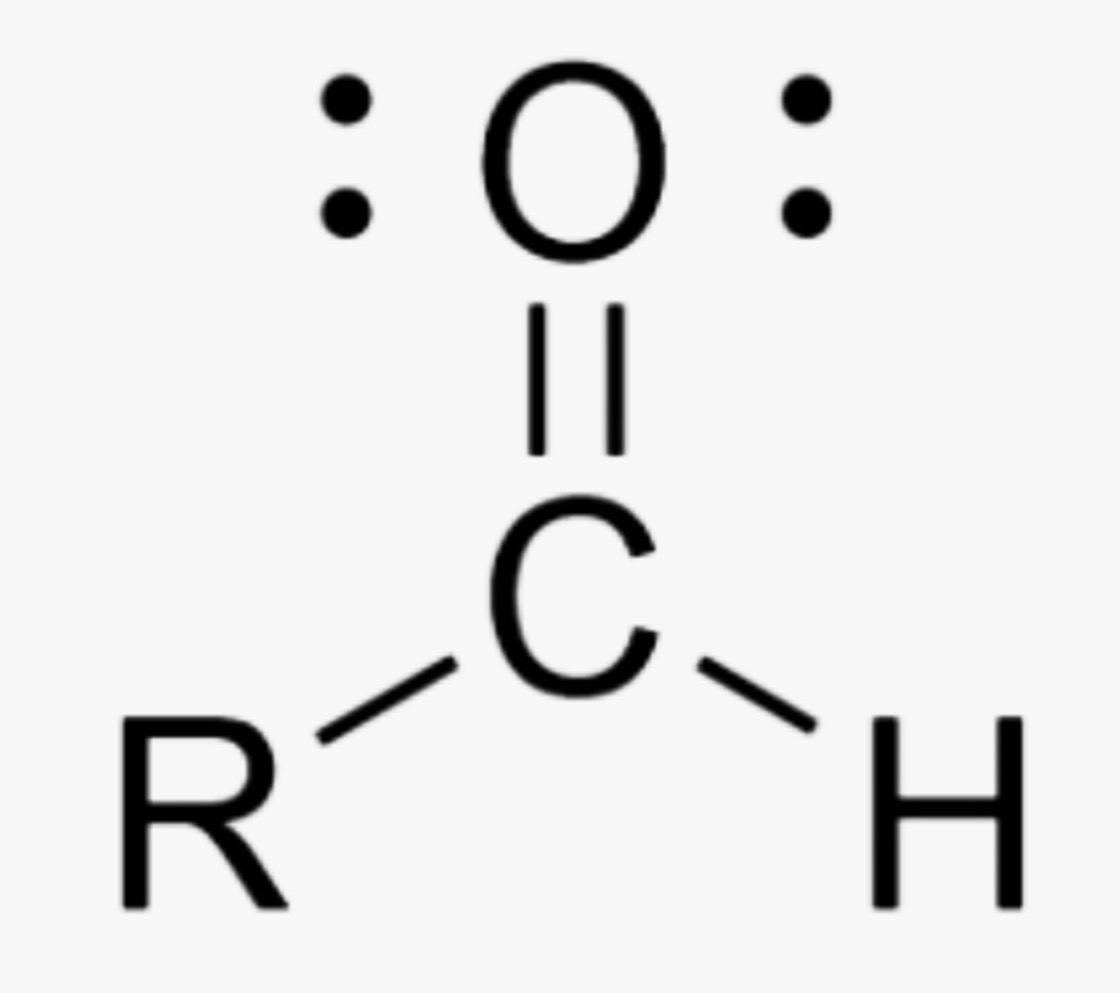
What is the name of this organic functional group?
Aldehyde
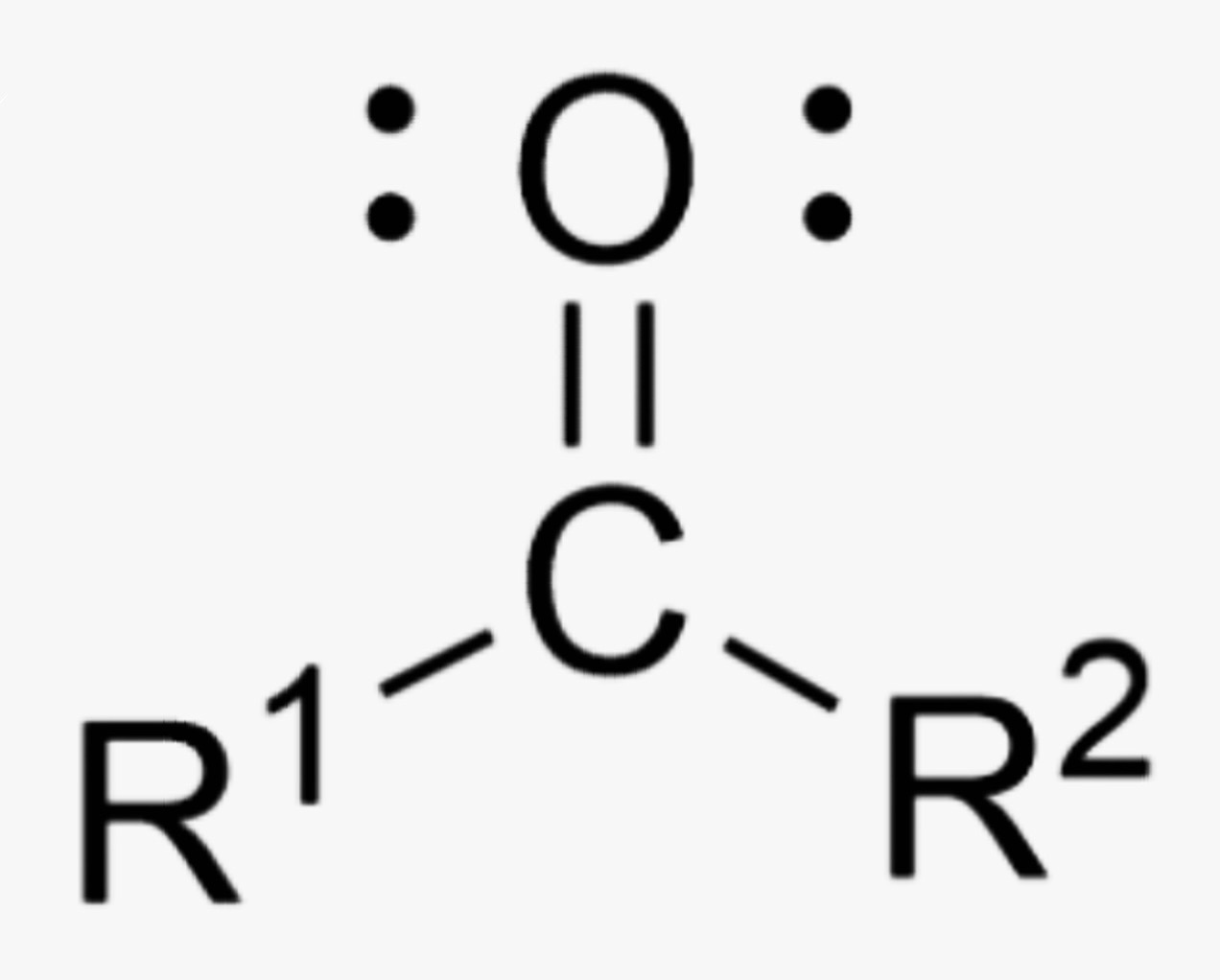
What is the name of this organic functional group?
Ketone
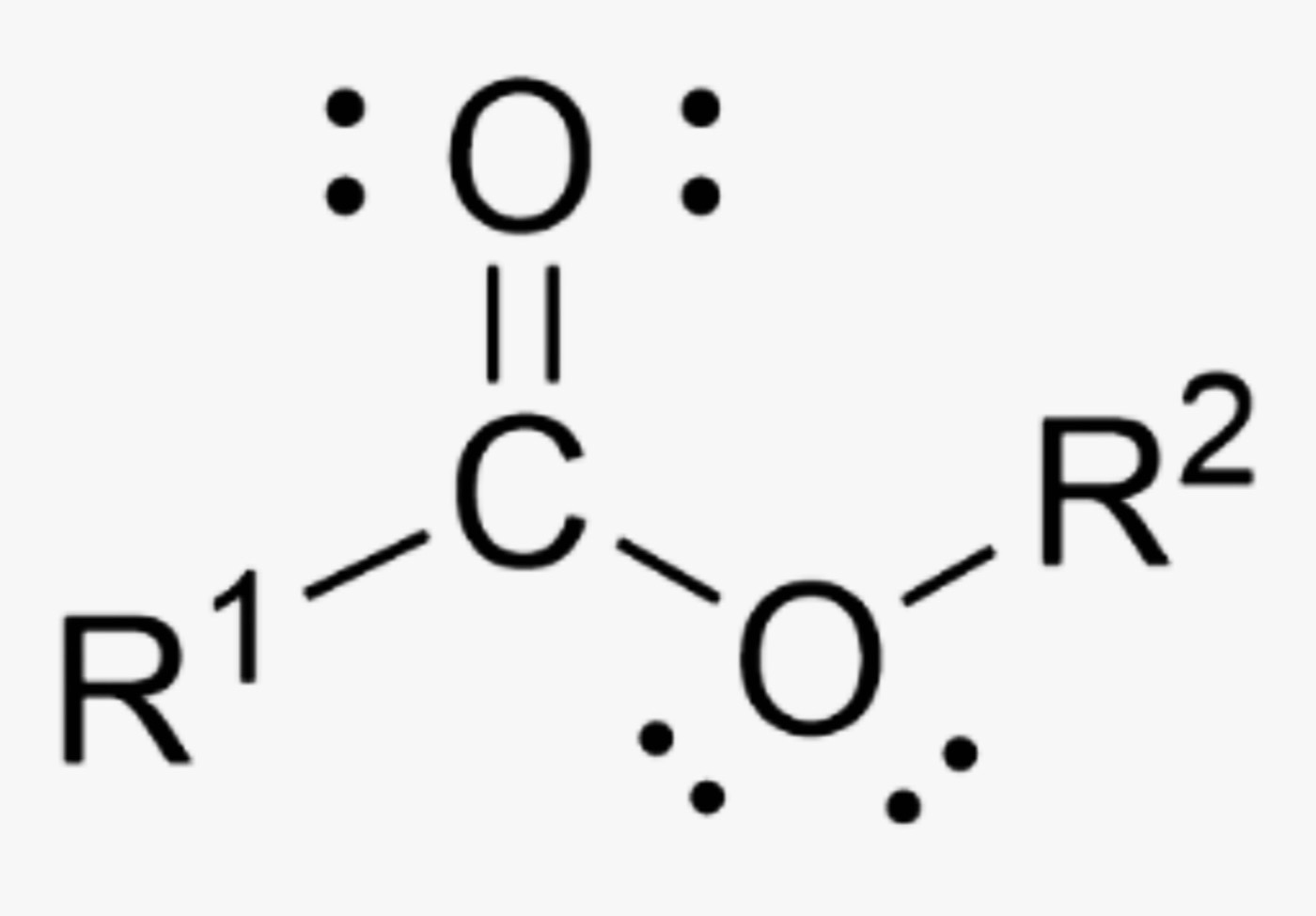
What is the name of this organic functional group?
Ester
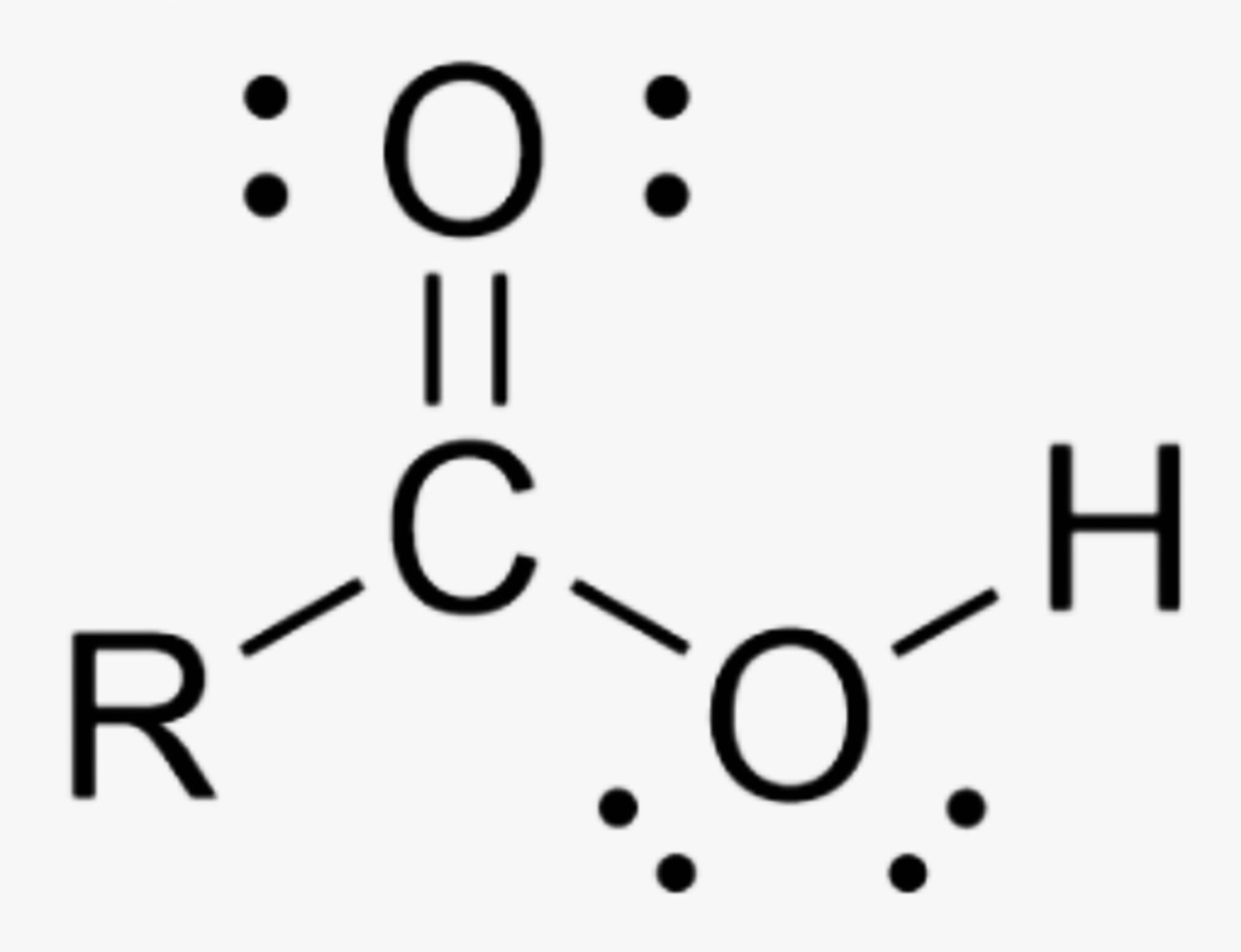
What is the name of this organic functional group?
Carboxylic Acid
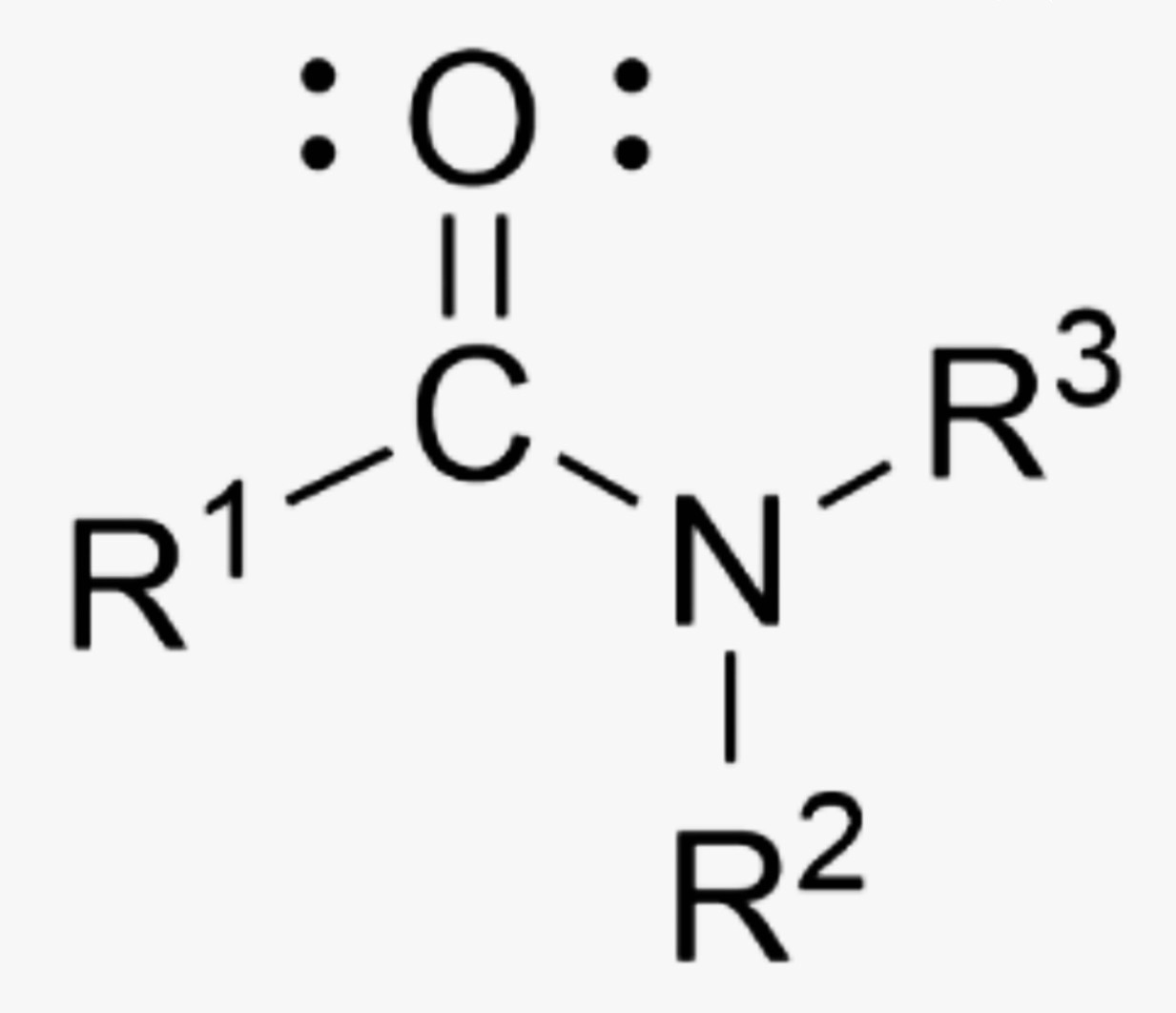
What is the name of this organic functional group?
Amide
What is the naming convention for alkane?
-ane
What is the naming convention for alkene?
-ene
What is the naming convention for alkyne?
-yne
What is the naming convention for arene?
-benzene or -phenyl
What is the naming convention for alkyl halide/haloalkane?
fluoro-, chloro-, bromo-, iodo-
What is the naming convention for amine?
-amine or -amino
What is the naming convention for alcohol?
-ol or hydroxy-
What is the naming convention for ether?
-ether or methoxy-
What is the naming convention for thiol?
-thiol
What is the naming convention for aldehyde?
-al
What is the naming convention for ketone?
-one
What is the naming convention for ester?
-anoate or -ate
What is the naming convention for carboxylic acid?
-oic acid
What is the naming convention for amide?
-amide

What is the name of the organic functional group present in this molecule?
Alkane

What is the name of the organic functional group present in this molecule?
Alkene

What is the name of the organic functional group present in this molecule?
Alkyne

What is the name of the organic functional group present in this molecule?
Arene

What is the name of the organic functional group present in this molecule?
Alkyl halide/Haloalkane

What is the name of the organic functional group present in this molecule?
Amine

What is the name of the organic functional group present in this molecule?
Alcohol

What is the name of the organic functional group present in this molecule?
Ether

What is the name of the organic functional group present in this molecule?
Thiol
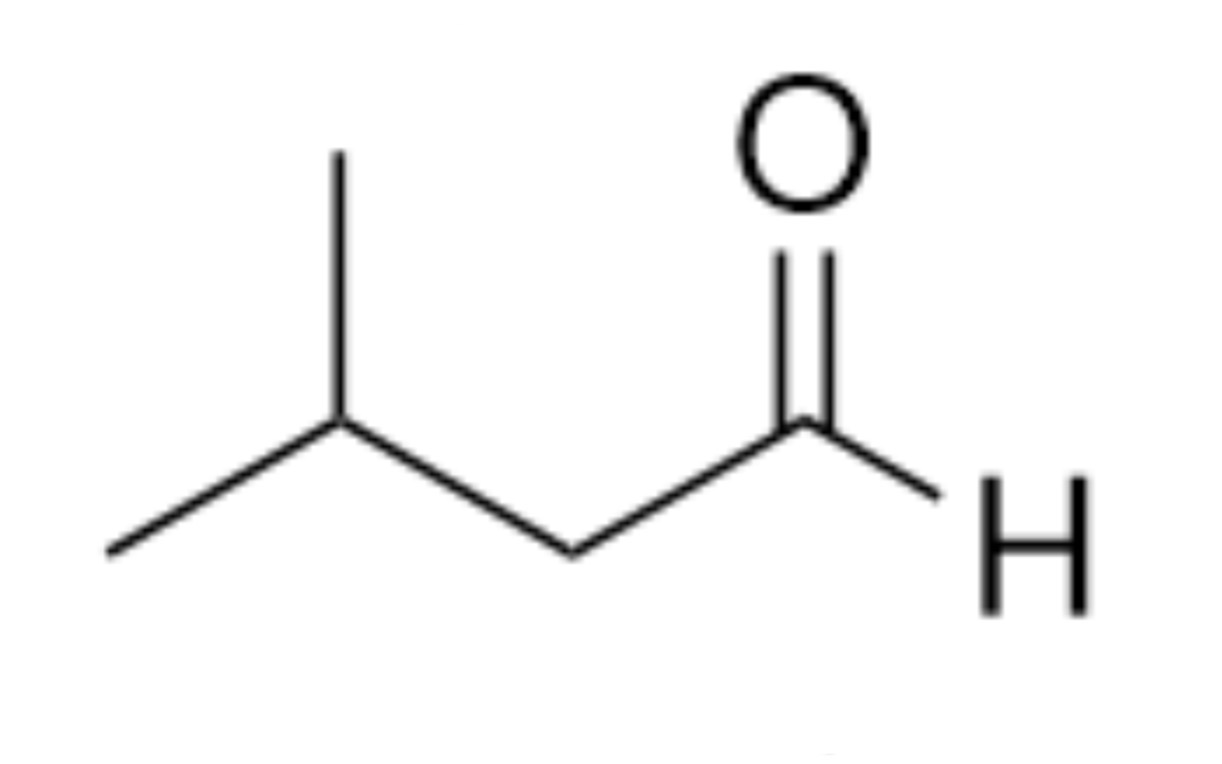
What is the name of the organic functional group present in this molecule?
Aldehyde
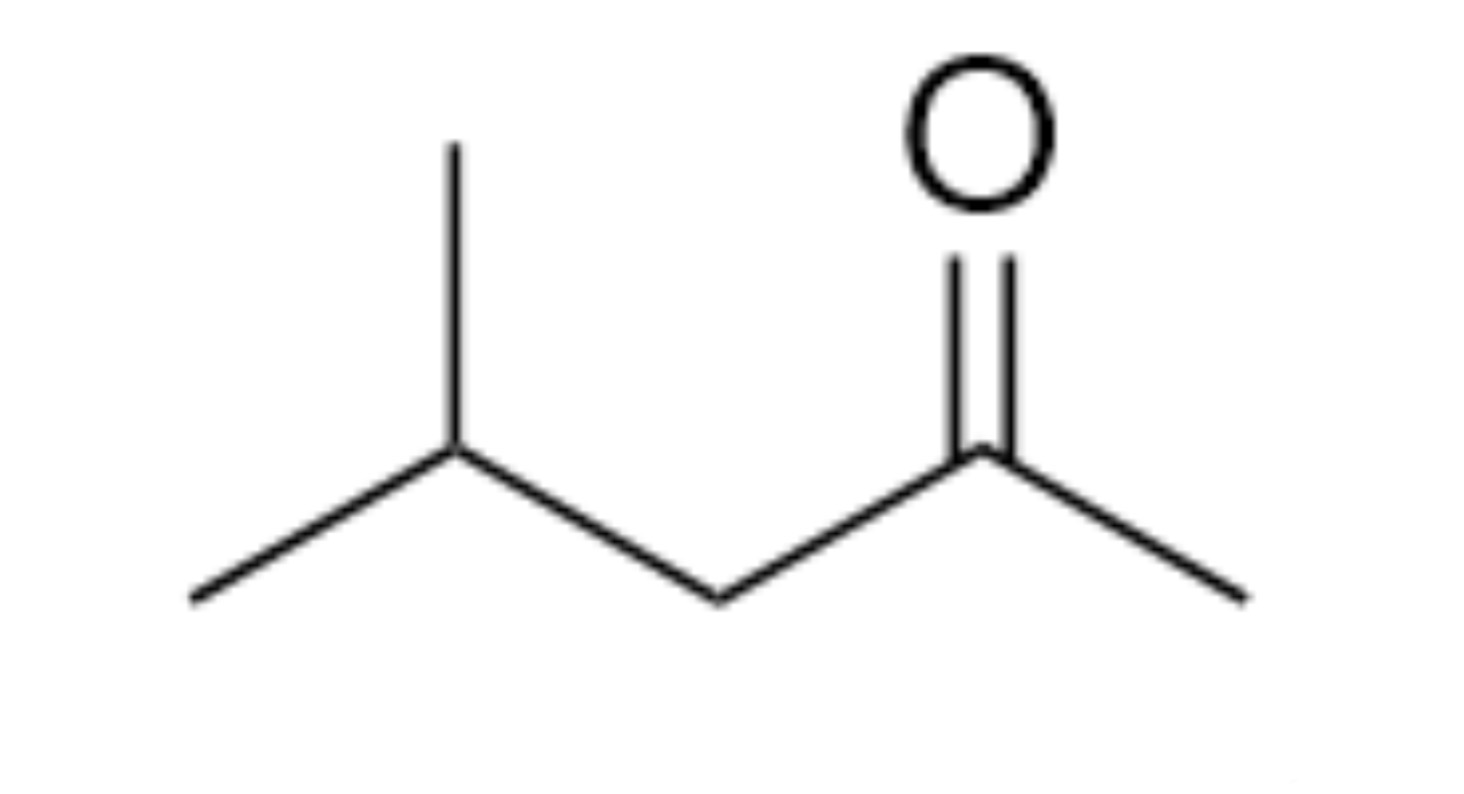
What is the name of the organic functional group present in this molecule?
Ketone
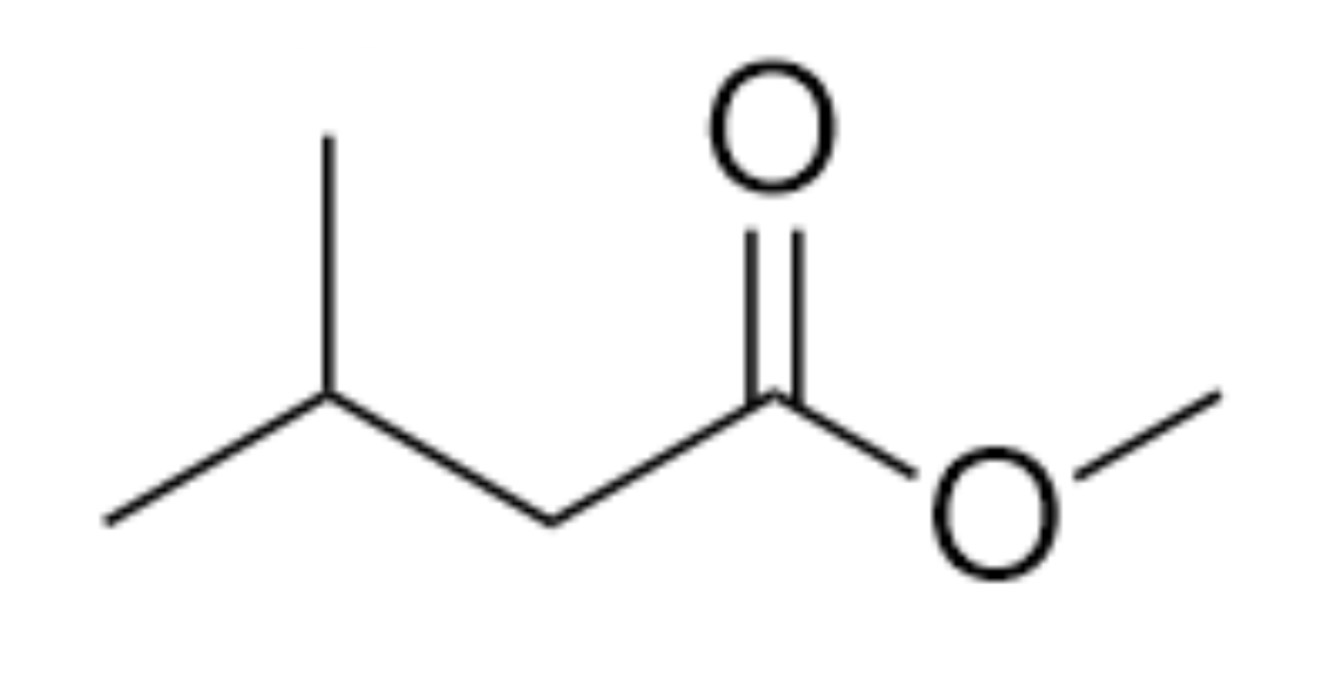
What is the name of the organic functional group present in this molecule?
Ester
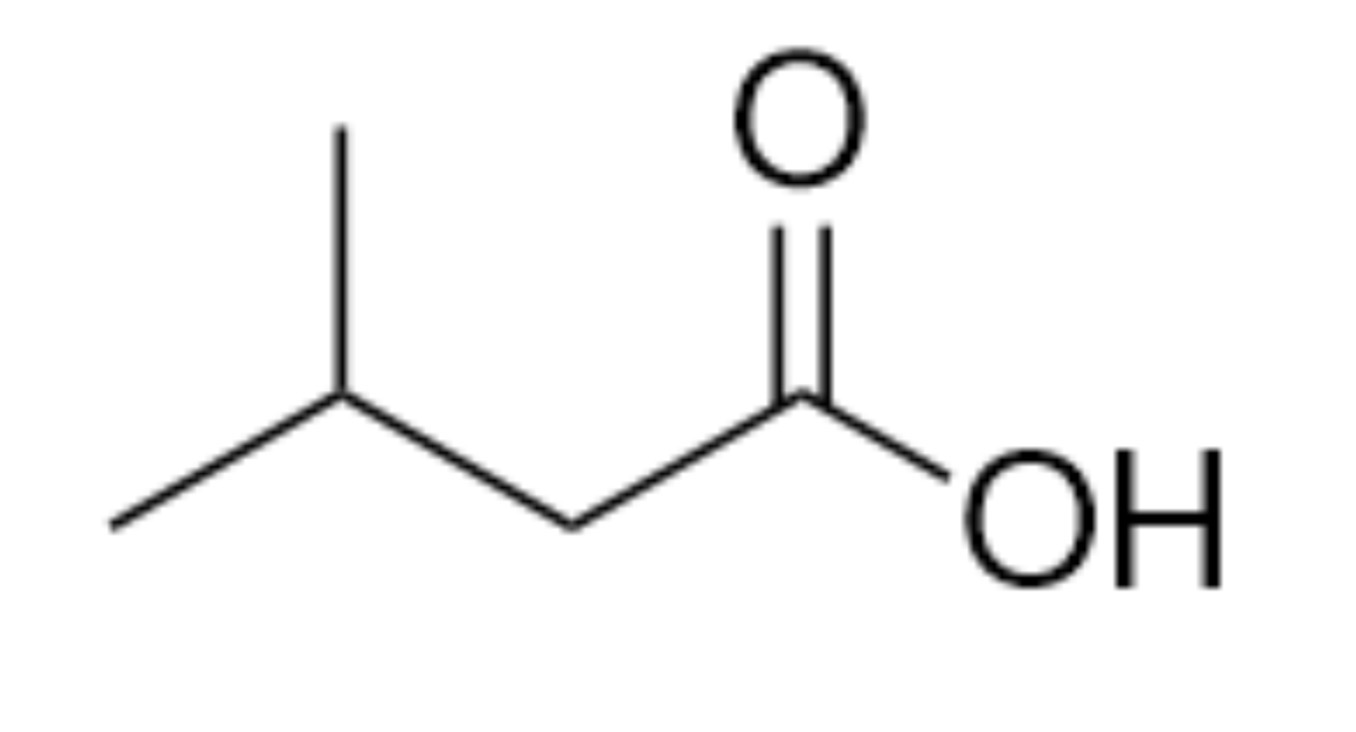
What is the name of the organic functional group present in this molecule?
Carboxylic acid
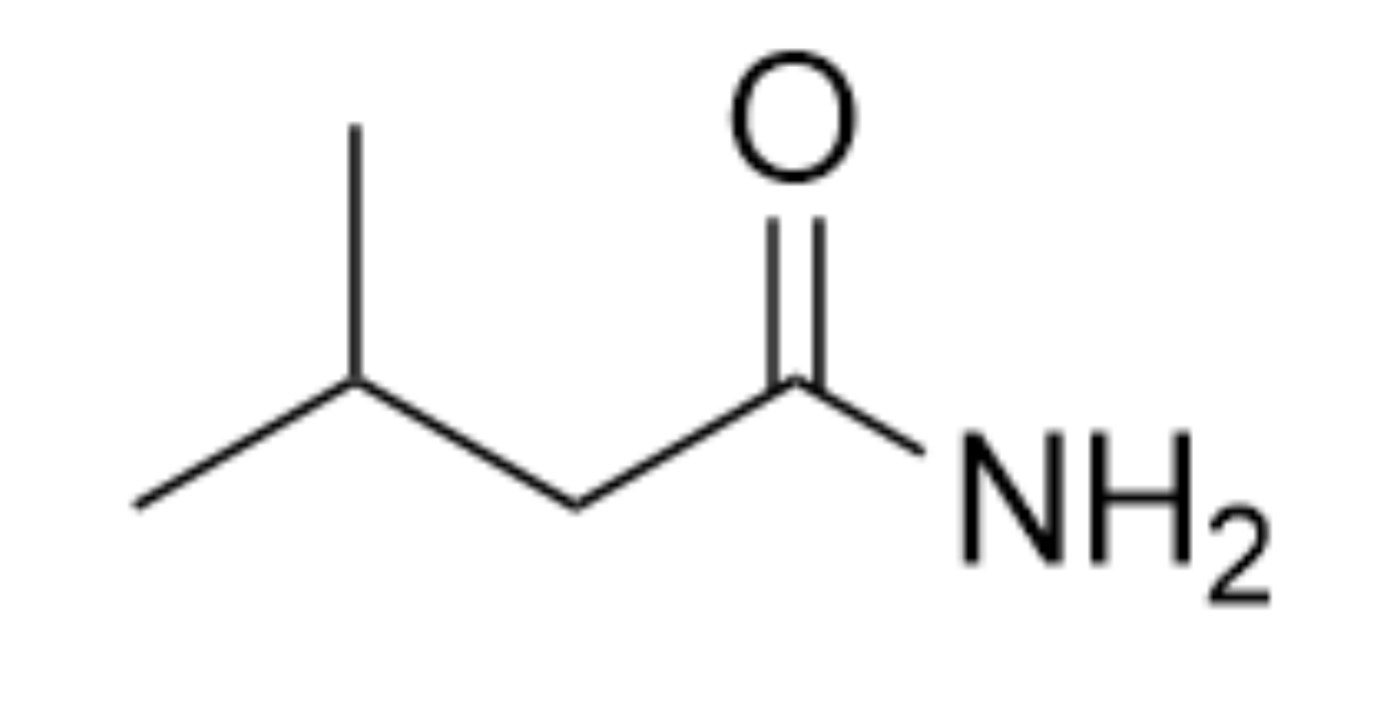
What is the name of the organic functional group present in this molecule?
Amide
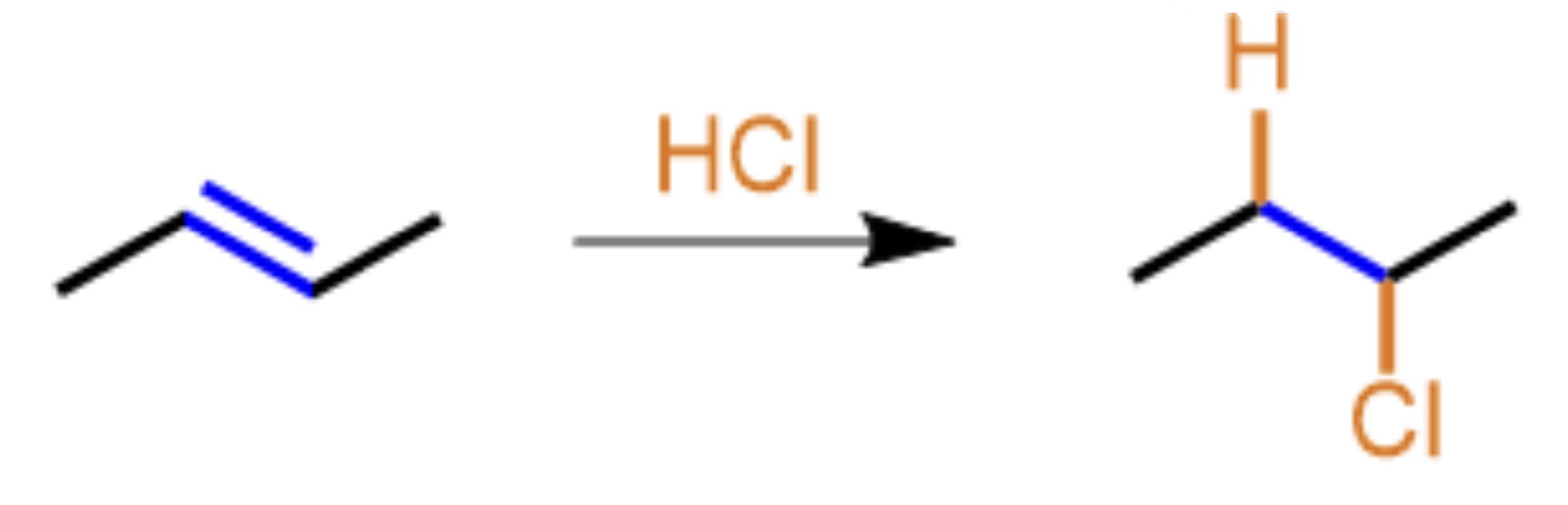
What type of organic reaction mechanism is presented?
Addition, the H and Cl are added on adjacent carbons and the alkene (double bond) becomes an alkane (single bond)
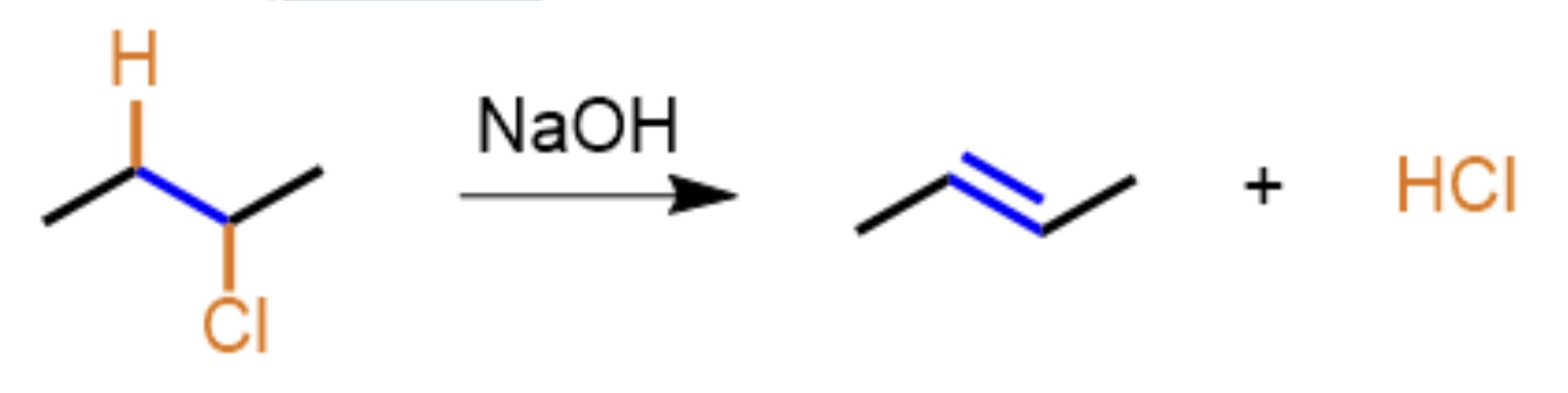
What type of organic reaction mechanism is presented?
Elimination, H and Cl are removed by NaOH in order to form HCLl and an alkene (double bond)
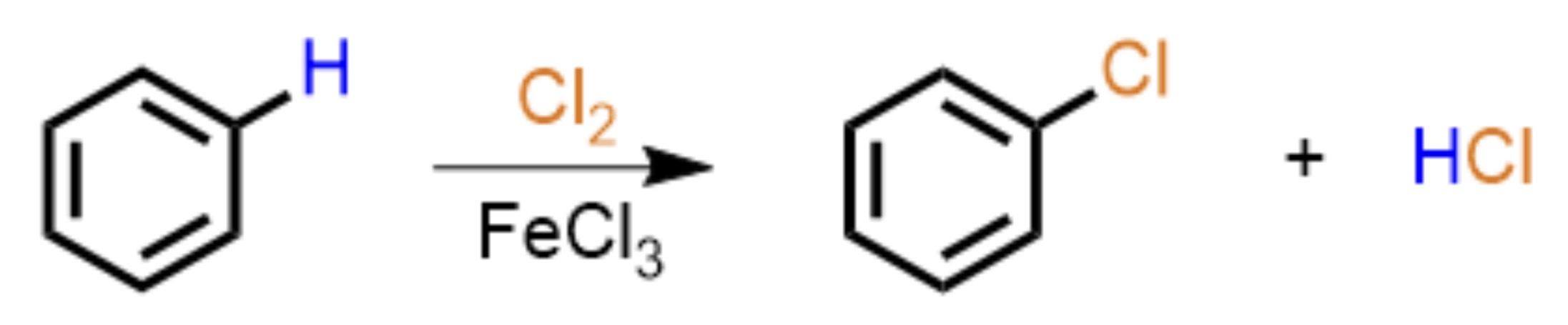
What type of organic reaction mechanism is presented?
Substitution, H is replaced with Cl by FeCl3 and HCl is formed as a byproduct.
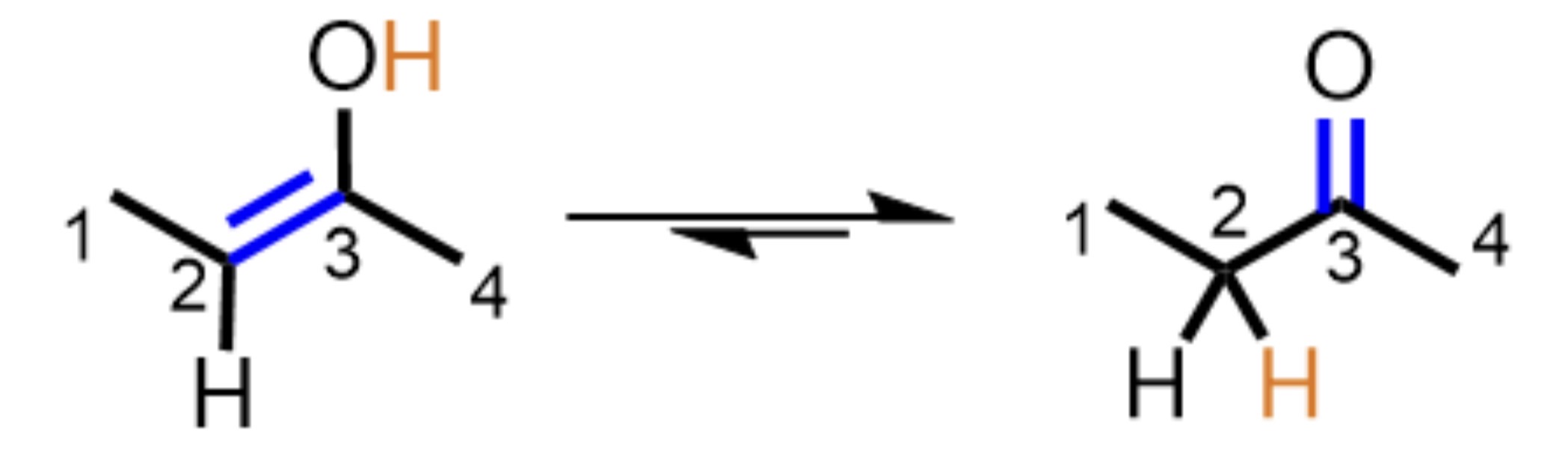
What type of organic reaction mechanism is presented?
Rearrangement, the H is moved from the oxygen to a carbon while the double bond is shifted from C=C to C=O.
What do most biopolymer addition reactions require?
π bonds (donates electrons to make single bonds that continue the chain reaction, making a polymer.)