Chapter 14 CHM2046 UF
1/109
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
110 Terms
Which periodic patterns generally decrease down a period and increase across a group?
Ionization Energy, Electron Affinity (negatively), Electronegativity, Covalent Character, Acidity of Oxides, and Nonmetallic Character.
Which periodic patterns generally increase down a period and decrease across a group?
Atomic radius (size), Metallic Character, Ionic Character, and Basicity of Oxides.
Hydration Energy
The energy released when ions are dissolved in water. Proportional to Lattice Energy.
Where on the periodic table can you find the most reactive elements?
At each end of the periodic table, excluding noble gases. This would be groups 1A and 7A.
For a metallic element that acts as a strong reducing agent, what conclusion can be made about it's Ionization Energy?
It is likely low
What is the most abundant element in the universe?
Hydrogen
What two charges can Hydrogen take on? In what groups will hydrogen be with these respective charges?
1+ in group 1A and 1- in group 7A.
Covalent Hydride
A compound consisting of hydrogen and another element, ex. CH4 or NH3. Formed from acidic hydrogens with a 1+ charge (H+).
Ionic Hydride
A compound consisting Hydrogen bonded to any alkali metal or the alkaline earth metals Ca, Sr, or Ba. For example, NaH. They are very reactive to air and water and are formed from basic hydrogens (H-).
From what process is Ammonia created?
The Haber Process
What is the one step of the Haber Process?
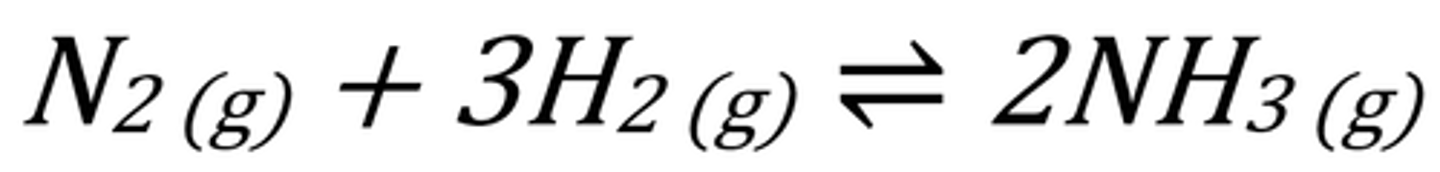
What is the fuel cell reaction?
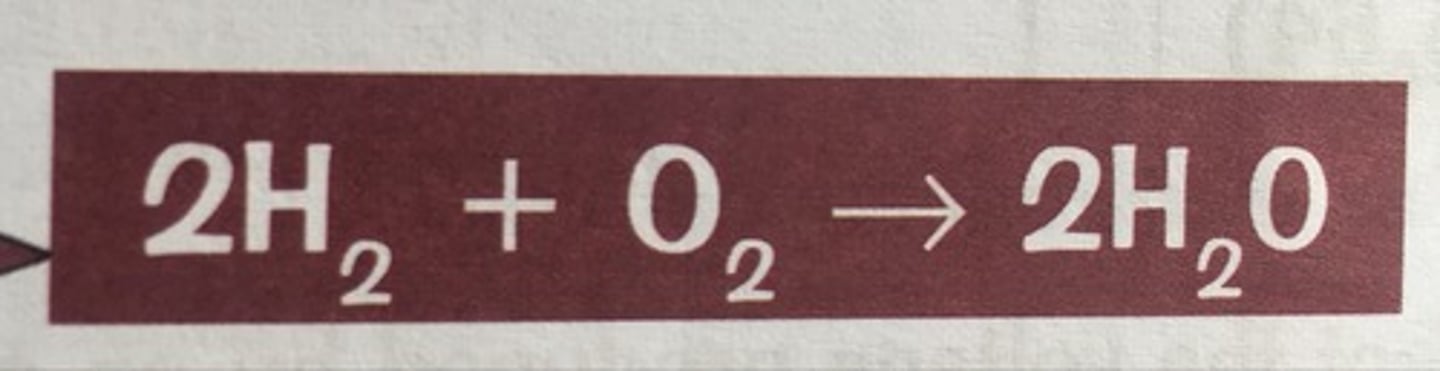
What is the reaction that describes hydrogen gas reducing some transition metal X with a 2+ charge?
X^2+(aq) + H2(g) -> 2H+(aq) + X(s)
What is the reaction that describes the formation of a metal hydride from the cation Y with a charge of 2+?
Y(s) + H2(g) -> YH2(s)
What are the three main properties of elements in group 1A?
1. Malleable metals
2. Reacts spontaneously in water to produce H2 gas
3. Strong Reducing Agents
Describe the Lithium Irregularity.
Lithium prefers to make polar covalent bonds and is highly reactive. Lithium salts are less soluble in water than those of Sodium or Potassium because it is more covalent than both of those elements.
How is metallic sodium usually obtained?
Through the electrolysis of molten (non-aqueous) sodium chloride in the "Downs Cell."
What is the relation between reactivity and an element's position on the Reduction Potential Sheet?
The lower an element is on the Reduction Potential table, the better reducing agent it is and the more reactive it is.
What element is more reactive: Sodium or Potassium?
Potassium, but both are highly reactive.
What happens when sodium or potassium metals react with water?
They form their respective hydroxides. For example: Na(s) + H2O(l) -> NaOH + H2.
Describe the reaction between metallic sodium and excess oxygen. (When sodium burns in air)
2Na(s) + O2(g) -> Na2O2(s)
If metallic sodium is only able to react with a limited amount of oxygen, what happens?
N2O2 cannot be formed, and instead, Na2O is formed.
Describe the reaction between metallic potassium and oxygen. (When potassium burns in air)
K(s) +O2(g) -> KO2(s)
What is a Peroxide? What is the peroxide ion?
An oxygen with an oxidation number of -1. The ion is shown in the image.
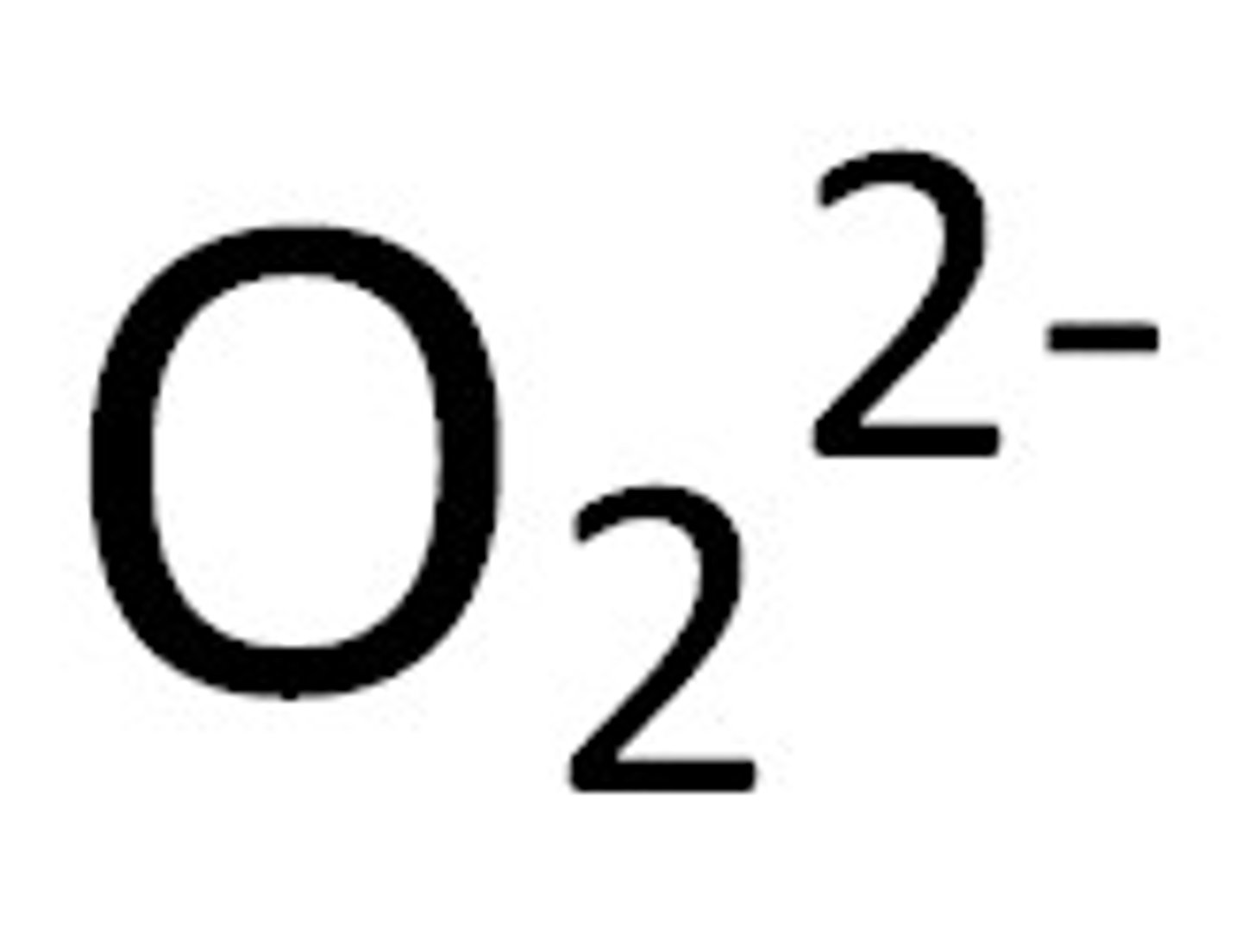
What is a superoxide?
An oxygen with an oxidation number of -1/2.
What is an oxide? What is the oxide ion?
A compound containing oxygen and any other element. In oxides, oxygen has an oxidation number of -2. The oxide ion is shown in the image.

What are the two general properties of group 2A metals?
1. Smaller atomic radii than group 1 due to higher charge.
2. All form 2+ cations and have higher density than group 1 metals.
What can be said about the basicity of group 2A oxides?
Oxides formed by group 2A are strong bases and react with strong acidic oxides to form salts.
Describe the Beryllium Irregularity.
Despite being a metal, Beryllium forms covalent compounds with both low melting points and low electrical conductivity.
Beryllium is an electron deficient element, only requiring _____ electrons to complete its octet.
4
What are the two ways to obtain Magnesium and Calcium?
1. From the Earth's Crust
2. Through electrolysis of their molten compound
Magnesium does not react with water. Instead, it reacts with what to produce what?
Steam; hydrogen gas (H2) and its hydroxide (Mg(OH)2)
Which element is more reactive: Magnesium or Calcium?
Calcium
Calcium, unlike Magnesium, reacts with what to produce what?
Cold water; hydrogen gas (H2) and its hydroxide (Ca(OH)2).
__________ is a compound that improves the quality of soil, plants, and grass.
Lime (CaO)
What is the difference in water solubility between the alkali metals and the alkaline earth metals?
The alkaline earth metals of group 2A are less soluble in water than the alkali metals of group 1A. This is because the metals of group 2A have a higher charge, therefore a higher Lattice Energy, therefore stronger bonds that are not as easy to break.
What is the only element in group 2A that cannot reduce hydrogen gas?
Beryllium
When group 1A and 2A metals form oxides, what can be said about their pH levels?
pH > 7 since they are basic oxides.
What reaction can group 2A metal compounds undergo when subjected to heat? What do these reactions form?
Decomposition Reactions; They form a Group 2A metal-containing oxide and a stable oxide.
What reaction describes the decomposition of Strontium Carbonate?
SrCO3(s) + heat -> SrO(s) + CO2(g)
What factor causes the properties of group 3A elements to deviate from their expected trends?
The presence of d-block electrons.
What element is both a metalloid and in group 3A?
Boron
Boron, like Beryllium, is electron deficient, only requiring _____ electrons to complete its octet.
6
Boron may do two things to complete its octet, which are:
1. Bond with a lone pair of electrons on a nearby atom if the atom is electron rich. I.E., boron acts as a Lewis Acid (Electron Acceptor).
2. Form a Bridge Bond.
Bridge Bond
When two central atoms bond to an intermediate element and use it as a "bridge" instead of the two central atoms connecting to each other directly. Only occurs in the presence of electron poor atoms.
Each bond in a bridge bond has how many electrons?
1 electron
What is the most abundant element in the Earth's crust?
Aluminum
Does the elemental form of aluminum occur in nature?
No
By what process is aluminum formed?
The Hall-Héroult process
Describe the two steps of the Hall-Héroult process.
1. Electrolyze a molten salt bath of cryolite (Na3AlF6) to Alumina (Al2O3).
2. Extract pure aluminum from alumina through the following reaction: 2Al2O3(s) + 3C(s) -> 4Al(s) + 3CO2(g)
Dimers
Molecules formed by joining two smaller, identical molecules.
Aluminum acts like a metal physically, but its halogen-containing compounds exist in the ______ phase as ____________ ______________.
gas; covalent dimers
Describe the reaction of solid alumina with aqueous hydrochloric acid.
Al2O3(s) + 6HCl(aq) -> 2AlCl3(aq) + 3H2O(l)
Describe the reaction of solid alumina with hydroxide and water.
Al2O3(s) + 2OH-(aq) + 3H2O(l) -> 2[Al(OH)4]-
Through the process of formation, [Al(OH)4]- is created. What is this ion called?
Aluminate Ion
What is unique about aluminum oxides?
They are amphoteric, meaning they can act as both an acid or a base.
What three pairs of elements have a diagonal relationship?
1. Lithium and Magnesium
2. Beryllium and Aluminum
3. Boron and Silicon
What is a diagonal relationship between two elements?
When two elements share similar characteristics and are diagonal from one another on the periodic table.
Explain the diagonal relationship between Li and Mg.
Both have similar size and solubility. Both form Nitrides (XN^3-), hydroxides, and carbonates that decompose easily. Both react with organic compounds to form polar covalent bonds with carbon.
Explain the diagonal relationship between Be and Al.
Their ions have high charge density, resulting in significant covalent character despite being metals. Both have amphoteric oxides with high melting points. Both utilize bridge bonds in their hydrides and chlorides.
Why can Beryllium and Aluminum form amphoteric oxides?
Because they are metals with high covalent character, which allows them to have both basic and acidic characteristics.
What is the difference between the beryllium and aluminum bridge bonding?
Bridge bonding in Be creates polymers while bridge bonding in Al creates dimers.
Polymer
A substance made up of repeating identical units.
Explain the diagonal relationship between Boron and Silicon.
Both act as semi-conductors, form weak acids, and have low melting points.
Why can carbon bond with different elements?
Because it can experience every possible oxidation state for a group 4 element between -4 and +4.
What are the three most important allotropes of carbon?
1. Graphite (C_gr)
2. Diamond (C_d)
3. Buckminsterfullerene (C_60)
Allotrope
Different forms of one element
What carbon allotrope can you expect to find at high pressures? What are the properties of this allotrope?
Diamond; colorless, electrical insulator, very hard
What carbon allotrope can you expect to find at standard conditions? What are the properties of this allotrope?
Graphite; black, conductive, soft
What is the difference in hybridization between the three most prominent carbon allotropes?
In graphite and buckminsterfullerenes, each carbon is bonded to three other carbons and thus has a hybridization of sp2. In diamond, some carbons are sp2 hybridized and others are sp3 hybridized.
What molecular property allows diamond to be so hard?
Diamond is a covalent network solid, meaning it is made up of a very stable arrangement of carbon-carbon single bonds.
Between what two elements is the strongest possible polar covalent bond?
Silicon and Oxygen.
What kind of structure does elemental silicon take on?
A diamond structure
Silicate minerals
minerals that contain silicon and oxygen -the dominant form of matter in the nonliving world.
Orthosilicate Grouping
A tetrahedral arrangement with Si as the central atom single-bonded to four oxygens. Formula: SiO4
What is a cyclical silicate and what is its general formula?
A cyclical silicate is a silicate mineral in a ring structure. Its general formula is (SiO3)_n^2n-.
Silicone Polymers
Manufactured substances consisting of alternating Si and O atoms with two organic groups (OH) also bonded to each Si atom.
What is the difference between Carbon and Silicon's bonds with Oxygen?
Silicon prefers to single bond with oxygen while carbon prefers to double bond with oxygen.
Why is N2 a gas while P4 is a solid at room temperature?
N2 is triple bonded and small while P4 is single bonded and large.
What metal in group 5A can only take on a +3 oxidation state?
Bismuth (Bi)
Paramagnetic
has unpaired electrons. Paramagnetic molecules are also referred to as "odd-electron molecules."
Which nitrogen oxides are paramagnetic?
Nitrogen Monoxide (NO) and Nitrogen Dioxide (NO2, in smog)
Which nitrogen oxide is a volatile solid that consists of NO_2^+ and NO_3^- molecules?
Dinitrogen Pentoxide (N2O5)
Which nitrogen oxide dissociates to NO2 and may be a colorless to yellow liquid?
Dinitrogen Tetroxide (N2O4)
Which nitrogen oxide is referred to as laughing gas?
Dinitrogen Monoxide (N2O)
By what process is Nitric Acid (HNO3) formed?
The Ostwald Process
Describe the 4 steps of the Ostwald process.
1. N2 + 3H2 -> 2NH3 + heat (Haber Process)
2. (with catalyst): 4NH3 + 5O2 -> 4NO + 6H2O
2. (without catalyst): 4NH3 + 3O2 -> 2N2 + 6H2O (cycles back to repeat haber process)
3. 2NO + O2 -> 2NO2
4. 3NO2 + 3H2O -> 2NO3^- + 2H3O+ + NO
In dilute acid, you can make _________ from Nitrate.
Nitrogen Monoxide (NO)
In concentrated acid, you can make _______ from Nitrate.
Nitrite (NO2)
Explain the two allotropes of Phosphorous
1. White Phosphorous: A tetrahedral compound that has four phosphorous elements single bonded to each other in a tetrahedral type of shape.
2. Red Phosphorous: A polymer of white phosphorous formed when it breaks.
What is phosphine and how is it formed?
PH3; formed via alkaline hydrolysis
What is the reaction that has Phosphorous Trichloride reacting to form phosphoric acid?
PCl3 + 3H2O -> H3PO3 + 3HCl
When an element in a compound forms an acid or base, what must it retain?
It's oxidation number
Why is O2 a gas and S8 is a solid at room temperature?
O2 is double bonded and small while S8 is single bonded and large.
Is SF6 a reducing agent or oxidizing agent, and why can it only be one?
Oxidizing agent; it cannot be a reducing agent because sulfur has the maximum oxidation state it can have and thus cannot lose any electrons to get a higher one.
What form of oxygen is paramagnetic?
Superoxide
Disproportionation Reaction
an element in one oxidation state is simultaneously oxidized and reduced
At room temperature, sulfur appears in what molecular form?
S8 rings
What is the most highly used acid in the world?
H2SO4
What process produces sulfuric acid?
The Contact Process