Chapter 40 - Animal Water Balance
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
Importance of Water and Electrolyte Balance
All biochemical reactions occur in aqueous environments
Electrolytes are required for nerve signaling, muscle contraction, and enzyme function
Na+, K+, Cl-, Ca2+
Imbalances can disrupt critical physiological functions; maintenance of homeostasis ensures proper function
Water balance relates to excretion (wastes need to be dissolved in water)
Osmolarity
Concentration of solutes in a solution that determines movement of water across membranes via osmosis
Measured in osmoles per liter
Higher osmolarity = lower concentration of free water molecules
When separated by selectively permeable membrane: water moves from low → high regions of osmolarity
Lower osmolarity = lower solute concentration
Higher osmolarity = higher solute concentration (more opportunity for interaction)
Hyperosmotic, Hypoosmotic, Isosmotic
Determine direction of water movement across membranes
Hyperosmotic: a solution with a higher solute concentration relative to another solution
Hypoosmotic: a solution with a lower solute concentration relative to another solution
Isosmotic: two solutions have equal solute concentrations
Osmosis
Passive movement of water across a selectively permeable membrane
Moves from regions of low osmolarity (lots of free water) → high osmolarity (little free water/lots of solutes)
Movement occurs because solutes bind water molecules, reducing free water availability
Water is not attracted to solutes; movement is driven by differences in free water concentration
Passive v. Active Transport
Passive transport moves solutes down their concentration or electrochemical gradient without energy input
Simple diffusion: small, nonpolar molecules (O2 and CO2)
Facilitated diffusion: uses membrane proteins to move larger/charged molecules
Active transport moves solutes against their gradient using energy (ATP or ion gradients); either primary or secondary
Primary vs. Secondary Active Transport
Primary: uses ATP directly to move ions against their gradient (e.g., Na+/K+ ATPase)
Creates electrochemical gradients across membranes
Secondary = uses these gradients to move other solutes
Efficient transport of multiple substances with minimal ATP use
Symporters move solutes in the same direction; antiporters move them in opposite directions
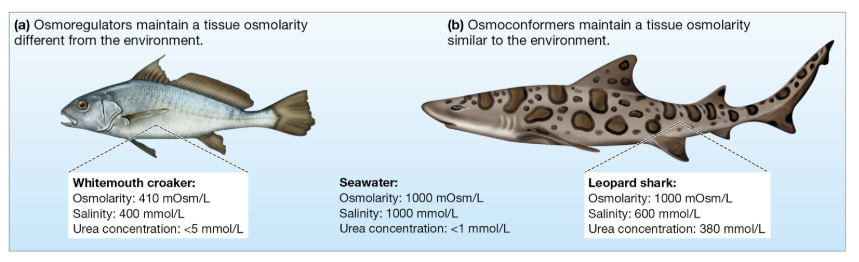
Osmoconformers vs. Osmoregulators
Osmoconformers maintain internal osmolarity equal to their environment
Minimizes osmotic stress
Typically marine invertebrates
Osmoregulators actively control internal osmolarity, keeping it different from the environment
Requires energy but allows survival in variable environments
Typically fishes and terrestrial animals
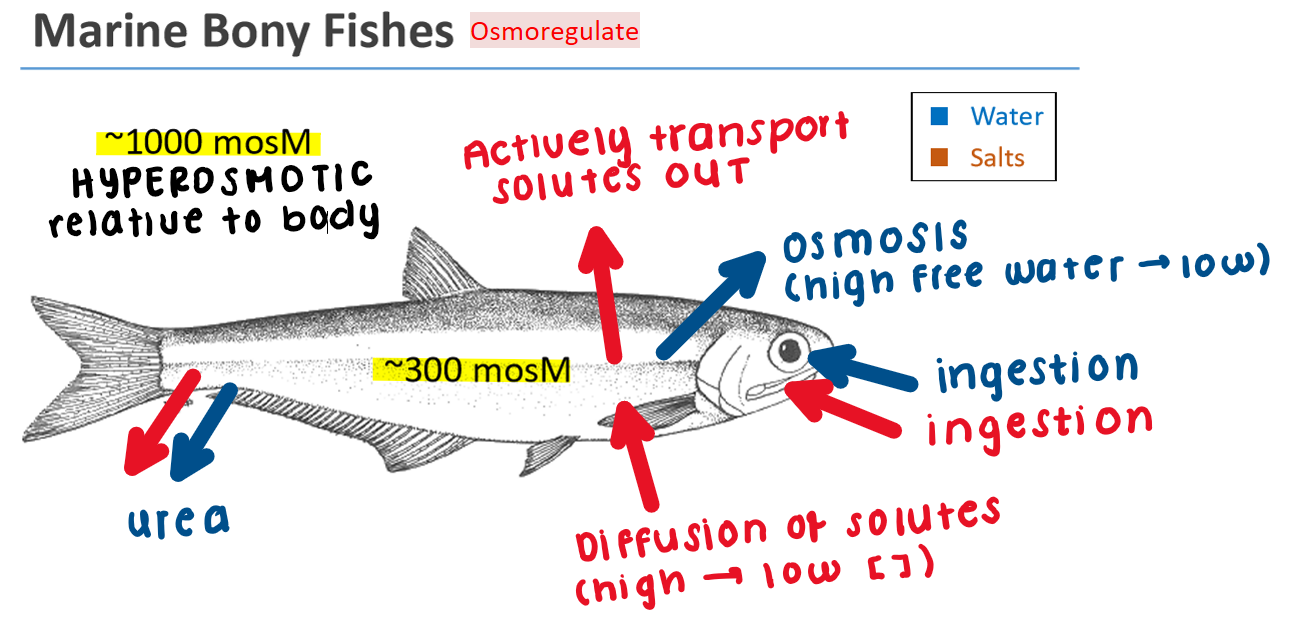
Marine Bony Fish: Osmoregulator
Seawater is hyperosmotic relative to fish body fluids
Body loses water by osmosis (high free water → low) across the gill epithelium
Gain salts by diffusion (higher [ ] in seawater → lower [ ] in the body)
Drink/ingest seawater to replace lost water and salts
Excess salts are actively excreted through specialized chloride cells in gills
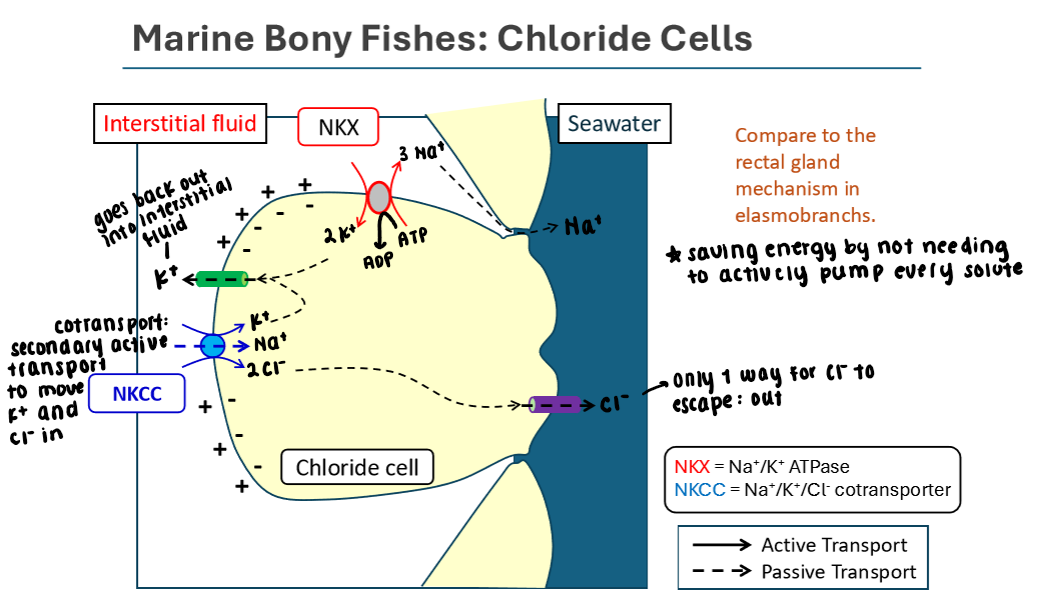
Chloride Cells in Marine Bony Fish
Pumping excess solutes OUT of the body
Uses Na+/K+/ATPase to create an electrochemical gradient
3 Na+ out, 2 K+ in
Na+ gradient (made by sodium potassium pump) allows Na+ to passively move down its gradient into the cell
K+ and Cl- move back into the cell via secondary active transport using the Na+/K+/Cl- cotransporter located b/w cell and interstitial fluid
K+ moves passively back to the interstitial fluid from the chloride cell
Cl- moves passively out of the chloride cell into the surrounding seawater
Saves energy by not needing to actively pump every solute
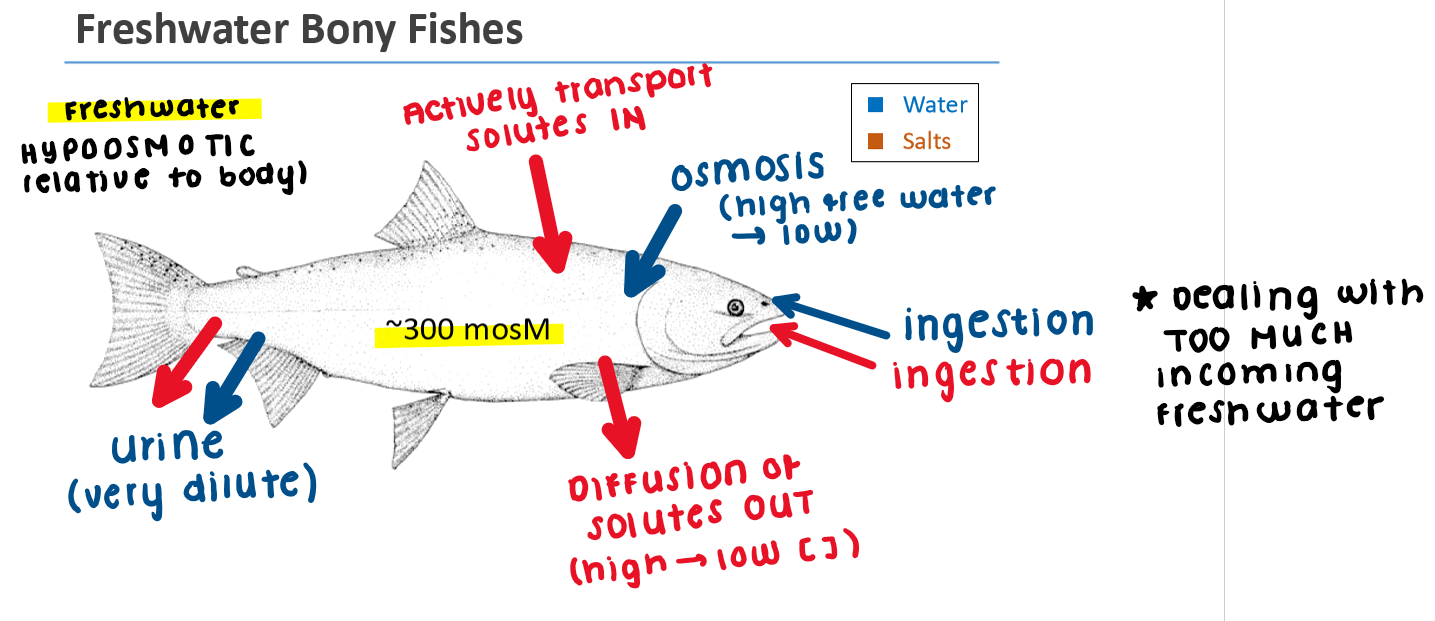
Freshwater Fish: Osmoregulator
Freshwater is hypoosmotic relative to fish body fluids
Body gains water by osmosis (high free water → low) across gill epithelium)
Loses salts by diffusion (higher [ ] in the body → lower [ ] in the freshwater)
Dealing with too much incoming water
Salts are actively transported into the body through chloride cells
Excrete large volumes of dilute urine to remove excess water
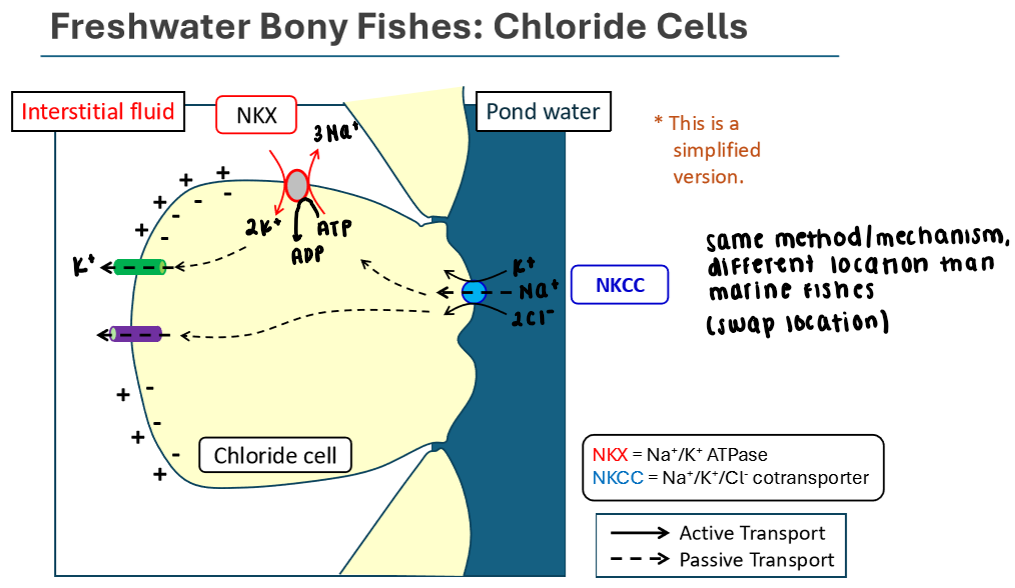
Chloride Cells in Freshwater Bony Fishes
Same mechanism as in marine bony fishes; but the location of the cotransporter differs because we’re actively pumping salts INTO the body
Uses Na+/K+/ATPase to create an electrochemical gradient
3 Na+ out, 2 K+ in
Na+ gradient (made by sodium potassium pump) allows Na+ to passively move down its gradient into the cell
K+ and Cl- move back into the cell via secondary active transport using the Na+/K+/Cl- cotransporter located b/w the cell and pond water
K+ and Cl- move passively to the interstitial fluid from the chloride cell
Sharks (Cartilaginous Fish): Osmoconformer
Body fluids nearly isosmotic to seawater
Maintain high urea concentrations to increase osmolarity without
B/c they’re relatively isosmotic to sea water, there’s not a high concentration of salt anywhere to drive osmosis
Helps reduce water loss by osmosis
Actively excrete excess salts from the rectal gland to maintain ion balance
Must produce proteins to protect cells from urea toxicity
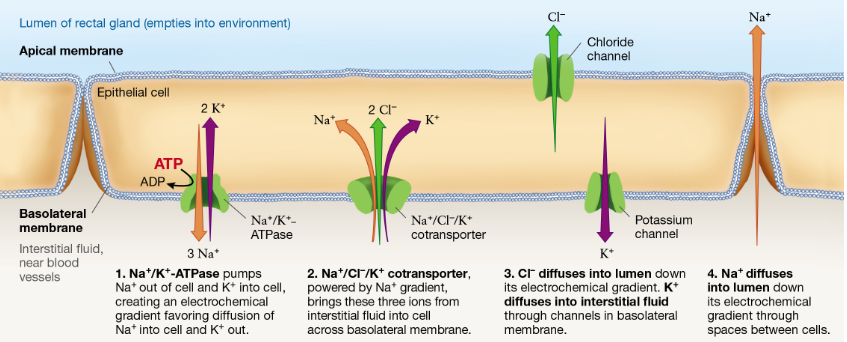
Cartilaginous Fish (Shark): Rectal Gland
Process occurs in the epithelial cell of the rectal gland. Apical membrane = near lumen (empties into environment); basolateral membrane = near interstitial fluid
Na+/K+/ATPase pumps Na+ out of cell into interstitial fluid and K+ into cell from the interstitial fluid, building an electrochemical gradient
Na+/Cl-/K+ transporter that’s power by the Na+ gradient actively moves all three ions from interstitial fluid into the cell (secondary active transport)
Cl- diffuses into the lumen via chloride channel; K+ diffuses to interstitial fluid thru K+ channel
Na+ diffuses into the lumen along its electrochemical gradient
Now salt (Na+ and Cl-) are in the lumen of the rectal gland ready to be excreted
Nitrogenous Wastes: Types and Trade-Offs
Ammonia: highly toxic, requires little energy to produce but large water loss for excretion
Urea: toxic and requires moderate water loss and energy to synthesize
Uric acid: least toxic and conserves water but requires high energy to produce
Different organisms use different waste forms based on environment and water availability
Mammalian Kidney: Structure
Kidneys filter blood and regulate water and electrolyte balance (filtration, reabsorption, excretion)
Blood enters via renal artery and exits via renal vein
Urine flows from kidney → ureter → bladder → urethra → out
Nephrons are functional units of the kidney
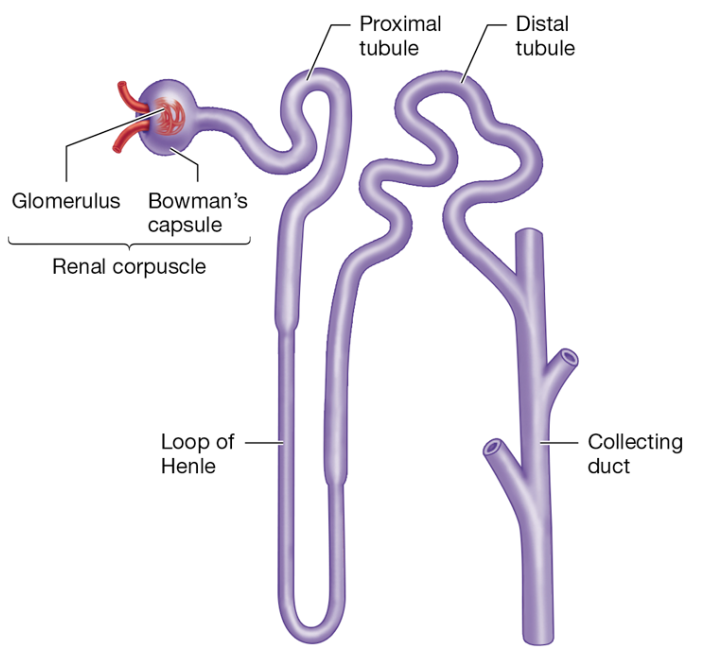
Nephron Structure
Renal corpuscle (glomerulus + Bowman's capsule) filters blood to form the pre-urine (filtrate)
Proximal tubule uses Na+/K+-ATPase and cotransporters to reabsorb nutrients, ions, and water into the blood
Loop of Henle establishes osmotic gradient in the surrounding interstitial fluid
Distal tubule reabsorbs ions and water from the filtrate based on body needs
Hormone (aldosterone) regulated transport
Collecting duct regulates water reabsorption to maintain homeostasis; influences urine concentration
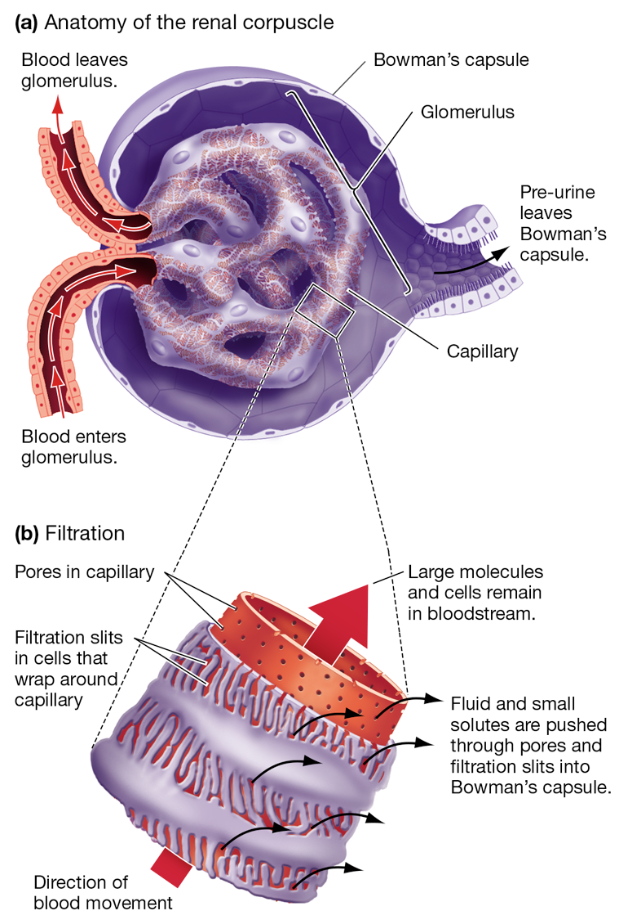
Filtration in Renal Corpuscle
Blood enters glomerulus under pressure
Small molecules (water, ions, glucose, urea) pass through pores and filtration slits into Bowman's capsule
Large molecules (proteins, cells) remain in bloodstream
Creates initial filtrate/pre-urine that leaves Bowman’s capsule
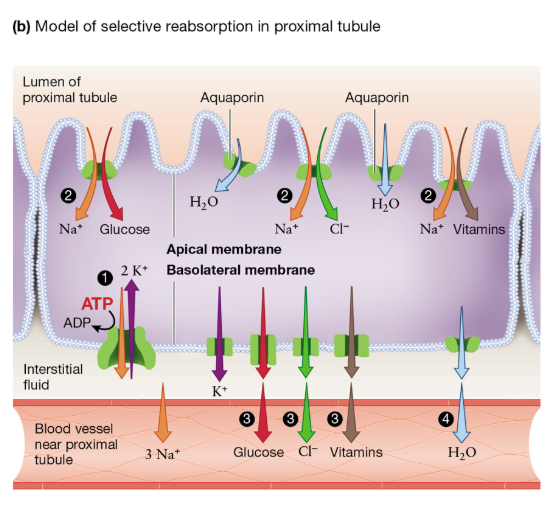
Proximal Tubule: Reabsorption Mechanisms
Epithelial cells contain microvilli to increase surface area for transport
Na+/K+ ATPase creates an electrochemical gradient that favors Na+ entry in from the lumen
Na+-dependent cotransporters in the apical membrane use that Na+ gradient to remove ions and nutrients (Cl-, glucose, vitamins) from the filtrate that’s in the lumen
Those solutes (glucose, Cl-, vitamins) move from the cell → interstitial fluid → nearby blood vessels
Water moves into those blood vessels via osmosis (following the movement of solutes)
Recovers valuable nutrients and prevents loss
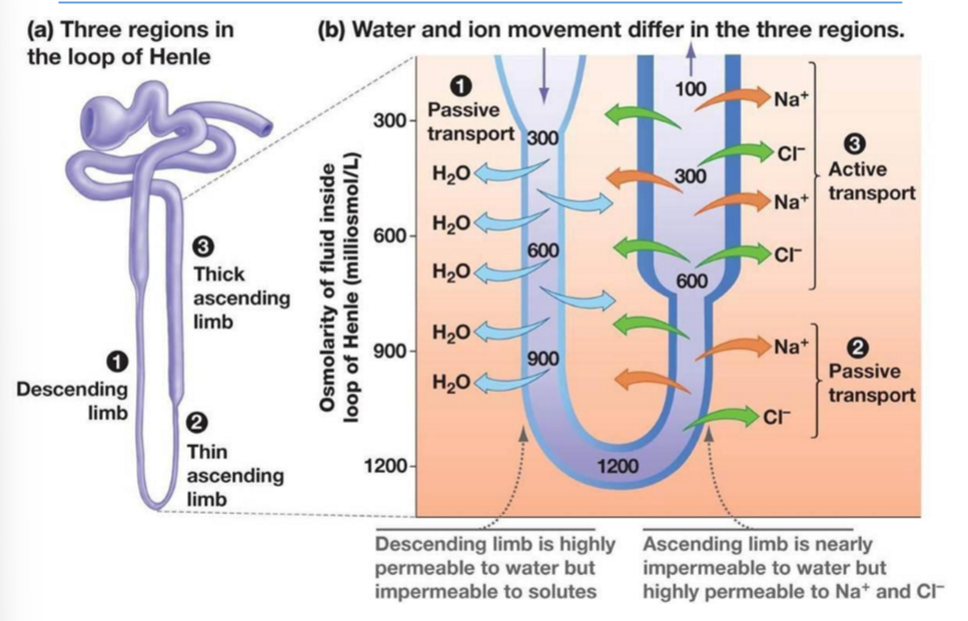
Loop of Henle: Countercurrent
Descending limb is permeable to water only; there’s is an OUTFLOW of water across that epithelium by osmosis
Ascending limb is nearly impermeable to water; it moves Na+ and Cl- out to the interstitial fluid
Thin ascending limb = passive
Thick ascending limb = active
Movement of NaCl from ascending limb raises interstitial osmolarity (there’s more solutes in the interstitial fluid, which helps pull water out from the descending limb)
Helps form concentrated filtrate
Countercurrent flow enhances gradient efficiency
Distal Tubule: Regulation of Ions
Reabsorbs Na+ and Cl- based on body needs
Aldosterone increases Na+ reabsorption and K+ secretion
Helps regulate blood pressure and electrolyte balance
Also plays a role in pH regulation via ion exchange
Adjusts composition of filtrate before final processing
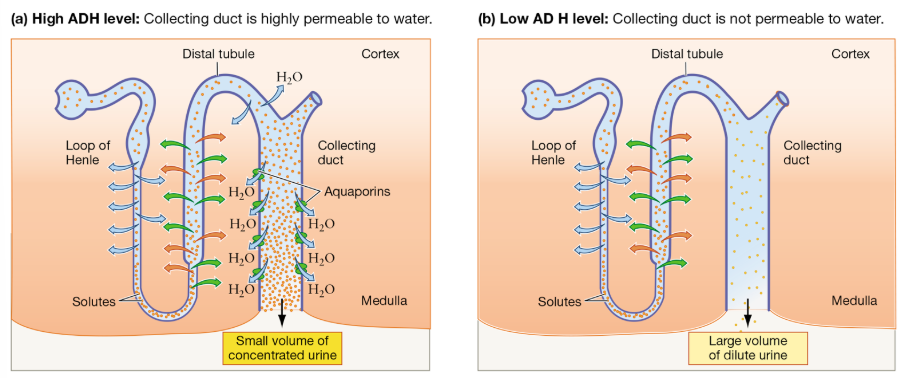
Collecting Duct: Final Water Balance Control
Water reabsorption depends on presence of ADH
When permeable, water exits filtrate into high osmolarity (lots of solutes) medulla
Produces concentrated urine when dehydrated
Low ADH leads to dilute urine
ADH: Role in Water Balance
ADH is released in response to dehydration or increased blood osmolarity
High ADH → small volume of concentrated urine (b/c you’re dehydrated and need to reabsorb water)
Low ADH → large volume of dilute urine
It triggers insertion of aquaporins in collecting duct cells, increasing water permeability for reabsorption
ADH also increases urea permeability, strengthening osmotic gradient
Negative Feedback in Osmoregulation
Sensor detects change in osmolarity or hydration status
Integrator (brain) compares to set point
Effector (kidney, hormones) adjusts water/ion balance
Response restores conditions toward set point
Maintains stable internal environment
ADH Blocking
If ADH is blocked, aquaporins are not inserted
Collecting duct remains impermeable to water
Less water is reabsorbed
Results in large volume of dilute urine
Explains effects of alcohol on urination