GCSE Chemistry: Rate, Equilibrium, and Reversible Reactions
1/25
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
26 Terms
Rate of reaction
Amount of product used or amount of product formed over time.
Rate of reaction formula (reactants)
Rate of reaction = amount of reactant used / Time
Rate of reaction formula (products)
Rate of reaction = amount of product formed / Time
Measurement of quantity
Quantity of reactant or product can be measured by mass in grams or by a volume in cm³.
Units of rate of reaction
Units may be given as g/s or cm³/s.
Moles in rate of reaction
Quantity of reactants can be in terms of moles, with units for rate of reaction in mol/s.
Graphical rate of reaction
To find the rate of reaction graphically, draw tangents to curves and use the slope of the tangent.
Factors affecting reaction rates
Concentration, pressure, surface area, temperature, catalysts.
Collision theory
Chemical reactions can occur only when reacting particles collide with each other and with sufficient energy.
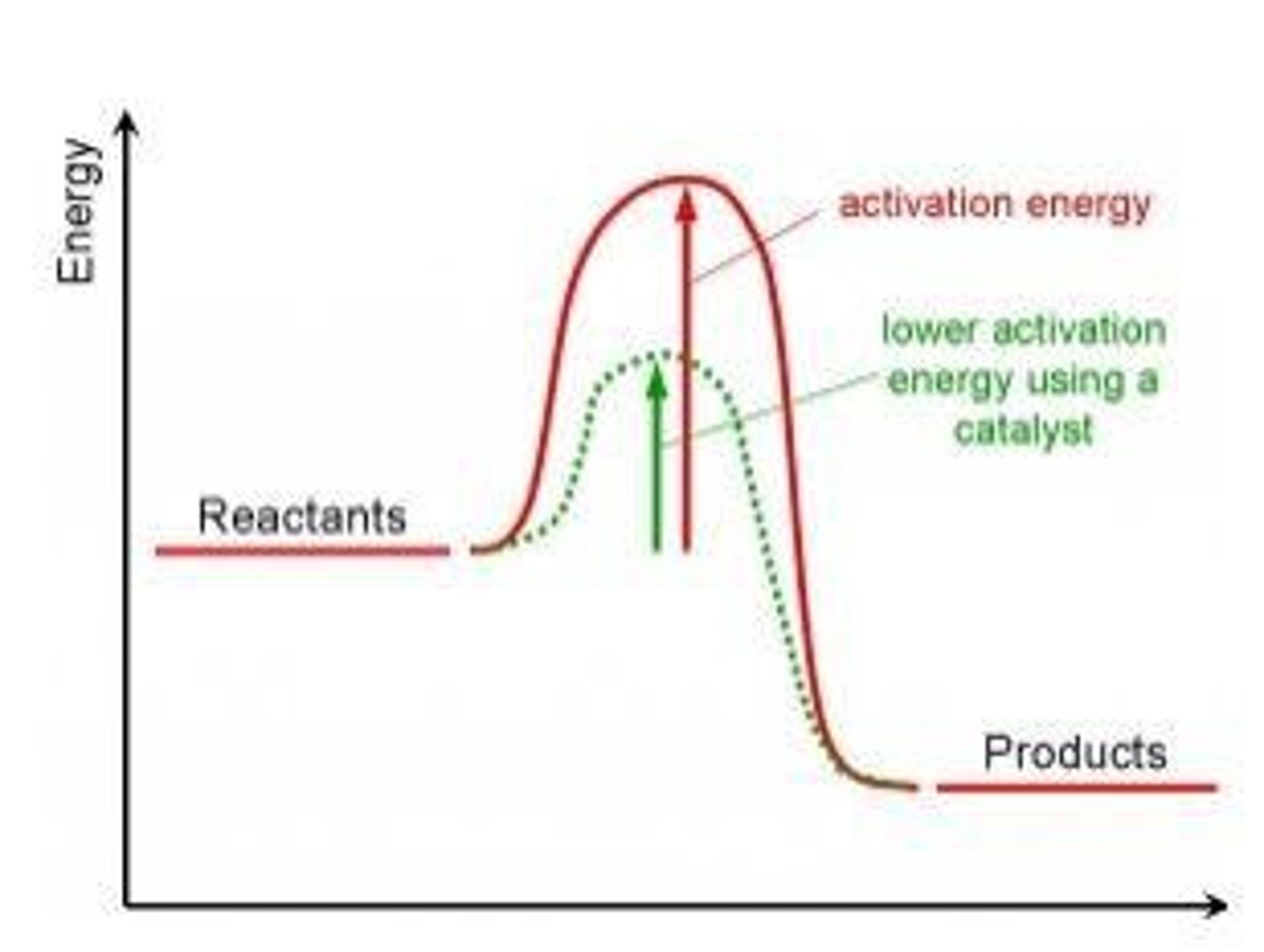
Activation energy
The minimum amount of energy that particles must have to react.
Effect of concentration on reaction rate
Increasing concentration increases the frequency of collisions and the rate of reaction.
Effect of temperature on reaction rate
Increasing temperature increases the frequency of collisions and makes collisions more energetic.
Catalysts
Substances that speed up chemical reactions without being changed or used up during the reaction.
Enzymes
Act as catalysts in biological systems.
Catalysts in reaction equations
Catalysts are not included in the equation for a reaction.
Catalysts and activation energy
Catalysts decrease the activation energy, increasing the proportion of particles with energy to react.
Reversible reactions
Products can react to produce the original reactants.
Reversible reaction symbol
Use the symbol ⇌ instead of → to represent a reversible reaction.
Haber Process example
Hydrogen + nitrogen ⇌ ammonia.
Endothermic and exothermic reactions
If a reversible reaction is endothermic one way, it is exothermic in the opposite direction.
Equilibrium in closed systems
Equilibrium is reached when reactions occur at exactly the same rate in each direction.
Le Chatelier's principle
If a system at equilibrium experiences a change, it responds to counteract the change.
Effect of changing concentration on equilibrium
Changing concentration shifts equilibrium towards products or reactants until equilibrium is reached again.
Effect of temperature changes on equilibrium
Increasing temperature shifts equilibrium in the direction of the endothermic reaction.
Effect of pressure changes on equilibrium
An increase in pressure favors the reaction that produces the least number of molecules.
Pressure and yield of reaction
An increase in pressure decreases yield of reaction if it produces a larger volume of gas.