Ichem VSEPR Shapes
1/14
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
15 Terms
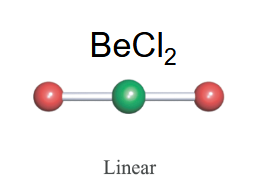
Linear
-2 bonds
-typically no lone pairs on the central electron
bent
-2 bonds
-lone pairs that push subsituent groups away
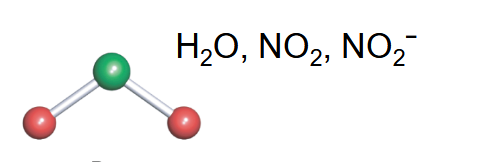
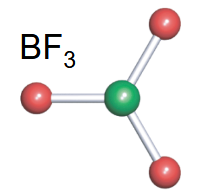
Trigonal planar
-3 bonds
-typically no lone pairs
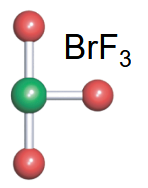
T-shaped
-3 bonds
-2 lone pairs
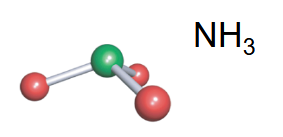
Trigonal pyramidal
-3 bonds
-1 lone pair
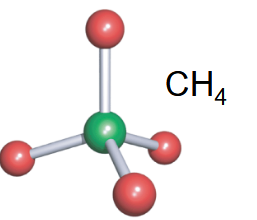
Tetrahedral
-4 bonds
-no lone pairs
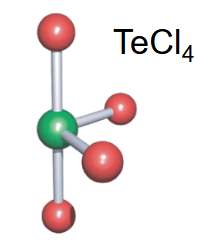
Disphenoidal
-4 bonds
-1 lone pair
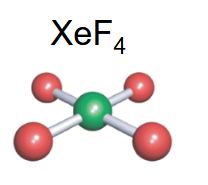
Square planar
-4 bonds
-2 lone pairs
Trigonal bipyramidal
-5 bonds
-no lone pairs
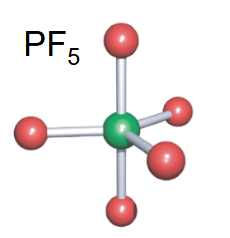
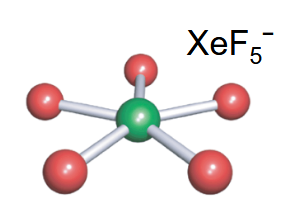
Square-Based pyramidal
-5 bonds
-1 lone pair
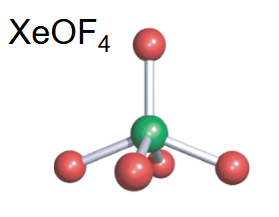
Pentagonal planar
-5 bonds
-2 lone pairs
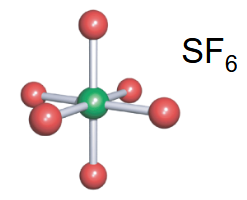
octahedral
-6 bonds
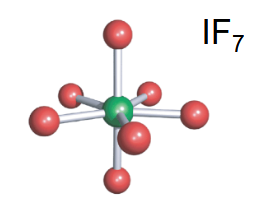
Pentagonal bipyramidal
-7 bonds
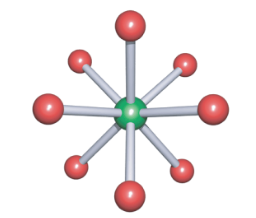
Square antiprismatic
-8 bonds
-(hardly any)
Angle rules
-bigger elements will increase angle
-but if they are further, angle decreases
-higher electronegativity, bigger orbital (lowers angle)
-if electronegativity density stays the same in the orbital, then, the angle gets bigger