WEAKNESSES - practicals + mistakes
1/60
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
61 Terms
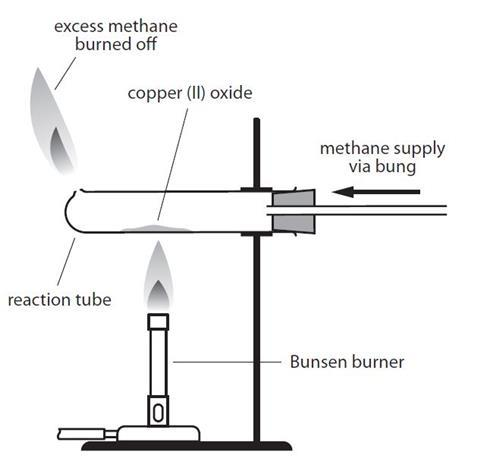
why do you need to relight the hydrogen immediately if it goes out?
to prevent hydrogen from escaping which is flammable and could cause an explosion.
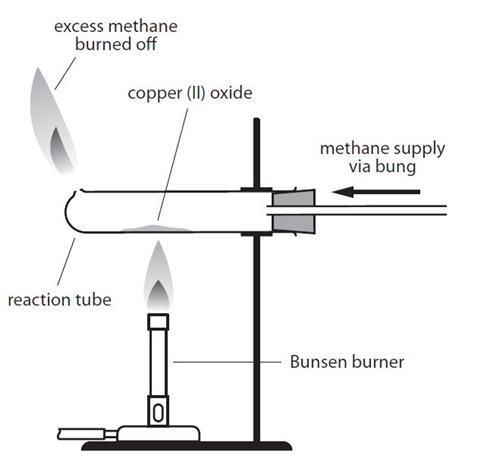
what colour is copper carbonate?
green
what colour is copper oxide?
black
what are short chain alkenes used to make?
polymers
why is ethane considered a saturated compound?
made of single carbon-carbon bonds only
no other atoms can be added
no addition reactions
how to collect gas in electrolysis
fill measuring cylinder with solution/water
place over electrode
collect the gas by displacement of the water/solution
how to test for carboxylic acids
add metal/carbonate
effervescence
definition of dynamic equilibrium
rate of the forward reaction is equal to the rate of the reverse reaction
the concentration of the reactants and products remains constant
ethanol molecular formula
C2H6O
isotope definition
atoms with the same number of protons but different number of neutrons
compound definition
atoms of two different elements chemically bonded together
why does it take a while for the ammonium chloride ring to form
gases move in random directions and collide with the walls and themselves
why does solid sodium oxide not conduct electricity
ions fixed in the lattice and cannot move
no delocalised electrons
what is the colour of silver chloride
white precipitate
advantage of mixing solutions containing the same amount of moles
make sure they fully react
highest possible yield
problems caused by the disposal of addition polymers
toxic gases when burned
lack of space in landfill sites
why do you use pencil to draw baseline in chromatography?
insoluble
Rf calculation
solute distance
solvent distance
why do you heat the crucible before adding the magnesium in combustion
to clean it of impurities
what do you add into HCl for the cross disappearing experiment temperature rate of reaction thing
sodium thiosulphate
what catalyst could you add for a rate of reaction experiment
manganese oxide
copper oxide
zinc oxide
calculation for percentage of oxygen in measuring cylinder
start volume - finish volume
start volume x 100
what is the substance inside the double gas syringe
phosphorus
reaction of magnesium with acid
vigorous
reaction of zinc and iron with acid
slow
what also do you add to make an ester other than carboxylic and alcohol
sulfuric acid
proportion of alcohol to carboxylic acid in making esters
equal volume
what do you pour the solution over to form the ester layer
sodium carbonate solution
what is the name of the paper used for chromatography
chromatography paper
what is the name for the distance moved by the solvent
solvent front
when asked to explain the shape of a graph, what are the steps to take?
describe the shape e.g steepest gradient, less steep curve, curve levels off
explain why e.g fewer particle collisions
niche marks for explaining why magnesium chloride has a much higher melting point than hydrogen chloride (2)
strong electrostatic attraction between oppositely charged ions
much more energy is required to break the ionic bonds in MgCl2 than the intermolecular forces in HCl
how can the student prove manganese oxide is a catalyst and not a reactant
filter out
dry it
reweigh it, should be the same mass
explain why diamond is hard but graphite is soft (5)
diamond has a tetrahedral lattice
every carbon atom is bonded to four other carbons
in diamond the bonds need a lot of energy to overcome and break
graphite is arranged in layers
the layers can slide over each other
why does chlorine have the highest boiling point (out of the gases in the table)
chlorine has the strongest forces of attraction between its molecules
how long do you wait in a chromatography practical?
until the solvent has risen up the paper nearly to the top
correct way to write copper sulphate
copper (11) sulphate
bonding and structure of sulfur dioxide
structure - simple molecular
bonding - weak intermolecular forces, require little energy to overcome and break
how to physically identify a pure solid vs a mixture (3)
measure the melting point
if fixed melting point the substance is pure
if melts over a range the substance is a mixture
explain how positions of chlorine, bromine and iodine depends on electronic configuration
(3)
all in group 7
all have 7 electrons in outer shell
number of shells determines period they are in
if it says how a specific compound can be separated from crude oil what do you do
describe the process of fractional distillation
say which fraction it is and where it will leave
eg top of column as a refinery gas
colour copper (11) sulphate solution
blue
actual yield less than expected (3)
spillages
impurities
not all reacted
soluble salt method: (8)
heat acid
add base and mix
filter to obtain soluble salt solution
heat solution
until crystals start to form
leave to cool
filter out crystals
dry crystals in a warm oven
arrangement and movement of particles in a gas
arrangement - random arrangement, far apart
movement - move freely, quickly
why does the lead iodide take less time to form at higher temperatures (3)
crystals dissolve faster
particles move faster
particles collide after a shorter period of time
why do both isotopes have the same chemical properties
same electronic configuration
why is carbon monoxide poisonous to humans
decreases the capacity of the blood to transport oxygen
explain why the ammonium chloride ring forms closer to the hydrochloric acid end (2)
hydrogen chloride has a higher formula mass
hydrogen chloride diffuses slower
equation for the ammonium chloride reaction
NH3(g) + HCl(g) —> NH4Cl(s)
why could metal x be silver? (2)
silver is less reactive than copper
no reaction
why can octane be used as a fuel (3)
petrol for cars
undergoes combustion
releases heat
method for calculating actual yield from percentage yield.(3)
find moles of anything given in question and do ratio
times the one you want by the Mr
times the theoretical yield by the percentage
why could a percentage yield be greater than 100% (3)
crystals not fully dried
acid used more concentrated
higher volume of acid than stated
rust chemical name
iron (111) oxide
appearance of copper metal
pink-brown/orange-brown
why is the empirical formula for the alkenes CH2 (2)
twice as many hydrogen atoms as carbon atoms
general formula is CnH2n
explain why the compound is an unsaturated hydrocarbon (3)
made up of hydrogen and carbon atoms
only
contains C=C double bonds
complete combustion of alcohols equation
look at the sneaky O in the alcohol too
why are the spots placed above the baseline (2)
they will not dissolve in the solvent
the dyes can travel up the paper
incomplete combustion what state is the water produced?
liquid