paper 2 flash cards chem
1/16
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
17 Terms
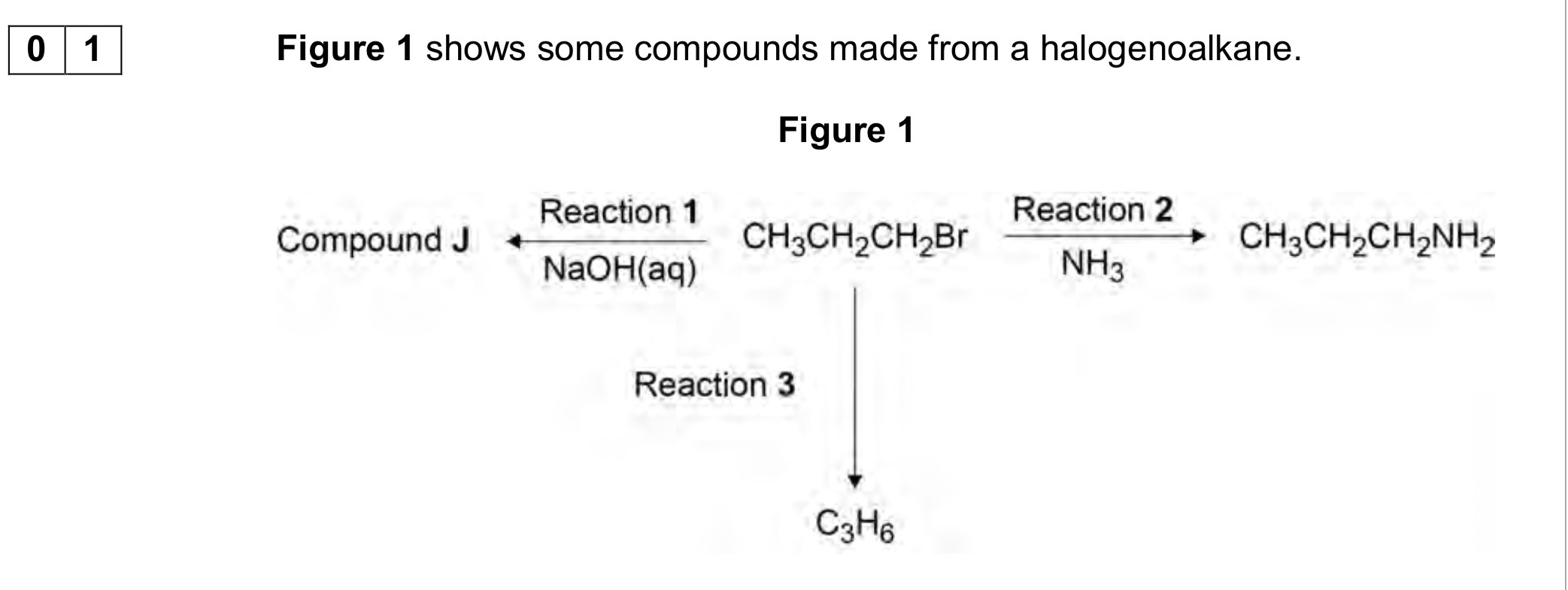
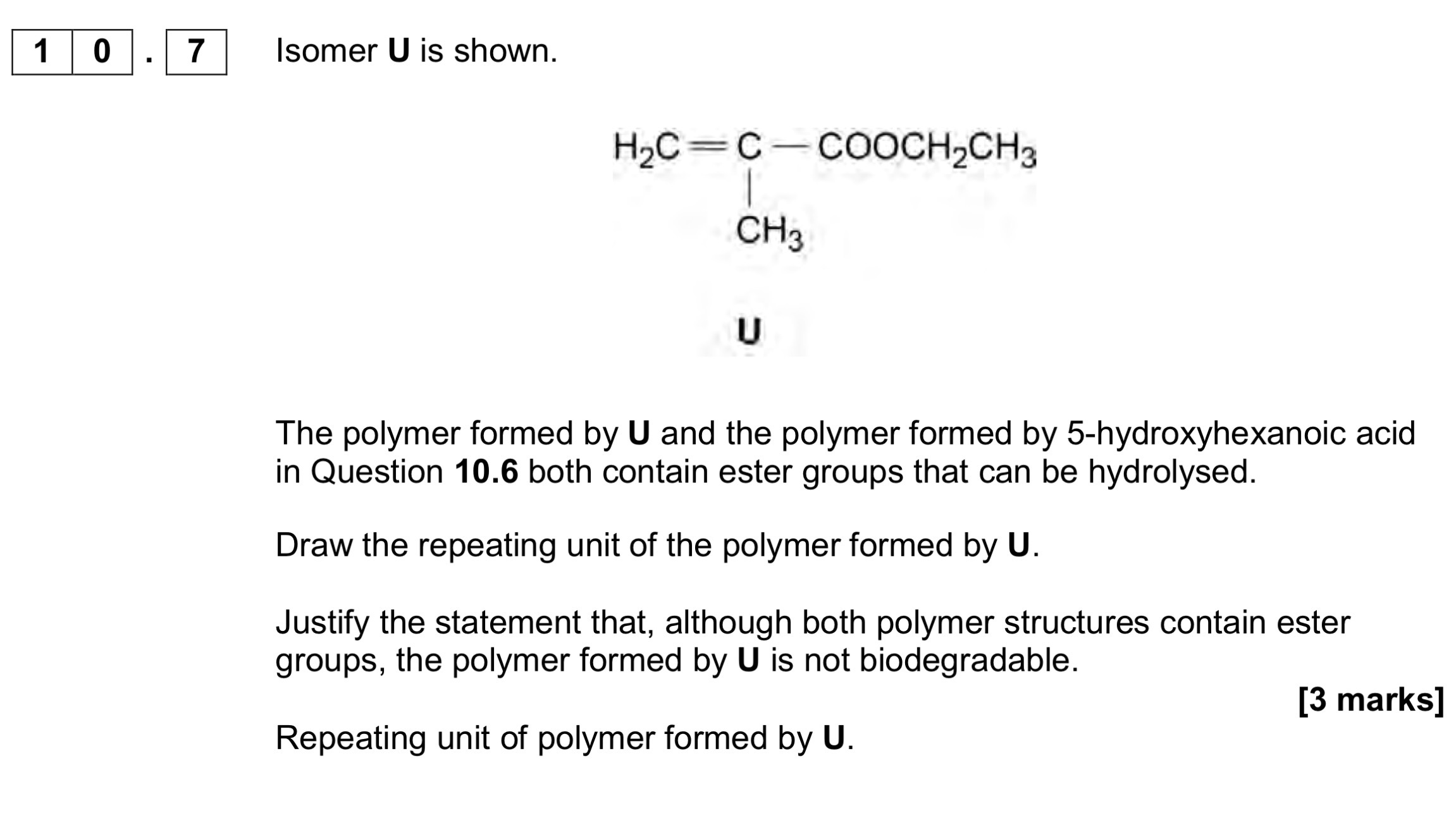
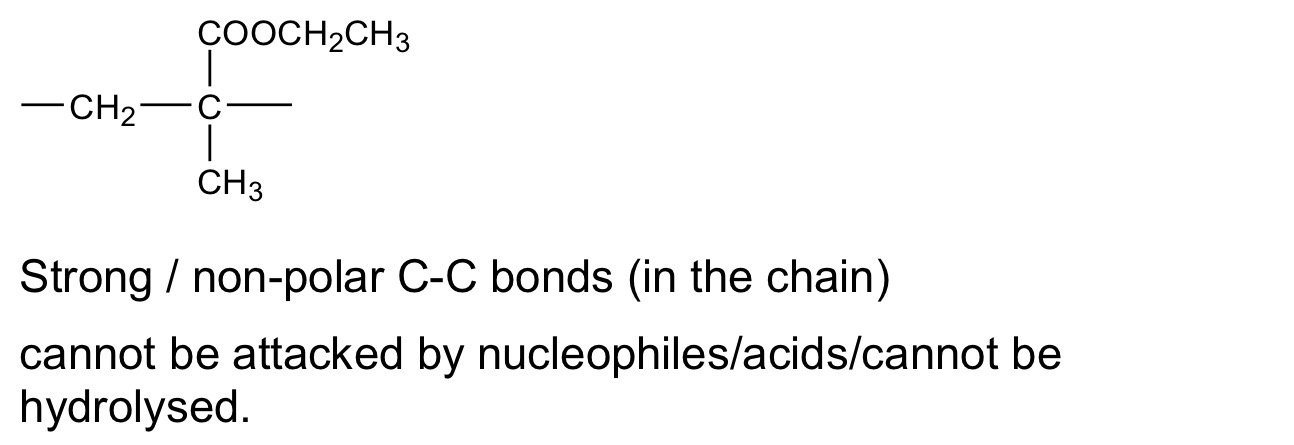
Name the mechanism for Reaction 2 and give an essential condition used to ensure that CH3CH2CH2NH2 is the major product,
Electrophillic addition, and excess NH3
When Reaction 2 is carried out under different conditions, a compound with
molecular formula C9H21N is produced.
Draw the skeletal formula of the compound.
Identify the functional group in the compound including its classification.

Identify the reagents and conditions used in reaction 3
NaOH/ethanol
Explain why (any) reaction doesn’t produce a racemic mixture
Planar carbonyl group
Can attack from either side
With equal probability
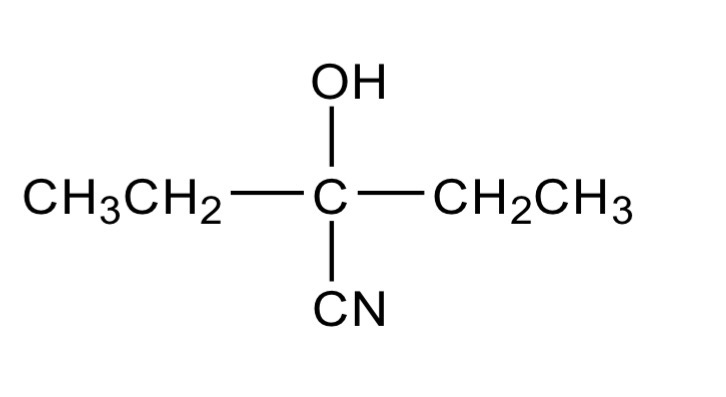
An isomer of CH3CH2CH2CH2CHO reacts with KCN followed by dilute acid to
form a compound that does not show stereoisomerism.
Draw the structure of the compound formed and justify why it does not show
stereoisomerism.
Doesn’t contain a chiral centre
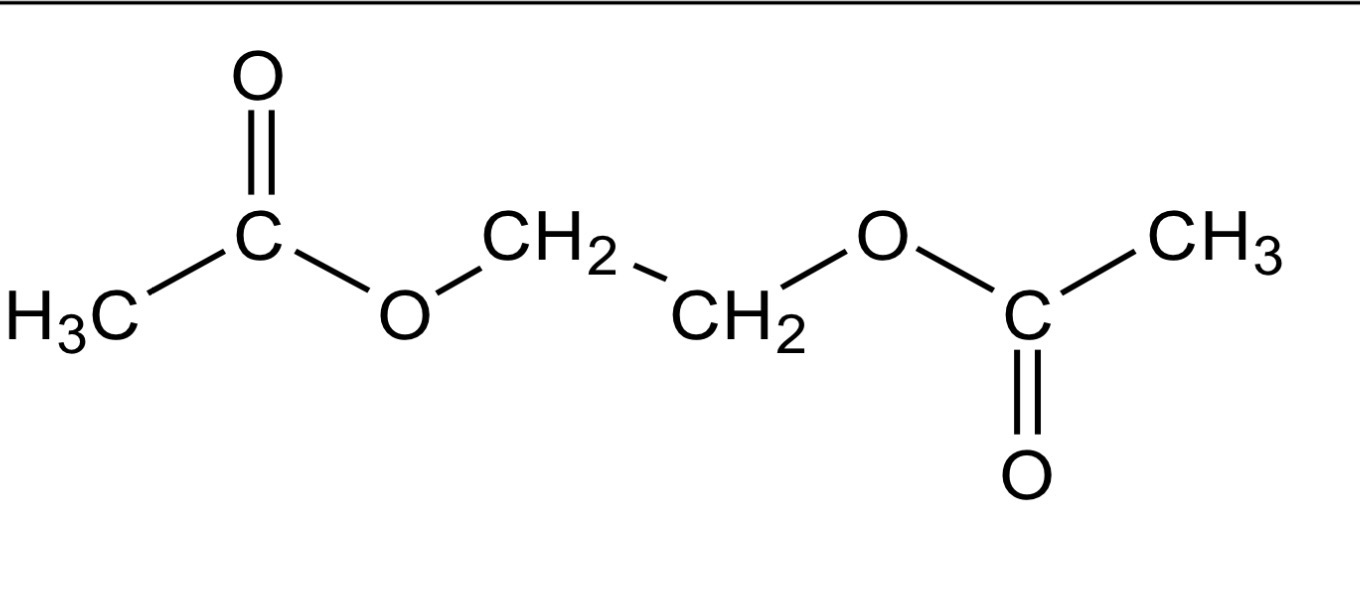
Ethanoic acid and ethane-1,2-diol react together to form the diester (C6H10O4)
as shown.
2CH3COOH(l) + HOCH2CH2OH(l) ⇌ C6H10O4(l) + 2H2O(l)
0 5 . 1 Draw a structural formula for the diester C6H10O4
State in general terms what determines the distance travelled by a spot in TLC.
[1 mark]
affinity for stationary and mobile phase
A second TLC experiment was carried out using 1,2-dinitrobenzene and
1,4-dinitrobenzene. An identical plate to that in Question 8.3 was used under
the same conditions with the same solvent. In this experiment, the Rf value of
1,4-dinitrobenzene was found to be greater than that of 1,2-dinitrobenzene.
Deduce the relative polarities of the 1,2-dinitrobenzene and 1,4-dinitrobenzene
and explain why 1,4-dinitrobenzene has the greater Rf value.
1,2 is more polar
1,4 is less attracted to stationary phase
A third TLC experiment was carried out using 1,2-dinitrobenzene. An identical
plate to that in Question 8.3 was used under the same conditions, but the
solvent used contained a mixture of hexane and ethyl ethanoate.
A student stated that the Rf value of 1,2-dinitrobenzene in this third experiment
would be greater than that of 1,2-dinitrobenzene in the experiment in
Question 8.6
Is the student correct? Justify your answer.
Yes
Solvent more polar
Polar isomer more attracted to the solvent than before
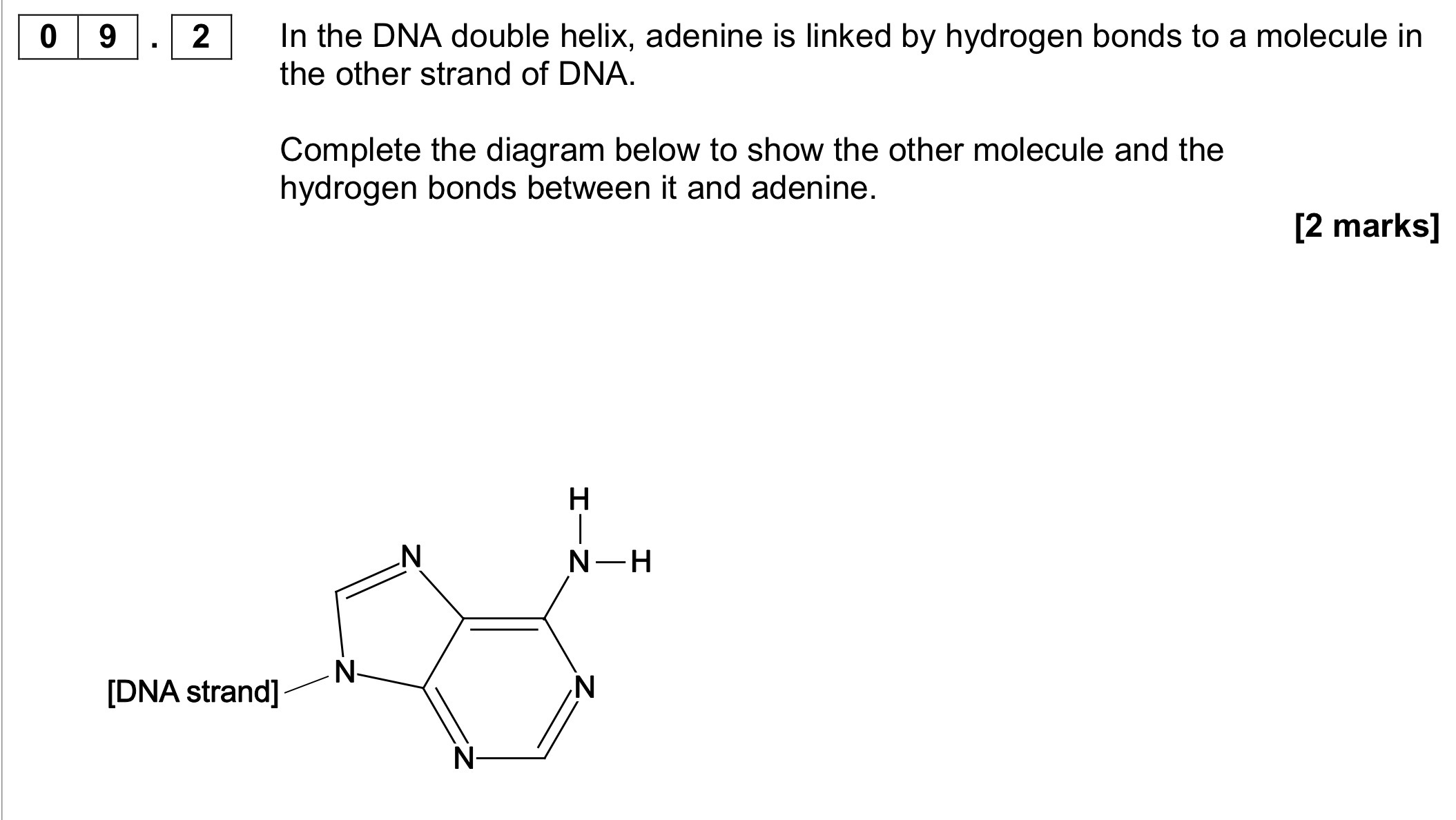
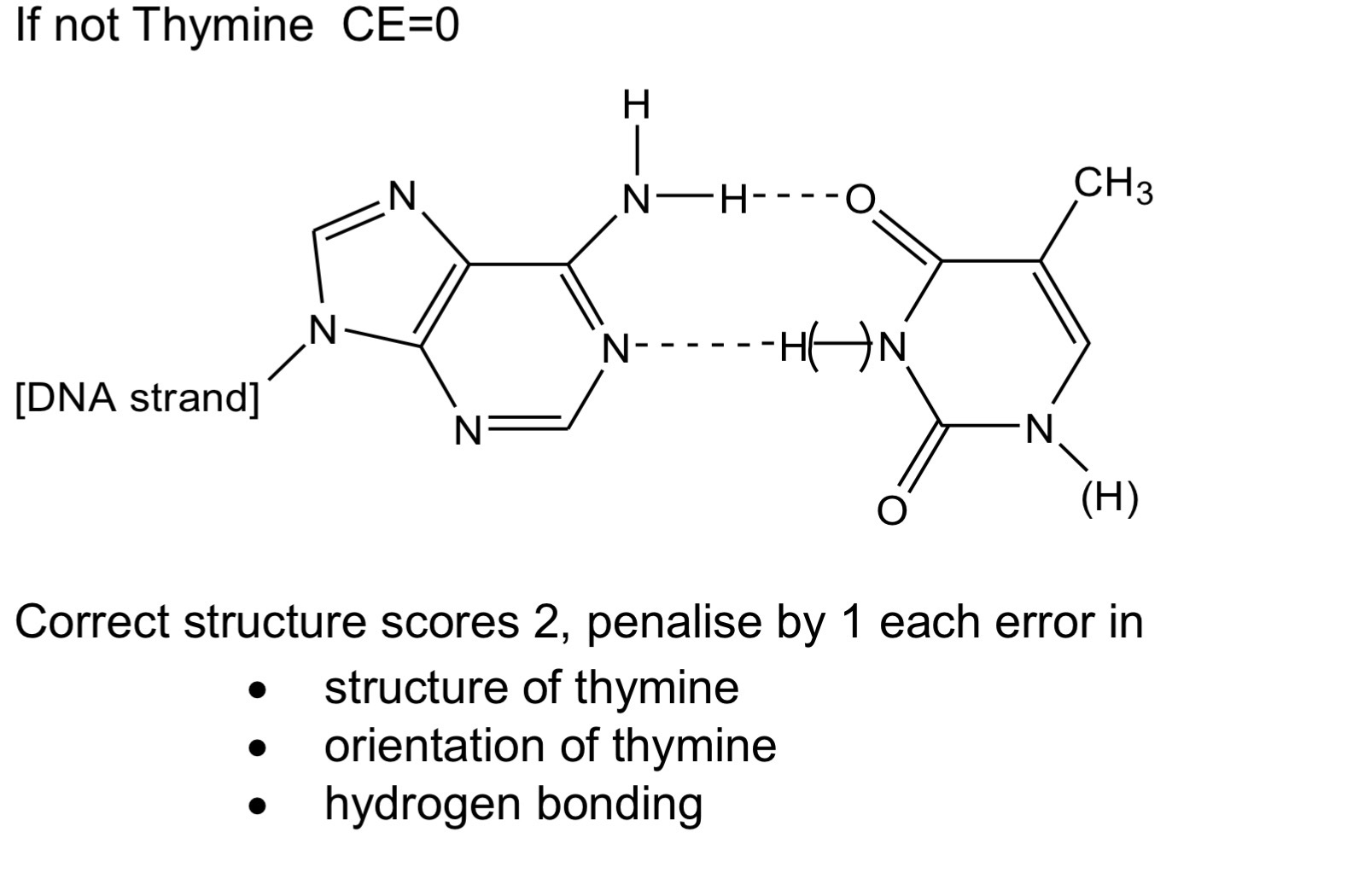
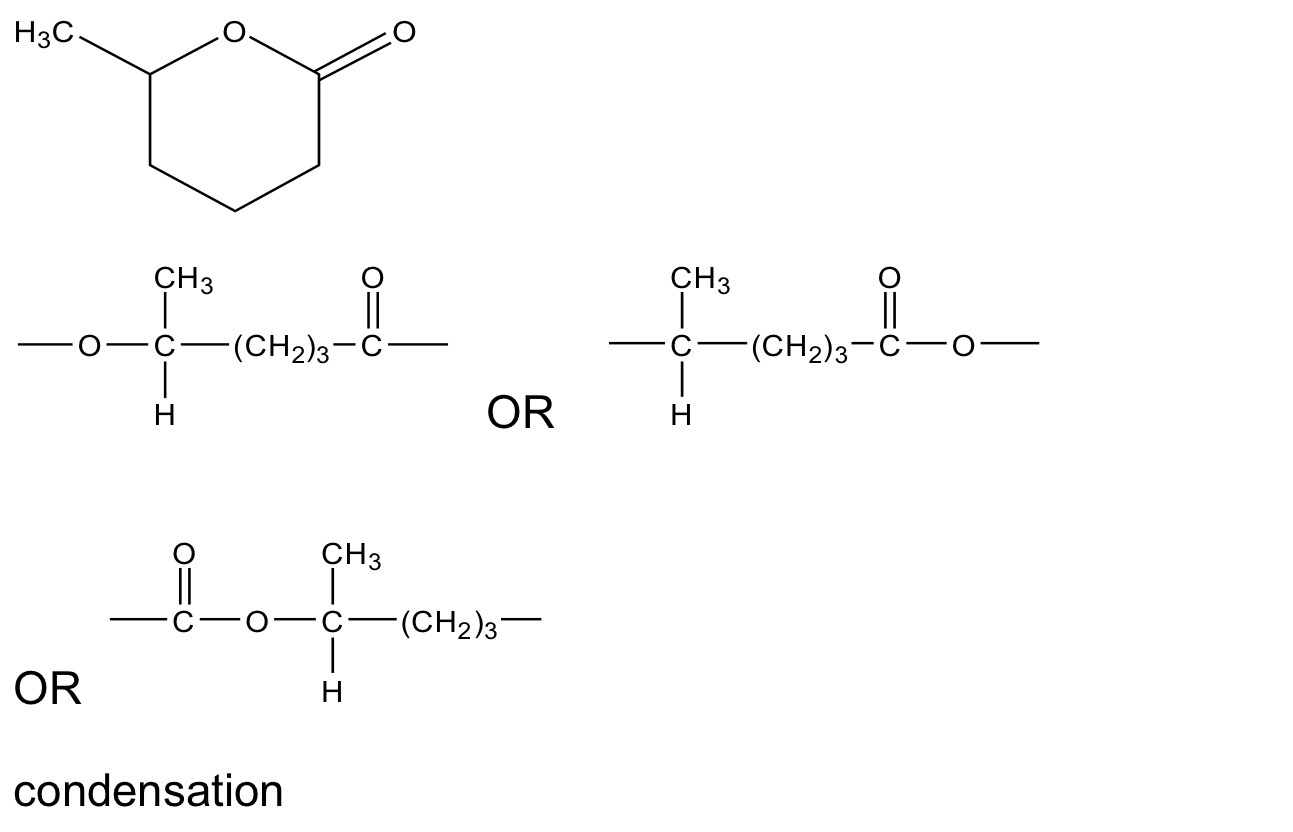
The action of heat on 5-hydroxyhexanoic acid can lead to two different
products.
On gentle heating, 5-hydroxyhexanoic acid loses water to form a cyclic
compound, T (C6H10O2).
Under different conditions, 5-hydroxyhexanoic acid forms a polyester.
Draw the structure of T.
Draw the repeating unit of the polyester and name the type of polymerisation.