Ethers
1/13
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
14 Terms
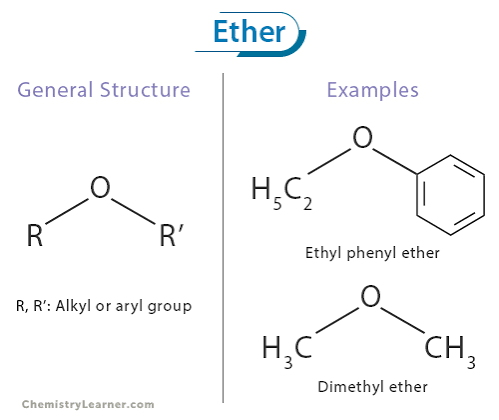
Ether Structure and General Formula
R-O-R' (Oxygen linked to two alkyl groups)
general formula CnH2n+2O
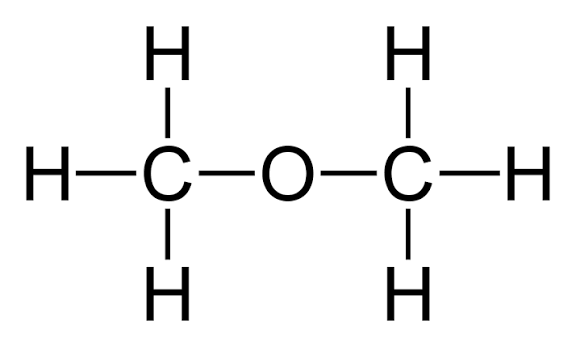
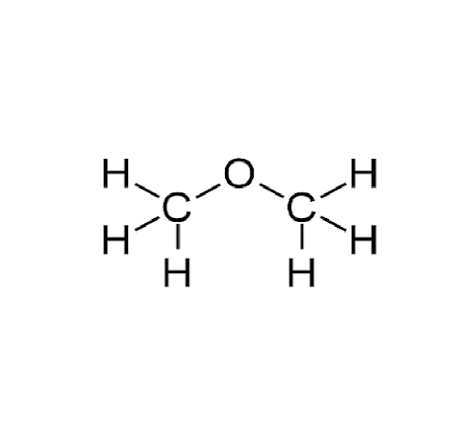
1st.2 carbon of ether (Common and IUPAC)
Common: Dimethyl ether | IUPAC: Methoxymethane (H3C-O-CH3).
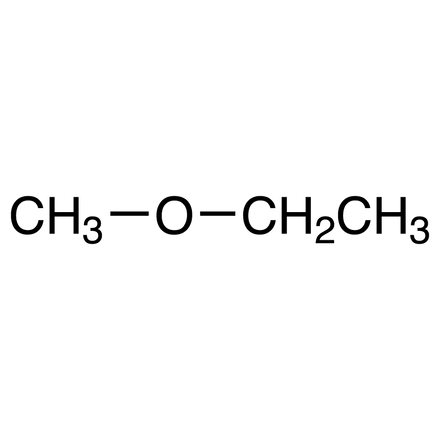
2C+O+C ether (Common and IUPAC)
Common: Ethyl methyl ether | IUPAC: Methoxyethane (C2H5-O-CH3).
Symmetrical vs Unsymmetrical Ethers
Symmetrical: Same alkyl groups
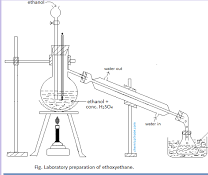
Preparation of Ethers
Dehydration of alcohols with concentrated H2SO4 at 140 °C.
Physical properties of Ethers
Methoxymethane and methoxymethane are colourless gases at room temperature.
Lower members are gases or volatile liquids, highly flammable, and unreactive.
Solubility of Ethers
Ethers with up to 3 Carbon atoms are soluble in water.
Ether Reaction with HI
C-O bond breaks to form an alcohol and an alkyl iodide (CH3OCH3 + HI -> CH3I + CH3OH). With heat
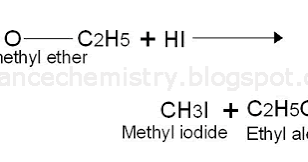
Uses of Ethers
Used as reaction solvents and anaesthetic agent before
The most common member of ethers
Ethoxyethane

Why ether are often used as reaction solvents
fairly inert
The simplest member of ethers
methoxymethane (dimethyl ether)
Nomenclature of ether
Common name: the two alkyl group’s name+ ether
IUPAC name: alkoxy(the smaller + oxygen) +alkane= alkoxy alkane
Now ethoxyethane ethane ( anaesthetic agent) are replaced by
Safer nonflammable alternative (hydrocarbon and halothane)