Acid-base and blood gasses
1/49
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
50 Terms
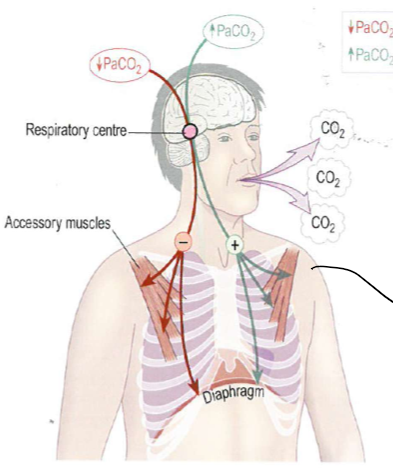
What stimulates ventilation?
responce to various mechanisms picking up on high concentration of pCO₂
If someone is struggling to breath what muscles will they use?
intercostal as well as accessory muscles
when oxygen tention is low, what happens to heamoglobin?
oxygen has been given up the tissues, becomes a really good and efficent buffer (H⁺), driven by affinity for oxygen
When oxygen tention is high what happens to heamoglobin?
becomes a less efficent buffer, driven by affinity for oxygen
How is oxygen picked up and carbon dyoxide released by haemoglobin in the lungs?
haemoglobin is not an efficient buffer, it wants to pick up oxygen and does, this produces oxidised haemoglobin and hydrogen, the hydrogen produced in the cell is on the right hand side of an equation with a bicarbonate ion from the reverse chloride shift producing carbolic acid which then produces water and CO₂, which is catalysed by carbonic anhydrase, the CO₂ is released into alveolar
Carbamino-haemaglobin complex, haemoglobin has greater affinity for oxygen so gives up CO₂
slow reaction in plasma, and diffusion in plasma
oxygen is dissolved in the plasma
reverse chloride shift - to balance electroneutrality - chloride ions out into extracellular fluid, bicarbonate ions into the red blood cell
How is oxygen released and carbon dyoxide picked up by haemoglobin in the tissues?
picking up CO₂ which reacts with water to form carbonic acid, which then dissociates into bicarbonate (conjugate base) and hydrogen ions - reaction goes back and forward depending on concentration of reactants, reaction is catalysed by carbonic anhydrase - as area of low oxygen tention, haemoglobin is a good buffer and will pick up the H⁺ ions that are produced
the bicarbonate and chlorine ions are exchanged for each other to maintain electrochemical neutrality, the bicarbonate ends up the extracellular fluid so can be transfered to other structures (called the choride shift)
Some CO₂ just dissolves into the blood plasma
Also CO₂ in the plasma doing the above reaction without catalysation so slower, the H⁺ ions bind to plasma proteins (diffuses out of cells and then dissolves in the plasma)
Some CO₂ combines with haemoglobin to form carbamino-haemoglobin compounds which remains in blood cell, haemoglobin gives up oxygen, this oxygen moves into the tissues
Also oxygen that is in the blood plasma, moving into tissues as well
Based on haemoglobin is a better buffer for H⁺ and also wants to release O₂
How could gas exchange with red blood cells be effected?
anaemia - lower concentrations of haemoglobin
drugs that inhibit carbonic anhydrase especially those involved in the treatment of glacoma
becomes statuated - giving oxygen does not help, but have a massive capacity to shift carbon dyoxide
What happens to hydrogen ions in normal metabolism?
hydrogen is produces from: digestion of food, metabolism of drugs, errors within metabolism, and incomplete oxidation of energy substrates, they are consumed by futher metabolism of intermediates, anerobic metabolism can produce lactate that causes an imbalance, the varience is normally balanced out and is normally transient, the kidney is mainly responsible for excreation of hydrogen ions through urine, but can’t be free hydrogen as would burn
What is the pH of urine?
about 4.2 - cannot fall below this
What happens to carbon dyoxide in normal metabolism?
produced due to oxidative metabolism, excreated by ventilation and is removed in expired air, lungs have segnificant capacity to move excess carbon dioxide, carbon dioxide can contribute to the production of H⁺ ions via CO₂ + H₂O ↔ HCO₃ ↔ HCO₃⁻ + H⁺
Why do you need bicarbonate ions?
to get rid of H⁺ ions
What is normal pH of the extracellular fluid?
between 7.35-7.46, slightly lower within cells
How are H⁺ ions managed in homeostasis?
buffering system: H⁺ + HCO₃⁻ ↔ H₂CO₃, normal H₂CO₃ concentration is 1.2mmol/L and HCO₃⁻ concentration is 24-25 mmol/L, production of bicarbonate to balance this system means we have large amounts of H⁺ formed as by product, this managed by kidneys, this is out of kilter not in equilibrium but creates electroneutralitiy
concentration of bicarbonate in the renal tubular lumen and the blood is about the same, we would loose lots of bicarbonate if this continues, sodium is exchanged for Hydrogen - hydrogen moves into lumen, combines with bicarbonate to form carbonic acid, bicarbonate in the tubular fluid cannot diffuse back into the renal tubule cell, carbonic acid dissociates into H₂O and CO₂, these then move into the tubule cell, CO₂ combines with the water to form bicarbonate, the bicarbonate is then reabstobed into the blood with sodium, the H⁺ ions move into the lumen again
Glutanine is metabolised to ammonium ion and HCO₃⁻, this ammonia diffuses across or can be shunted across into the lumen, binds to H⁺ ions to form ammonium which is unable to transfer back into the cells, this is excreted in urine
Phosphate buffer system, H⁺ ions in the tubular lumen bind with monohydrogenphosphate to form dihydrogenphosphate, this is lost in urine
Neither of these massivly influence the pH of urine
How fast can systems adjust the pH of the body?
respiritary system can ajust very quickly, kidneys can change but they change slowly
What happens in a ventilation/perfusion mismatch?
Physiological deadspace - ventilation with reduced perfusion caused by pulmonary embolism, pulmonary artenitis, necrosis or fibrosis
Physiological shunt - perfusion with ventilation reduced - caused by airway limitation, lung calapse or consolidation, loss of elastic tissue, disease of chest wall
How are H⁺, PCO₂ and HCO₃⁻ concentrations related?
CO₂ + H₂O ↔ H₂CO₃, H⁺ + HCO₃⁻ ↔ H₂CO₃, assume [H⁺] = K PCO₂/[HCO₃⁻], so H⁺ concentration is directly proportional to PCO₂ and inversly proportional to HCO₃⁻ concentration
What is acidosis?
high [H⁺] and low pH
What is alkalosis?
low [H⁺] and high pH
What are the types of acidosis/alkalosis?
non-respiratory (metabolic), respiratory, or mixed (unclear which came first)
What are the componants of the process that could cause hydrogen ion abnormalties?
generation, buffering, compensation, correction (in-balance corrected incorrectly)
What are the causes of Non-respiratory acidosis?
Increased [H⁺]: keto/lactic acidosis, poisons - acids, salicylate, polyethylene glycol poisoning, inherited metabolic disorder
Decreased excretion - renal tubular acidosis, renal failure, agents inhibiting CA - acetozolamide, topirimate, and brinzolamine
Deacresed [HCO₃⁻] - diarrhoea, loss from GI tract into peritoneum - fistulae
What is keto acidosis?
absolute defisit in insulin, so we start metabolising fatty acids as a substrate, causes intermeadiate ketones which are acidic, excess glucose in bood causes dehydration as kidney is excreating glucose and with glucose goes water, sodium goes too, urea goes up, treatment is insulin and rehydration
What common drug can lead to non-respiratory acidosis?
asprin poisoning, large volumes causes vomitting due to high breakdown of stomach lining
How does renal failure lead to non-respiratory acidosis?
decreased GFR reduces filtration of sodium ions and there is less availible to be exchanged with hydrogen, so hydrogen ions accumulate, also means reduced phosphate and this mechnaism is comprimised for clearing hydrogen ions
How can decreased bicarbonate occur that eventually leads to non-respiratory acidosis?
fistulae are openings in the GI system that allow GI fluids to escape, gastic secreations are acidic, pancreatic secreations are alkaline, two systems balance each other, but if fistula exists they can be lost and become imbalanced, fluid loss also depleates the extra cellular fluid and results in a falling GFR which compromises compensatory mechanisms, also applies to fluid loss from diarrhoea
What does changes in bicarbonate concentrations suggest?
immediate neutrilisation of hydrogen ions, and compensatory changes by the kidney (slower than respiritory ones)
What happens in non-repiratory acidosis (from too much H⁺)?
[H⁺] rises - this is buffered by bicarbonate which falls stochiometrically, carbonic acid is produced and dissociates producing carbon dioxide, this is lost on expiration, as [H⁺] rises non-respiratory acidosis develops. stimulates respitatory centre to remove more carbon dioxide producing compensatory respiratory alkalosis, respiratory response is rapid, compensation is limited as [H⁺] rise
What is kussmaul respiration?
rapid, shallow breathing
What do you expect to see in someone with non-repiratory acidosis caused by fluid loss?
high pulse rate, low blood pressure - pulse rate increases to try to increase blood pressure but fluid in system isn’t there , kussmaul breathing, pH low - hydrogen ions high due to dehydration, metabolic processes, ketones in the blood as body metabolising fats not glucose, low PCO₂, bicarbonate is low, breath smells like pears, bicarbonate concentration - decemated in trying to neutralise H⁺ as kindeys slow to adapt will balance out eventually, PO₂ probably normal, low sodium, high potassium, urea level high, creatine is increasing and glucose level is very high - losing fluid and sodium with it, insulin pushes potassium into the cell, no insulin so its all in the serum, also why glucose is high
Why would someone’s breath smell like pears?
high levels of ketones in circulation, from break down of fatty acids instead of glucose
How do you treat non-respiratory acidosis?
remove underlying problem without disrupting renal function (e.g. ketoacidosis give insulin), could use bicarbonate but carefully and only in those whose pH is <7 with constant arterial [H⁺] monitoring - could over correct it, chronic acidosis can develop if cause not corrected or would die
What do you always see in non-resipiratory acidosis?
dehydration
What do the concentrations of different substances do in non-repiratory acidosis?
pH decreases, [H⁺] increases, PCO₂ decreases, [HCO₃⁻] really decreases, every 1mmol/L fall in bicarbonate is matched by around 0.17kPa fall in PCO₂
What are the causes of respiratory acidosis?
airway obstruction, depression of respiratory centre, neuromuscular disease, pulmonary disease, extra pulmonary thoratic disease
How do you differentiate between non-repiratory and respratory acidosis?
all conditions associated with respiratory acidosis are characterised by an increase in PCO₂, is there PO₂ low - if so it is always respiratory
What is hypercapnia?
high levels of PCO₂
What are the two types of respiratory failure?
type 1 - altered PO₂
type 2 - altered PO₂ and PCO₂
What do we breath in reponse to?
CO₂ concentration
What occurs in respiratory acidosis?
acute - pH decreses, [H⁺] increases, PCO₂ increases, slight [HCO₃⁻] increase - kidneys are not compensated
chronic - pH slight decrease or low normal, [H⁺] slight increase or high normal, increased PCO₂ (permanent), increased [HCO₃⁻] - kidneys are compensated, occurs in COPD, if you expose body to high levels of CO₂, then start to have hypoxic drive instead of CO₂ drive, have to be careful when giving oxygen due to this
What happens in repiratory acidosis?
low repiratory function, obtain CO₂, stimulate various chemoreceptors causes repiratory compensation only if you can breath, normally, if you can breath faster you will, this gets rid of the CO₂ and takes you back to homeostasis
if you can’t do that you get permenant high levels of CO₂, renal compensation carbonic acid, bicarbinate system, involves H⁺ secreaton and HCO₃⁻ generation, buffer systems other than the carbonic acid system accepts H⁺ ions, homeostatis is returned, chronically sometimes the CO₂ is left high
What is a flail chest?
a section of the chest has come off
What should you worry about first with respiratory acidosis?
hypoxia will kill them first, restore oxygen levels: oxygen (high con.), ventilation or other measures if chronic disease
What happens in non-respiratory alkalosis?
an increase in bicarbonate levels for various reasons, self limiting increase in PCO₂ designed to increase [H⁺], decrease in [H⁺] concentration
if chronic, excess renal bicarbonate reabsorbtion possibly related to hypovolemia (or dehydrated) or potassium depleation, in hypovolemia Na reabsorbtion increase which causes bicarbonate to be reabsorbed as normally there is a Cl deficit, in exchange for hydrogen and potassium secreation
drive to produce more H⁺ but produce a bicarbonate every time you do so doesn’t help, urea could be high and so could sodium as dehydrated so trying to hold onto fluid, potassium will be lowered, worried
What causes non-respiratory alkalosis?
excess alkali administration: overcorrection of acidosis, excess ingestion, including excess indigestion of antacids
loss of few hydrogen: GI loss (chron’s, fisulae, vomitting diarrhoea), renal loss caused by mineralocorticoid steroid administration or disease causing the same, diuretic therapy, potassium depleation, overly rapid correction of chronic respiratory acidosis
How do you manage non-respiratory alkalosis?
correction of volume depletion with normal saline infusion, potassium suplements sometimes
What happens in respiratory alkalosis?
decrease in PCO₂ levels, decreased [H⁺], decrease in [HCO₃⁻]
What are the causes of respiratory alkalosis?
Hypoxia - increased CO₂ loss due to increased reporatory drive: altitude, anaemia, pulmonary disease, pulmonary oedema
increased drive - infections/trauma, respiratory stimulants, cerebral malignancy, hyperventilation (most common), mechanical hyperventilation (ICU)
How do you correct repiratory alkalosis?
if hyoerventlation - breath in and out of a paper bag so you are rebreathing your air as you will rebreath carbon dyoxide
otherwise remove underlying cause
What are the changes in substances in the serum for respiratory alkalosis?
acute - increased pH, decreased [H⁺], decreased PCO₂, slight decrease in [HCO₃⁻], bicarb is falling as CO₂ is falling
chronic - slight increase or low normal pH, slight decrease or high normal [H⁺], decreased PCO₂, decreased [HCO₃⁻]
What is the normal respiratory rate?
12-18 breaths per minute