CARBOXYLIC ACID, DERIVATIVES
1/45
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
46 Terms
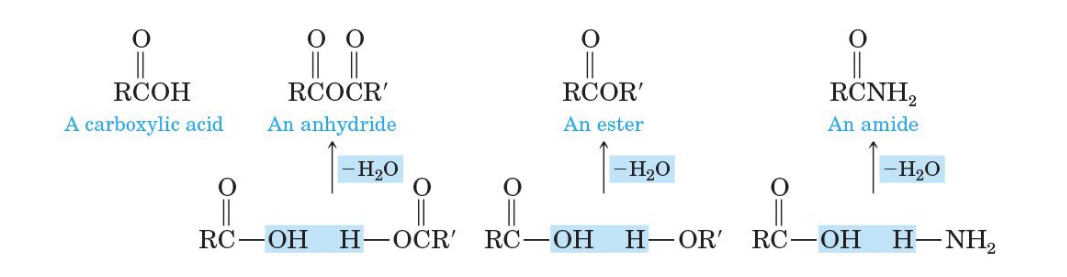
3 compounds derived from carboxylic acids and how theyre related by
anhydride, ester, amide. related by loss of h2o
anhydride
2 carbonyl groups bonded to same oxygen + drawing
types of anhydrides
symmetrical (2 identical acyl groups), mixed (2 different acyl groups)
ester
carbonyl group bonded to OR group (R group alkyl/aryl)
aryl
aromatic ring
what is a cyclic ester called
lactone
ending of ester
oate
amide
carbonyl group bonded to nitrogen
reminder for naming amide
drop oic acid from iupac name/ic acid from common name then add amide
what if amide is bonded to alkyl/aryl group
show location on nitrogen via 'N' or 2 by N, N,di
cyclic amide is called
lactam
penicillin and cephalosporin is referred to as
beta lactam antibiotic
fatty acids
long chain carboxylic acids from animal fat/vegetable oil/phospholipid/biological membrane. 500 isolated from various cell. 12/20 carbons in unbranched chain.
which isomer predominates in unsaturated fatty acids
cis. trans is rare
how is ester formed
carboxyl + alcohol under sulfuric acid
melting point of unsaturated fatty acids
lower than saturated
saturated fatty acids are __ at room temp + why
solid. minimizes london dispersion forces
unsaturated fatty acids are __ at room temp + why
liquids. cis double bond interrupts regular hydrocarbon chain
tallow
insoluble triglyceride layer on top. solid fat (from cattle) melted with steam
triglycerides are
triesters of glycerol
how is soap prepared
boil triglyceride with NaOH. undergoes saponification. boiling with KOH gives potassium soap
describe micelles
hydrophobic tail inside, hydrophilic head outside
what happens when micelle gets dirt
mixed in water, nonpolar hydrocarbon inner part dissolves nonpolar substances
natural soap + hard water
water INSOLUBLE salts
what does hard water contain
ca2+, mg2+, fe3+
how do detergents avoid forming scum (insoluble precipitate) from hard water + soap
sulfonate group (so3) replaces carboxylate co2 group
common additives of detergents
foam stabilizer, bleach, optical brightener
acidity of carboxylic acids
weak acids. Ka 4/5 to 10, pKa 4 to 5
what increases acidity of carboxylic acids
substituents w high electronegativity e.g. OH, Cl, NH3+
form of carboxylic acid when carboxylic acid dissolves in aqueous solution depends on
pH of solution
carboxylic acid + NaOH,KOH/strong base, ammonia, amine
water soluble salt
carboxylic acid + sidum bi/carbonate
water soluble sodium salt + carbonic acid (carbonic acid splits to co2 + h2o, evolve as gas)
what are carboxylic acids resistant to
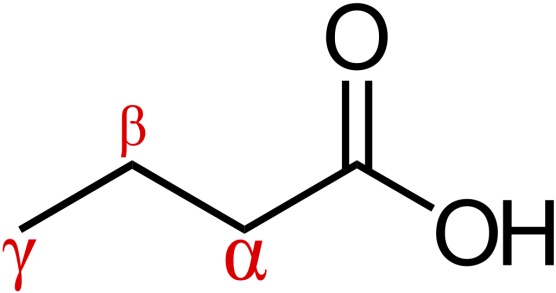
moderate heat. melt/boil without decarboxylation, except for carbonyl group beta to the COOH group
common preparation of amide
anhydride + amine
3 representations of carboxyl group
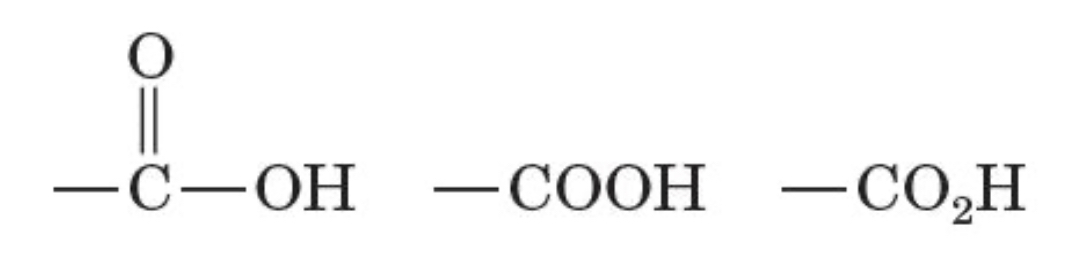
diagrams of derivatives
structure of common agents in detergents
beta carbon diagram
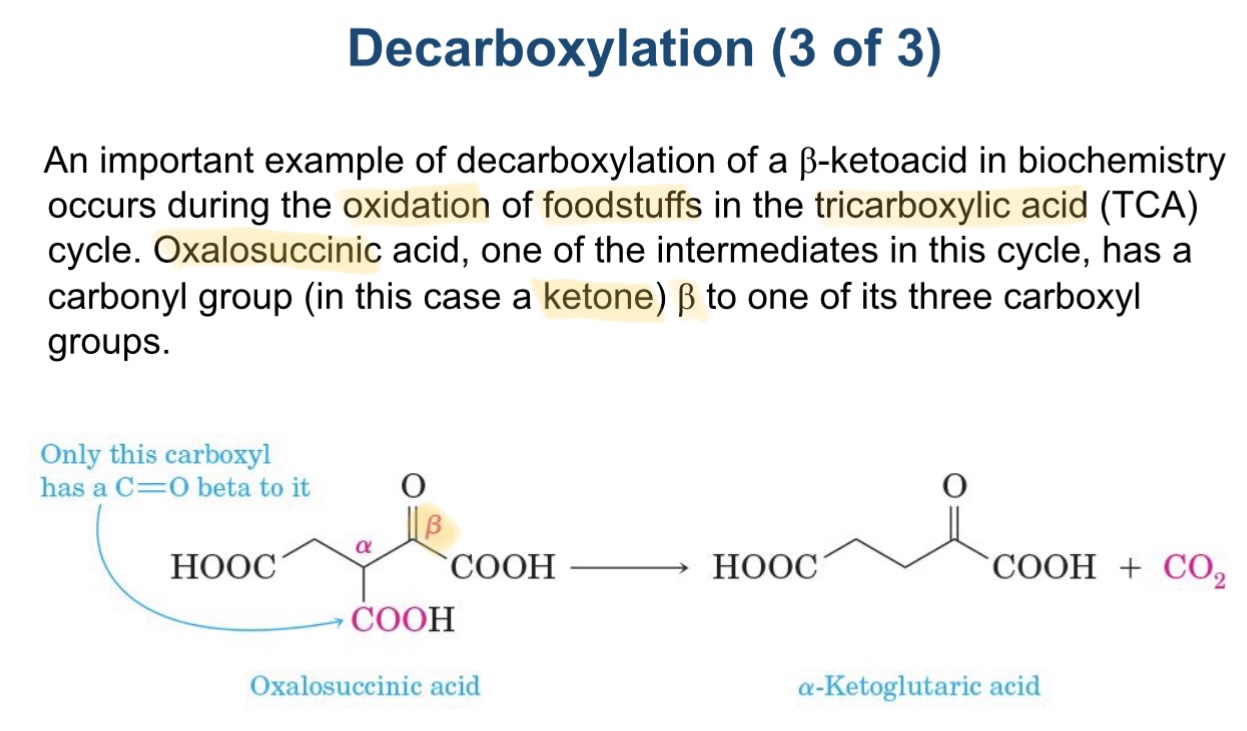
example of decarboxylation diagram
physical properties of carboxyl group-
3 polar covalent bonds, C-O, C=O, O-H
physical properties of carboxylic acids + why
high bp than other types due to: polarity, hydrogen bonding between carboxyl groups creates dimer behaving as high molecular weight compound
soluble than other types
describe soap composition
sodium/potassium salt of fatty acids
tallow + palm oil (triglyceride)
describe what happens during esterification
alcohol adds to carbonyl group of carboxylic acid
forms tetrahedral carbonyl
^ loses water to give ester
describe decarboxylation
loss of co2 from carboxyl group
carboxylic acids heated to high temps
electrons redistribute cyclically followed by keto enol tautomerism
describe preparation of amide (carboxylic acid + amine)
carboxylic acid + amine
remove OH from carboxylic acid
remove H from amine
forms ammonium salt
salt heated to high temp to eliminate water = amide