Electrode potentials and electrochemical cells
1/51
Earn XP
Description and Tags
Contains electrode potentials and cells, commercial applications of electrochemical cells.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
52 Terms
What is a half cell?
One half of an electrochemical cell. A half cell consists of a metal dipped in a solution containing it’s ions OR a platinum electrode dipped in two aqueous ions.
What is the redox reaction that takes place at a half cell (when the metal electrode comes into contact with it’s ions)?
M+ (aq) + e- →← M (s) (reversible reaction)
True or false? The reaction at a half cell is in equilibrium.
True.
Why might a platinum electrode have to be used in a half cell where there are two aqueous ions?
Some ions do not have a solid form. Platinum is inert (so does not react with the aqueous ions) but electrically conductive.
Give the general equation for the redox reaction that takes place at a half cell containing two aqueous ions (M³+ and M²+) and a platinum electrode.
M³+ (aq) + e- →← M²+ (aq) (reversible reaction)
Give one disadvantage to using a platinum electrode.
Platinum is very expensive.
What equipment would you need to make an electrochemical cell? How would you assemble this equipment to make it?
Two half cells, a salt bridge, two wires, a voltmeter.
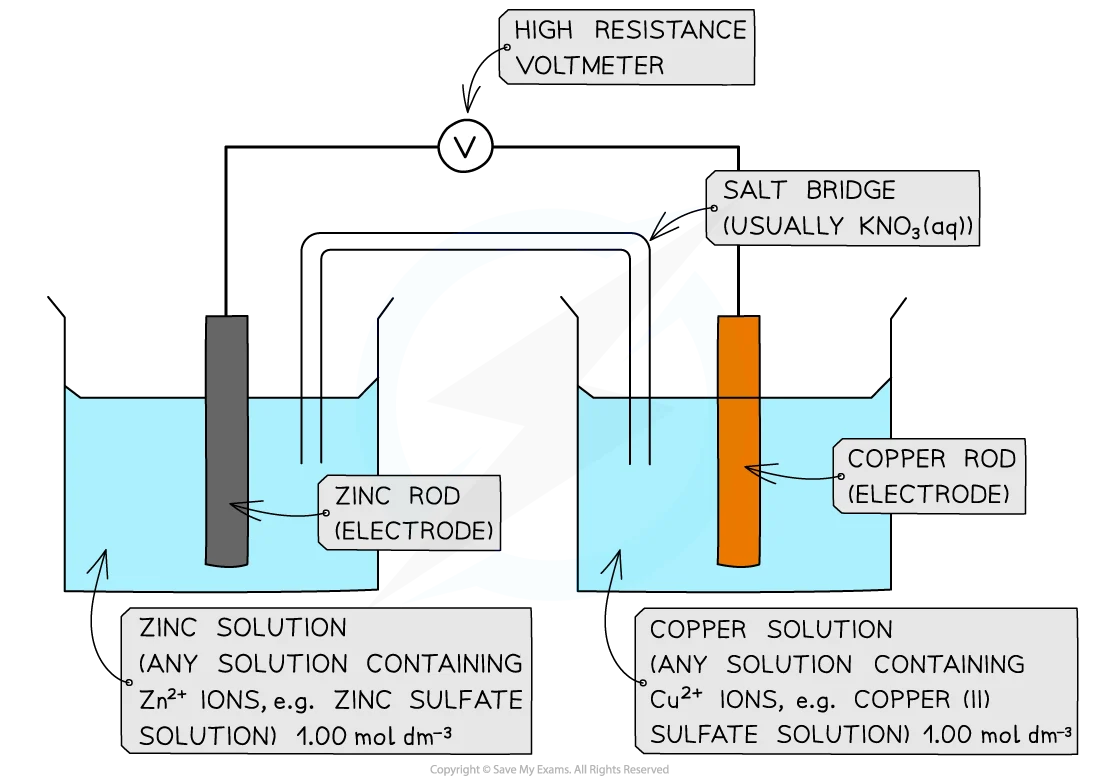
This is how you would assemble this equipment to make an electrochemical cell.
True or false? In an electrochemical cell both half cells are undergoing oxidation.
False. IN AN ELECTROCHEMICAL CELL ONE HALF CELL IS UNDERGOING OXIDATION AND THE OTHER HALF CELL IS UNDERGOING REDUCTION, OVERALL A REDOX REACTION IS TAKING PLACE IN THE ELECTROCHEMICAL CELL.
What is the purpose of the voltmeter in an electrochemical cell?
The voltmeter measures the potential difference (EMF OR Ecell) between the two half cells.
True or false? In an electrochemical cell, electrons flow from a less reactive metal to a more reactive metal.
False. IN AN ELECTROCHEMICAL CELL, ELECTRONS FLOW FROM A MORE REACTIVE METAL TO A LESS REACTIVE METAL.
Describe and explain what happens at the more reactive electrode.
The more reactive electrode loses electrons meaning that it is oxidised and more of it’s aqueous ions are formed. This decrease in the amount of solid (caused by increase in amount of aqueous ions) causes the electrode to become thinner.
M (s) → ← M+ (aq) + e-
Describe and explain what happens at the less reactive electrode.
The less reactive electrode accepts the electrons lost by the more reactive electrode and is therefore reduced. The positively charged aqueous ions react with the electrons forming more of the solid, this causes the electrode to become thicker.
M+ (aq) + e- → ← M (s)
What does the salt bridge tend to be?
Filter paper soaked in saturated potassium nitrate solution.
What is the purpose of the salt bridge in an electrochemical cell?
It allows the movement of ions between half cells without mixing the two solutions at each half cell. It maintains electrical neutrality within the electrochemical cell. Completes the circuit.
Why is it important that the salt bridge doesn’t come into contact with either of the electrodes at the half cells?
If the salt bridge came into contact with either of the electrodes at the half cells then uncontrolled reactions may take place that cause the electrochemical cell to short circuit.
What is the electrode potential (E^0) measured in?
Volts (V)
What do electrode potential values tell us about half cells?
They tell us how easily a half cell loses electrons (is oxidised).
True or false? In electrochemical cells the half cells are always written with the reduction reaction being the forward reaction.
True. (this isn’t actually what is happening in the cell as you know that a redox reaction is really taking place).
What does the acronym NO PRoblem mean?
The half cell with the most Negative electrode potential will undergo Oxidation (so is more reactive). The half cell with the most Positive electrode potential will undergo Reduction (so is less reactive).
What is the use of the standard hydrogen electrode (SHE)?
It is used as a reference to measure electrode potentials (under standard conditions) as the SHE value is 0.00V.
True or false? Electrode potentials of half cells can be measured on their own.
False. ELECTRODE POTENTIALS OF HALF CELLS CANNOT BE MEASURED ON THEIR OWN, THEY MUST BE MEASURED AGAINST A REFERENCE SHE.
Describe what a SHE looks like.
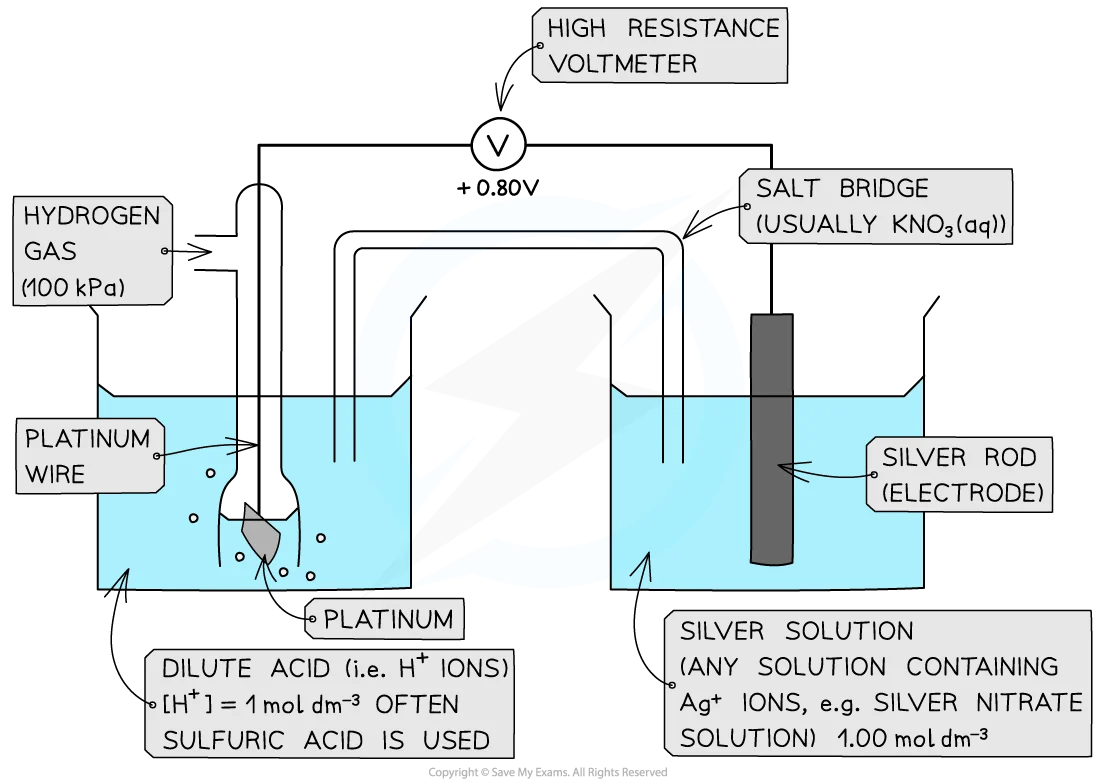
True or false? The concentration of acid in the SHE should always be 1 mol dm-³.
False. The concentration of H+ ions (from the acid) in the SHE must always be 1 mol dm-³, if an acid is diprotic then it only needs to be at a concentration of 0.5 mol dm-³ for the H+ ion concentration to be sufficient.
What are standard conditions for temperature, pressure, and concentration?
Temperature 298 K (25 degrees celsius)
Pressure 100 kPa
Concentration 1 mol dm-³
What is the electrochemical series?
A list of half cell reactions and their standard electrode potentials.
True or false? The species on the left side of the reduction half equation for a half cell in the electrochemical series is the strongest oxidising agent in the half equation.
True.
How does the oxidising power of a species change as the standard electrode potential value gets more positive in the electrochemical series?
It increases.
True or false? The species on the right of the reduction half equation for a half cell is the strongest reducing agent out of all the species in the reduction half equation.
True.
How does reducing power change as the value for standard electrode potential increase in the electrochemical series?
It decreases.
What is the equation for calculating (standard) cell potential?
(under standard conditions) E cell = E reduced - E oxidised
What is a cell notation?
A simplified way of drawing the setup of an electrochemical cell.
Tell me all the rules for cell notation.
1) The half cell with the most negative electrode potential goes on the left of the notation, the half cell with the most positive electrode potential goes on the right of the notation.
2) Reduced form I Oxidised form II Oxidised form I Reduced form, the greater the oxidation state of the species the closer it goes to the double line.
3) Single lines represent state changes.
4) The double line represents the salt bridge.
5) Commas replace single lines when there is no state change between species.
6) Include the platinum electrode in the notation if the solution contains two aqueous ions.
True or false? If the EMF value of an electrochemical cell is negative the reaction taking place is feasible (under standard conditions).
False. IF THE EMF VALUE OF AN ELECTROCHEMICAL CELL IS POSITIVE THE REACTION TAKING PLACE IS FEASIBLE (UNDER STANDARD CONDITIONS).
Outline the steps for determining whether a reaction is feasible/justifying why a reaction is feasible (under standard conditions) when you have got the electrochemical series.
1) Identify which species is being oxidised.
2) Rearrange the equation of the species being oxidised so that it is an oxidation half equation.
3) Combine the two reduction and oxidation half equations to obtain the equation for the feasible reaction.
4) Compare your obtained equation to the reaction given in the question, if they match the reaction is feasible.
(5) Calculate the EMF and if it is a positive value the reaction is feasible)
What are the two types of batteries?
Rechargeable batteries, non-rechargeable batteries.
Give an example of a rechargeable battery.
Phone battery (lithium ion battery), when they run out you supply it with an external source of electricity and it recharges.
Describe the three components of a lithium ion battery.
Lithium cobalt oxide (LiCoO2) electrode, graphite (C) electrode, lithium salt dissolved in an organic solvent electrolyte.
What is the purpose of the electrolyte in a battery?
Electrolytes act as a conductive pathway for ions to move from one electrode to another.
True or false? The negative electrode is the one producing electrons and therefore being oxidised.
True.
What is the half equation for the negative electrode in a lithium ion battery?
Li (s)→← Li+ (aq) + e-
What is the half equation for the positive electrode in a lithium ion battery?
Li+ (aq) + CoO2 (s) →← Li+[CoO2]- (aq)
Give the overall equation for the discharge that takes place in a lithium ion battery.
Li + CoO2 →← Li+[CoO2]
How does supplying rechargeable batteries with a current recharge them?
The current forces electrons to flow in the opposite direction and this reverses the overall discharge equation.
True or false? Electricity is generated by a continuous external supply of chemicals in a fuel cell rather than a ready store like in batteries.
True.
Talk me through all the steps on how an alkaline hydrogen-oxygen fuel cell works.
1) Hydrogen is fed in by the hydrogen feed.
2) Hydrogen reacts with OH- ions in the electrolyte (2H2 (g) + 4OH- (aq) →← 4H2O (l) + 4e-).
3) Electrons produced in the reaction between hydrogen and hydroxide ions travel through a platinum electrode (inert but conductive).
4) This flow of electrons is used to power something.
5) Oxygen is fed in by the oxygen feed.
6) Oxygen reacts with water and the electrons produced from the reaction between hydrogen and hydroxide ions to remake hydroxide ions (O2 (g) +2H2O (l) + 4e- → 4OH- (aq)).
7) Electrons flow to the negative platinum electrode.
8) Potassium hydroxide (KOH) electrolyte carries the hydroxide ions from from the cathode to the anode.
9) Electrons flow from the positive platinum electrode.
10) The water (product) is released into the surroundings.
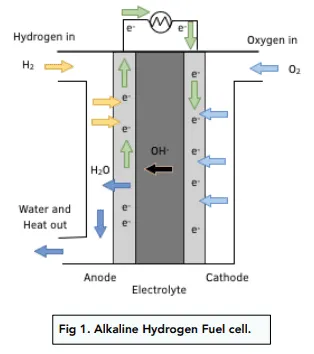
What is the purpose of the ion exchange membranes in an alkaline hydrogen-oxygen fuel cell?
They line the platinum electrodes and allow hydroxide ions to pass through but not hydrogen or oxygen.
Give the equation for the overall reaction in a hydrogen-oxygen fuel cell (from combining the oxidation and reduction half equations).
2H2 (g) + O2 (g) → 2H2O (g)
Give one advantage and one disadvantage of a non-rechargeable battery.
Advantage: they are cheaper in the short term.
Disadvantage: they have a short life.
Give one advantage and one disadvantage of a rechargeable battery.
Advantage: they have a long life.
Disadvantage: an external current has to be applied to them to recharge them.
Give three advantages and three disadvantages of fuel cells.
Advantages:
fuel cells are more energy efficient than combustion engines (more energy is converted into kinetic energy).
fuel cells don’t need to be recharged.
water is the only waste product from fuel cells.
Disadvantages:
hydrogen is highly flammable so must be stored and transported carefully.
it is expensive to store and transport hydrogen.
fossil fuels are used to make hydrogen and oxygen in an electrolysis so carbon dioxide is emitted.
Why can’t an aqueous electrolyte be used in a lithium cell?
Lithium would react with the electrolyte (MORE NEGATIVE THINGS WILL REACT WITH LESS NEGATIVE THINGS).