Organic Test on 23/04/26
0.0(0)
Studied by 0 peopleCard Sorting
1/6
There's no tags or description
Looks like no tags are added yet.
Last updated 7:35 AM on 4/23/26
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
7 Terms
1
New cards
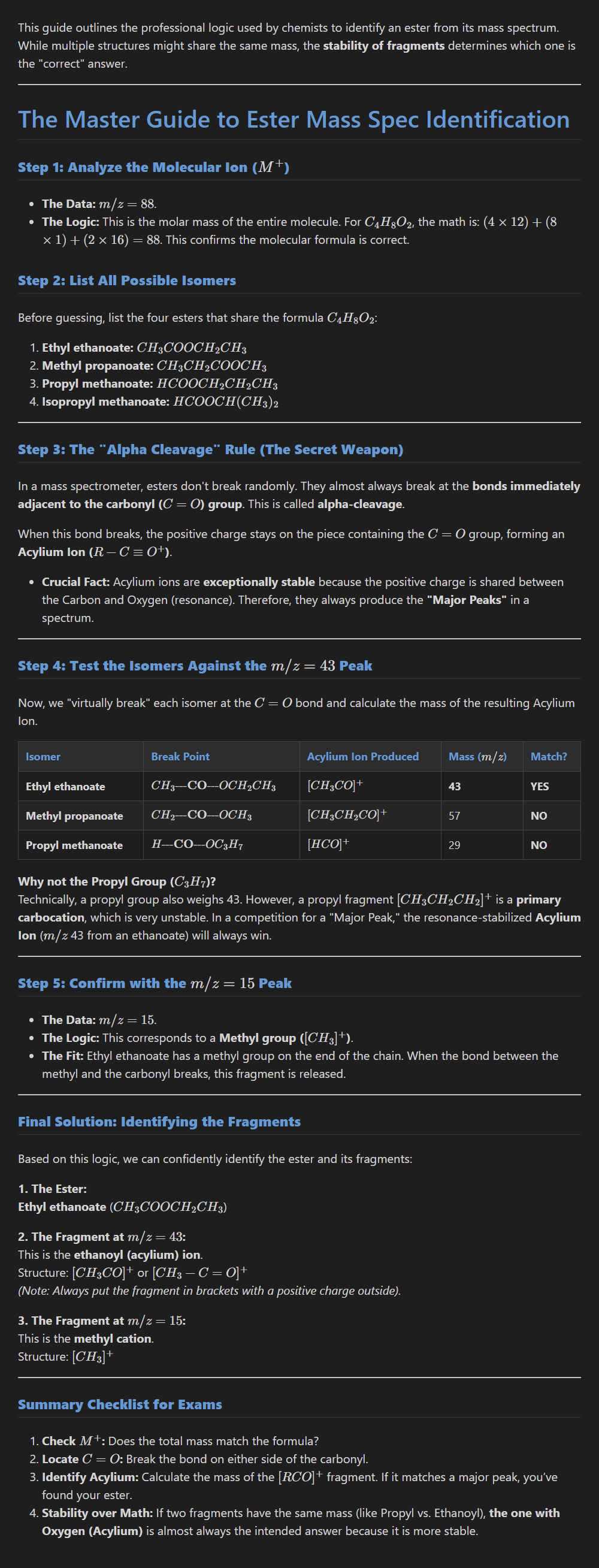
Carbocation stability? Why?
Higher-order carbocations are more stable because they have more electron-donating alkyl groups. These groups have a positive inductive effect that spreads out (stabilises) the positive charge.
2
New cards
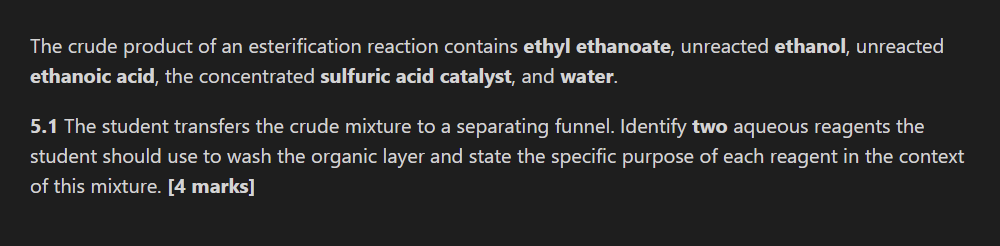
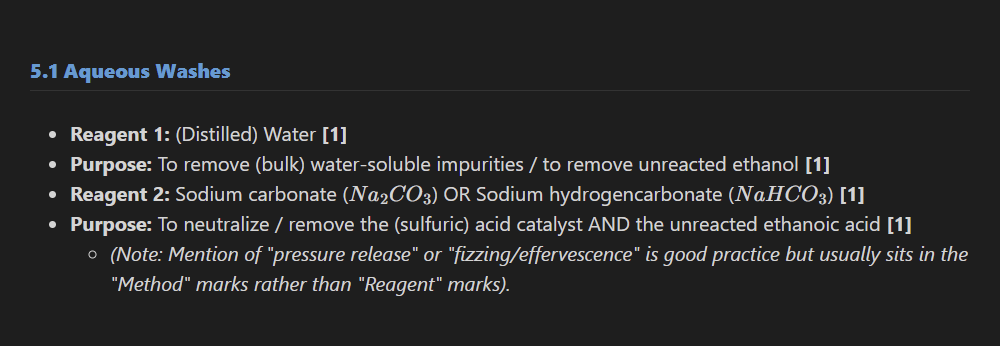

3
New cards
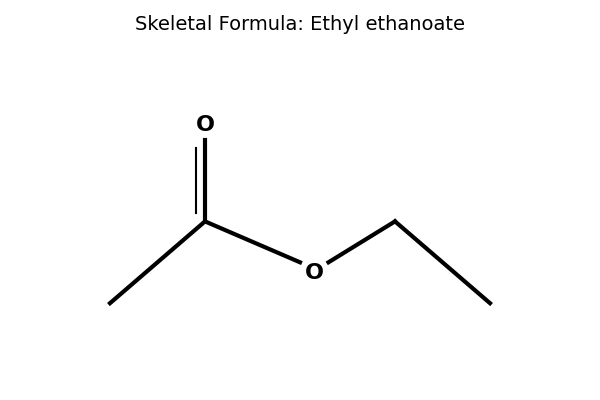
Ester formed from -COOH and -OH, where does the full -OH drop out from?
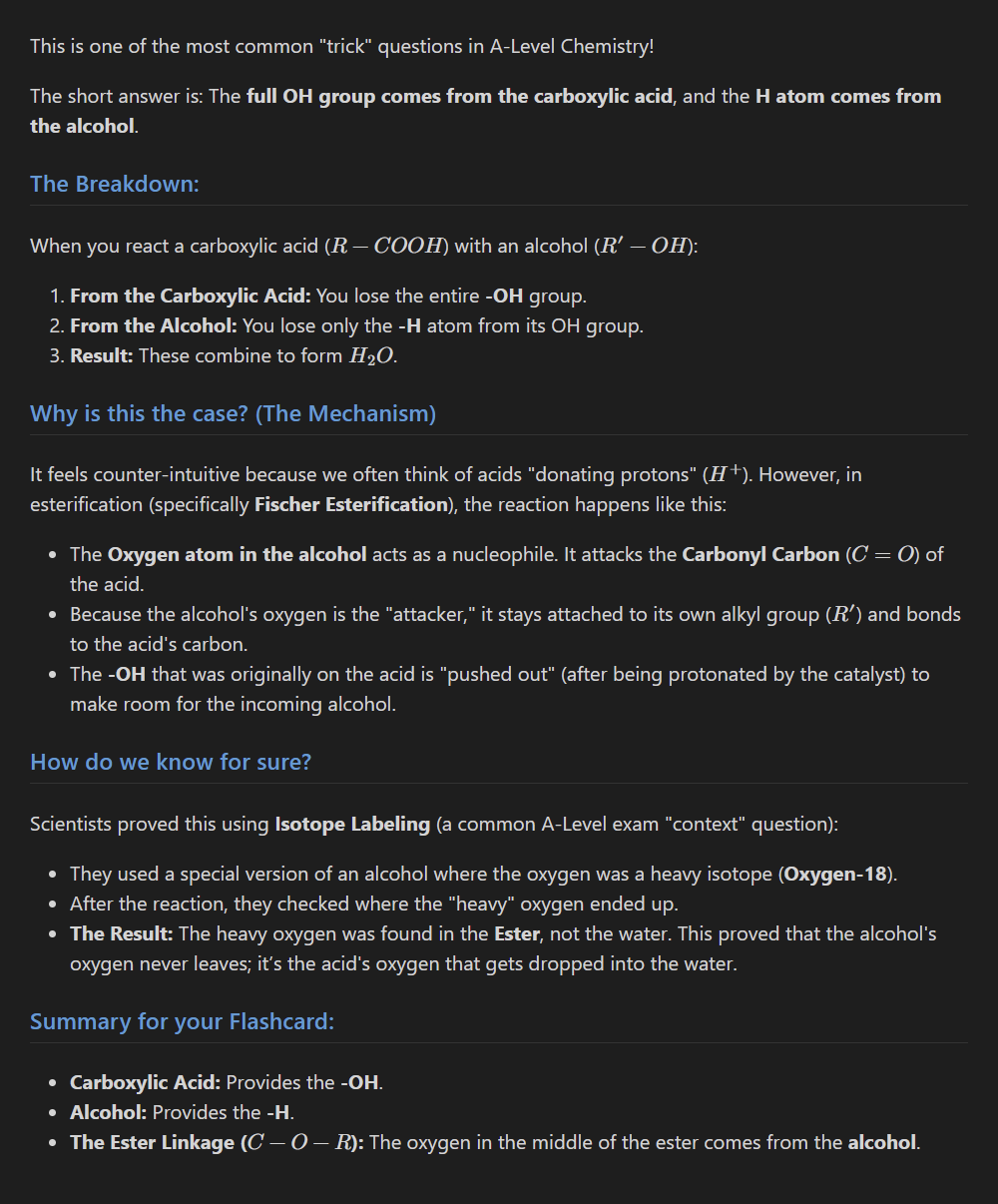

4
New cards
Ethane reacts with chlorine under UV light. One of the termination steps produces butane.
(a) Write the equation for the termination step that produces butane. [1]


5
New cards



6
New cards
Define electronegativity.
What factors influence it?
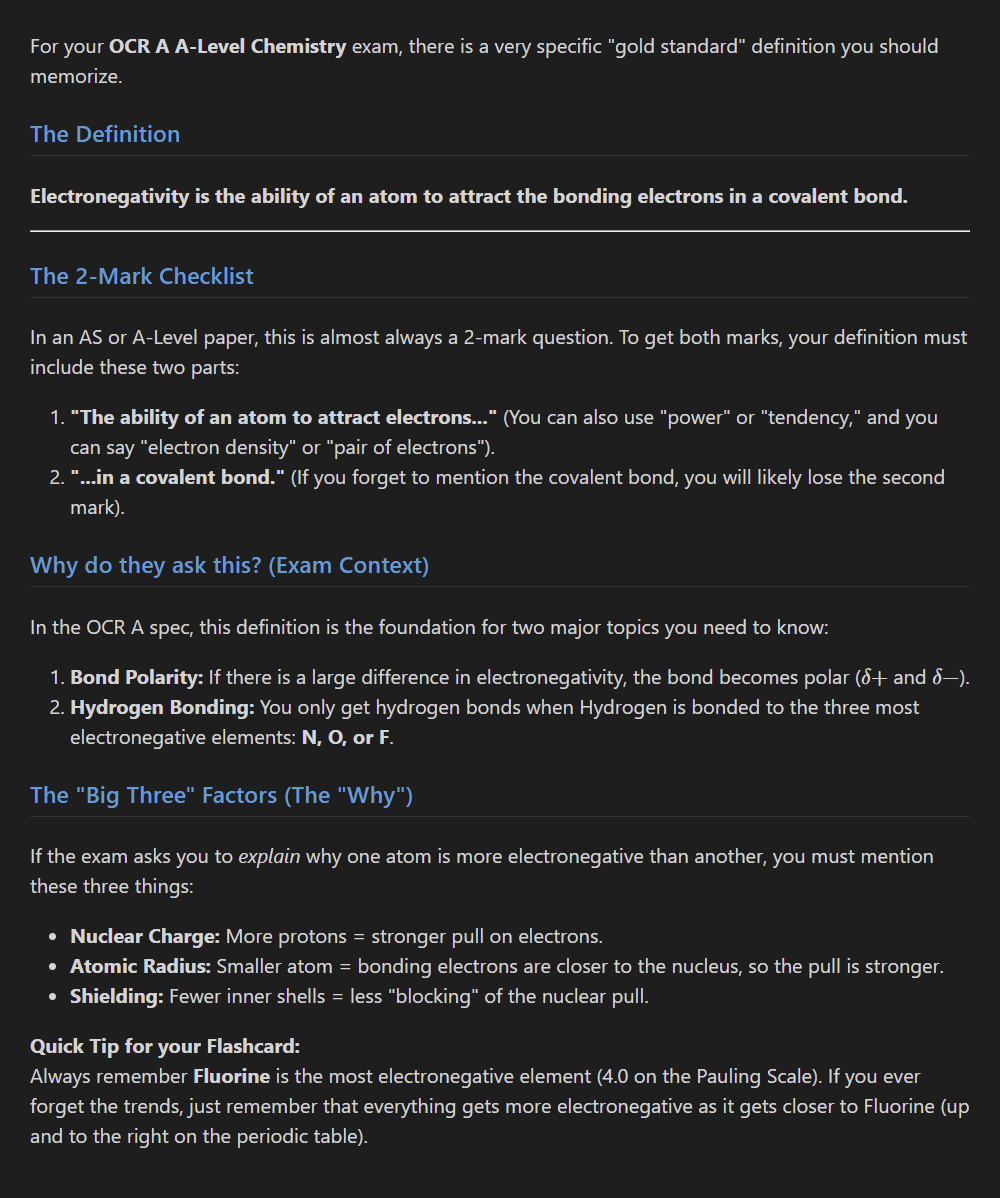

7
New cards