Ochem All Reactions
1/34
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
35 Terms
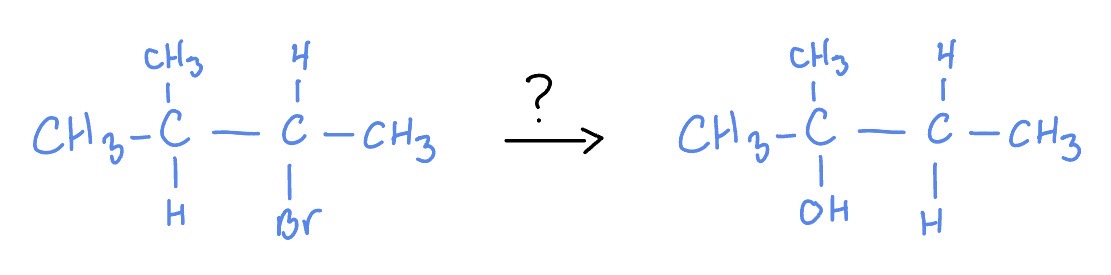
Hydride Shift
H2O
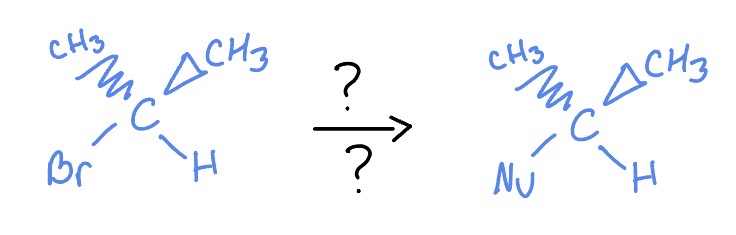
Sn2
*flips s/r
Nu:Na
acetone
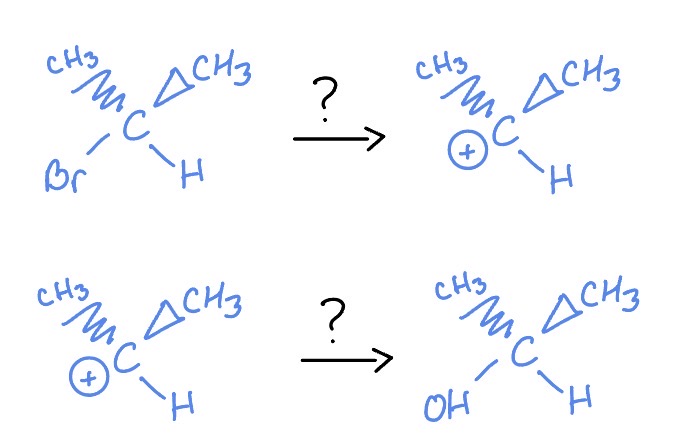
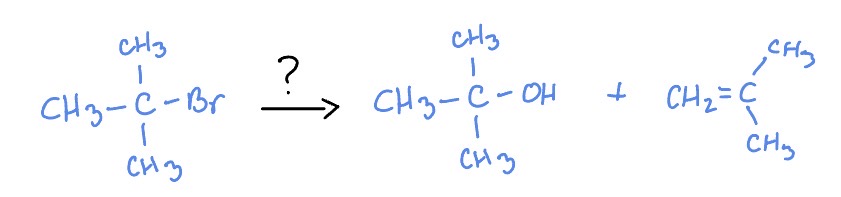
Sn1
H2O
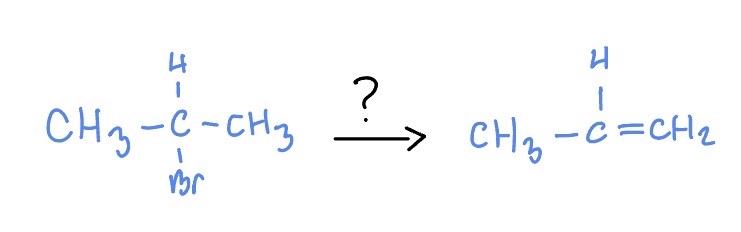
E2
t-BuOK
E2
H2O
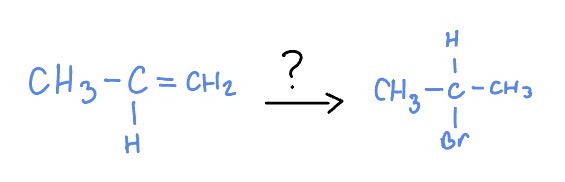
Methyl Shift
*Markovnikov
HBr
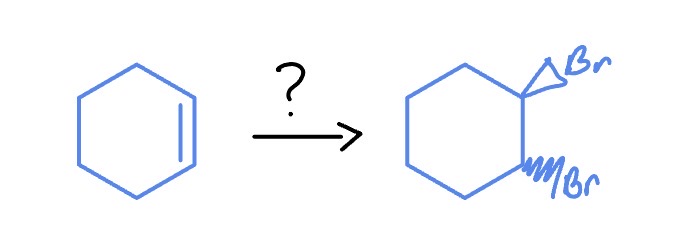
Electrophile Reaction
*Anti-Addition
Br2
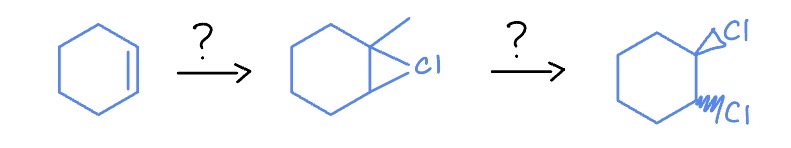
Electrophile Reaction
*Anti-Addition
Cl2
H2O
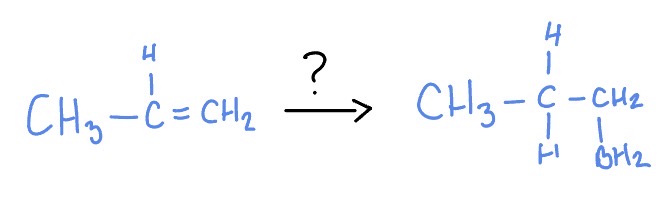
Hydroboration
*Anti-Markovnikov
*Syn-Addition
BH3
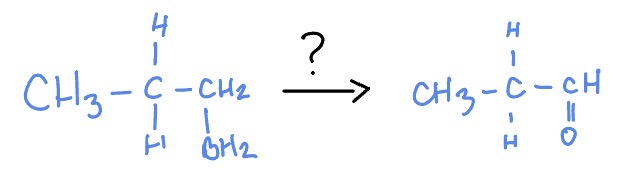
Hydroboration Oxidation
H2O2
Dehydration
*Syn-Addition
OsO4
H2O
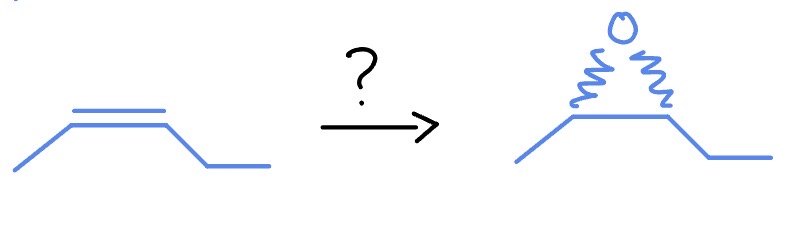
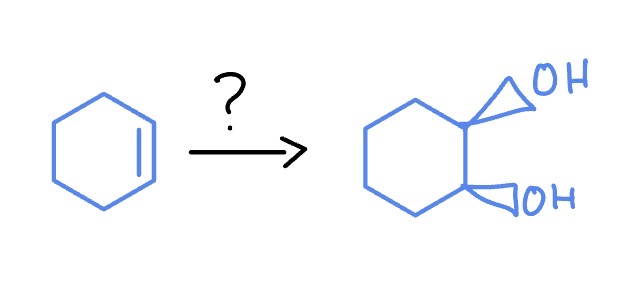
Epoxidation
*Syn Addition
RCO3H
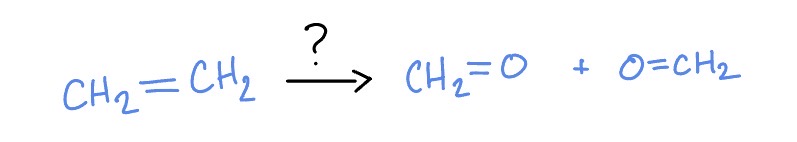
Ozonation
O3
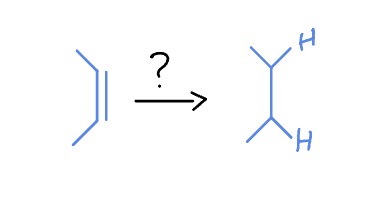
Reaction of Alkenes
*Syn Addition
H2
Pd
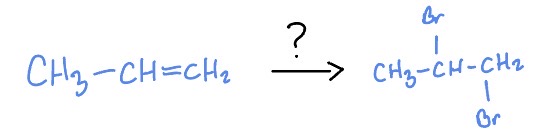
Radical + Alkenes
Br2
Dark
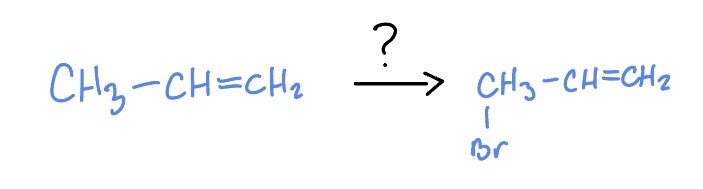
Radical Halogenation of Alkenes
Br2
NBS
Light
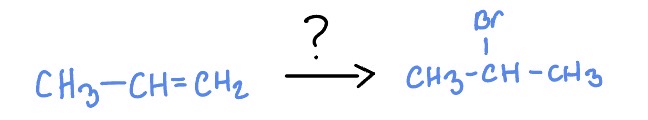
Radical Addition of Alkenes
*Markovnikov
HBr
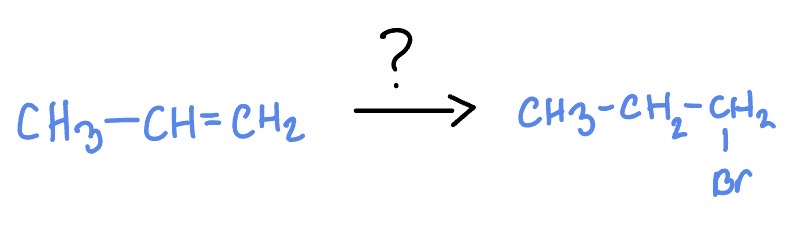
Radical Addition of Alkenes
*Non-Markovnikov
HBr
Peroxides
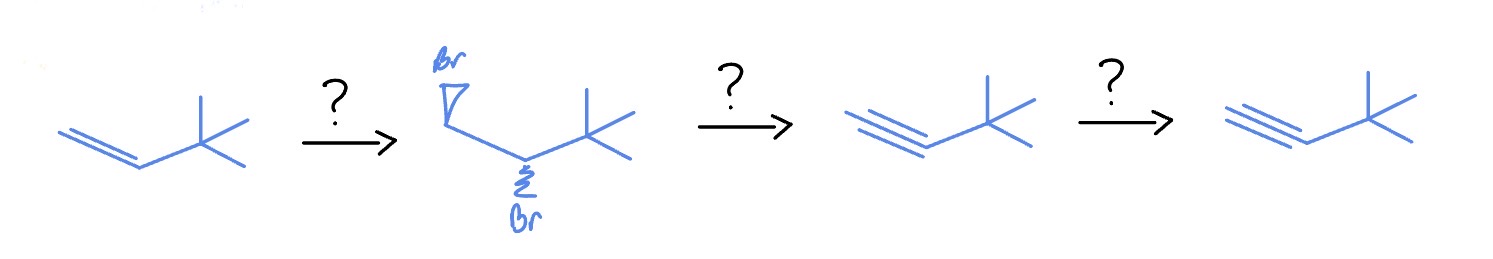
Alkynes + Strong Base
*Syn
Br2
NaH2 (Strong base)
H2O
Alkyne + Lindt Catalyst
*Syn Addition
H2
Lindt Catalyst/Poison Catalyst

Alkyne + 1mol Halogen
*Anti Addition
Br2 (1mol)
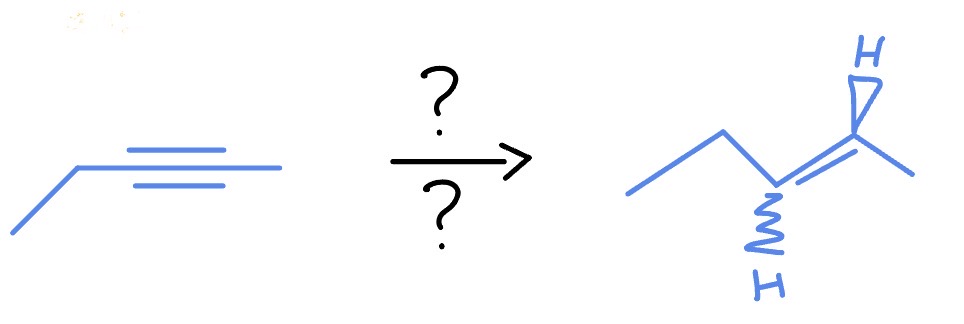
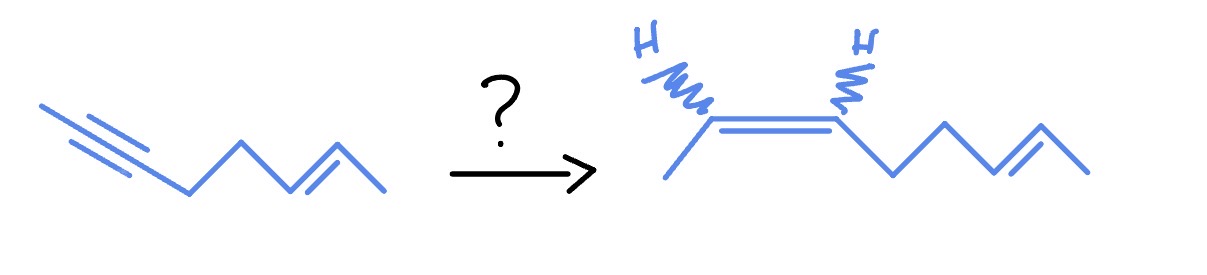
Alkyne with NH3 Liquid
*Anti Addition
Na
NH3 (Liquid)
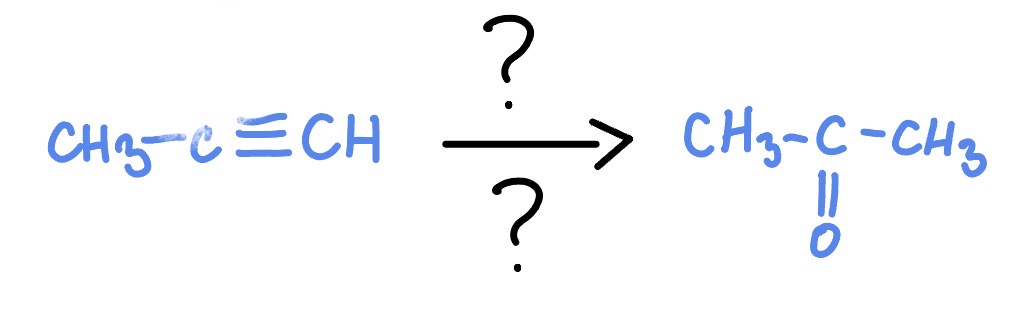
Alkyne w/ H2SO4 or HgSO4
*Markovnikov
H2SO4 + H2O
HgSO4
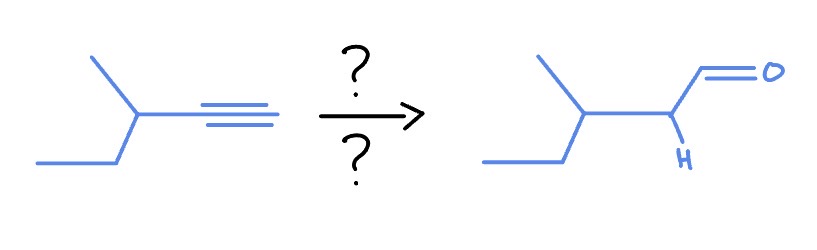
Alkyne
*Non-Markovnikov
BH3, THF
H2O
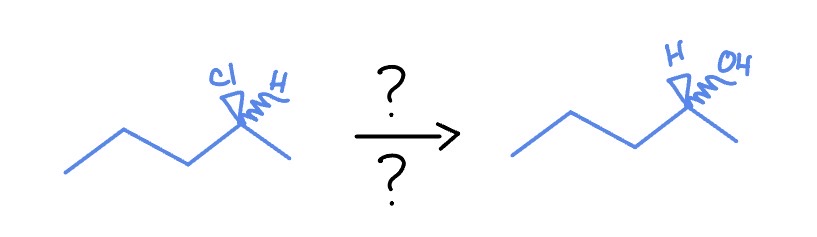
Sn2
*Flips S/R (where H is)
NaOH
DMSO
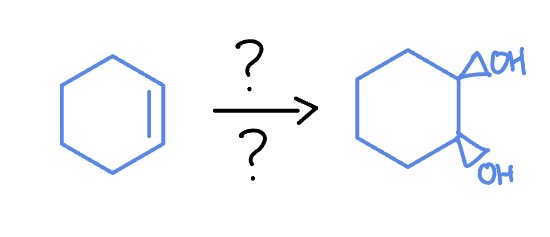
*Syn Addition
OsO4
NaHSO3, H2O
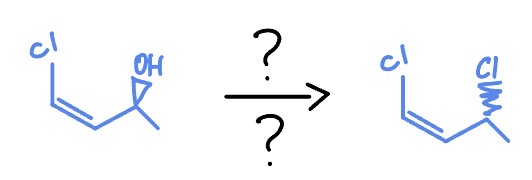
2* Alcohol Sn2
*Flips S/R (where H is)
SoCl2, PBr2
Pyridine
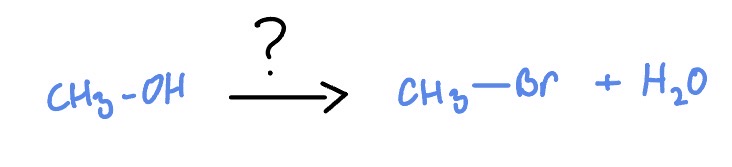
1* Alcohol
HBr or HCl

Formation of Dbl. Bond
H2SO4
Heat
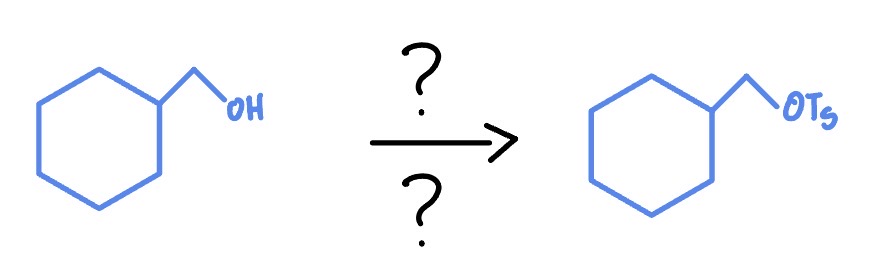
TsCl
Pyridine
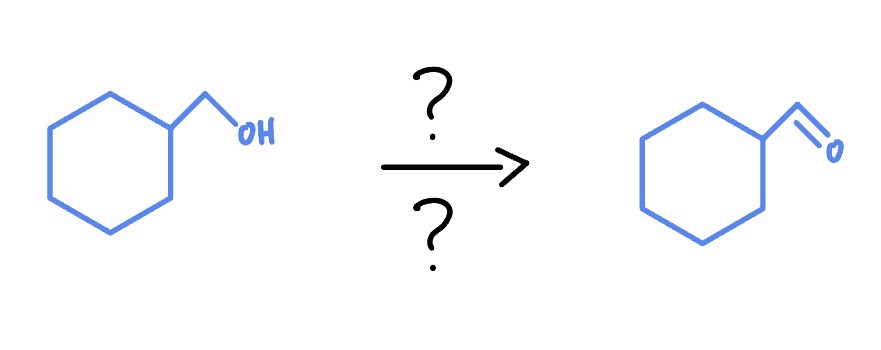
Makes aldehyde
Primary Alcohol
PCC
CH2Cl2

H2CrO4
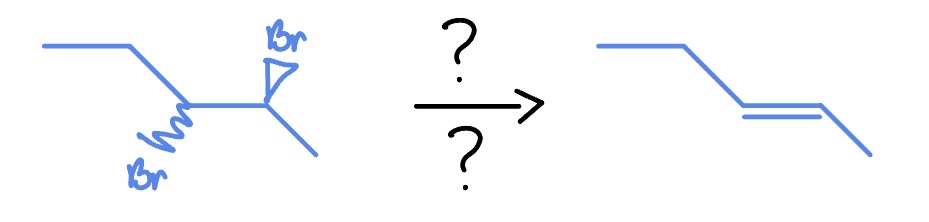
Na
NH3
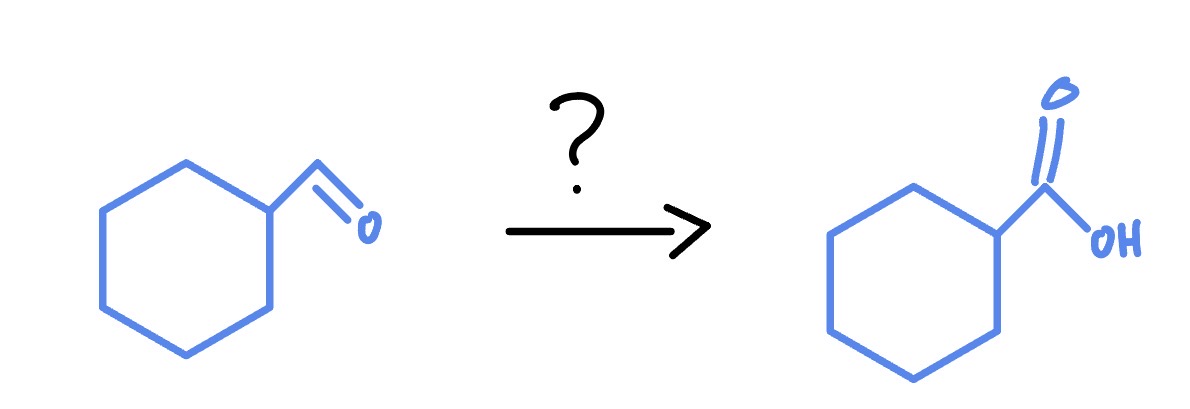
Makes Carboxylic Acid
Primary only
IBX
DMSO
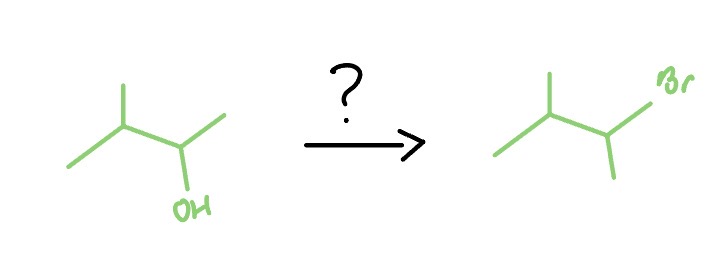
PBr3/SoCl2