Basic Atom + Periodic Table + Nomenclature
1/43
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
44 Terms
Atomic Notation
A (mass #)
X
Z (atomic #)
Period Table Notation
Z (atomic #)
X
avg A (average mass #)
neutrons # =
mass # - atomic #
Ions - Cation
positive charge - lost electrons
Ions - Anion
negative charge - gained electrons
cation size
smaller than atom
anion size
larger than atom
covalent bond
shared electrons
[ non-metal + non-metal ]
![<p>shared electrons</p><p>[ non-metal + non-metal ]</p>](https://assets.knowt.com/user-attachments/ae3e75e6-d6c8-470c-85e7-8cb1c2a4c59a.jpg)
ionic bond
transferred electrons
[ metal + non-metal ]
![<p>transferred electrons</p><p>[ metal + non-metal ]</p>](https://assets.knowt.com/user-attachments/324c6c49-9fef-483f-8955-3bfbcdb1a879.jpg)
Periodic Table - Groups
vertical column |
Periodic Table - Periods
horizontal row —
Periodic Table - Group 1 |
alkali metals ➡ +1 ion
Periodic Table - Group 2 |
alkaline earth metals ➡ +2 ions
Periodic Table - Group 17 |
halogens ➡ -1 ions
Periodic Table - Group 18 |
noble gases ➡ very stable
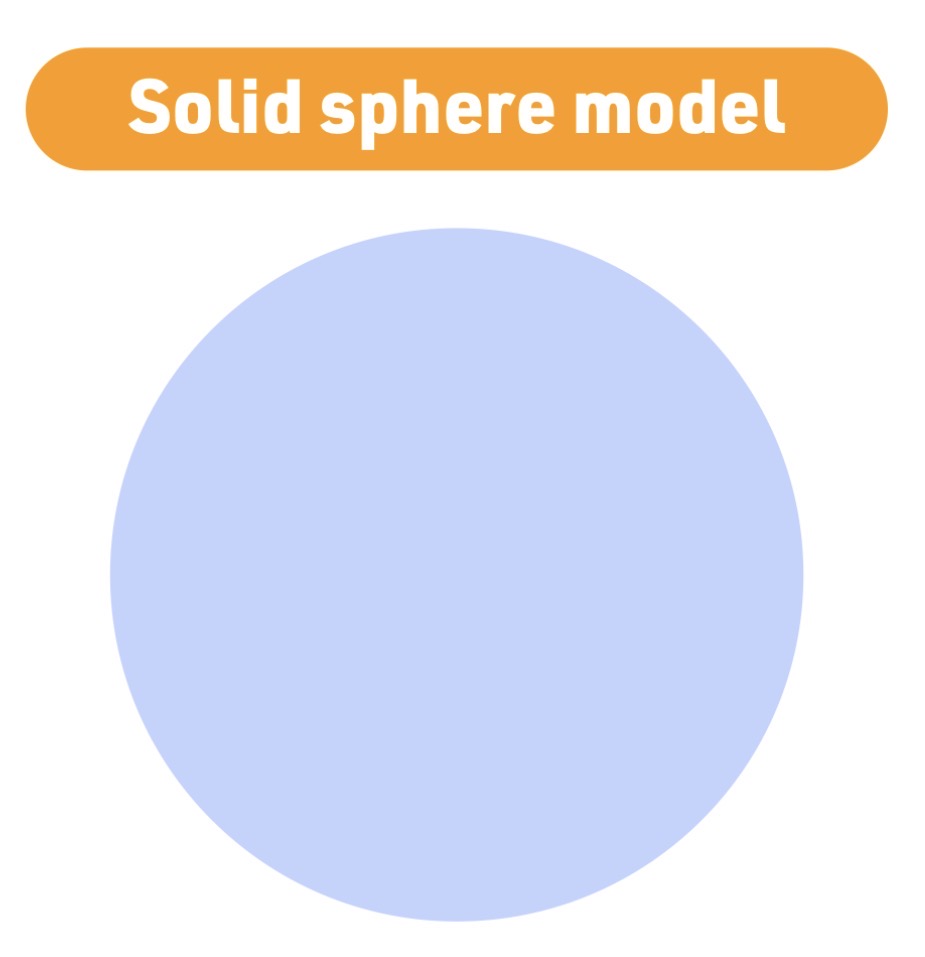
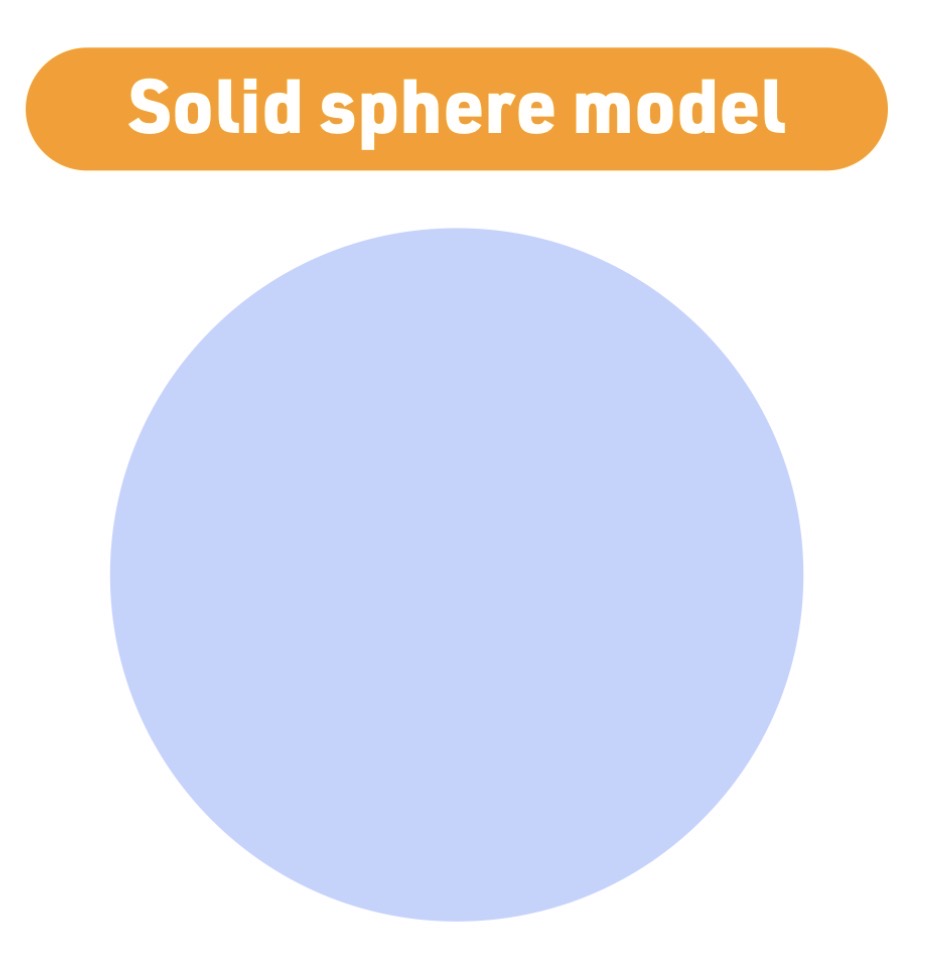
Atomic model - 1803
John Dalton
Solid Sphere model
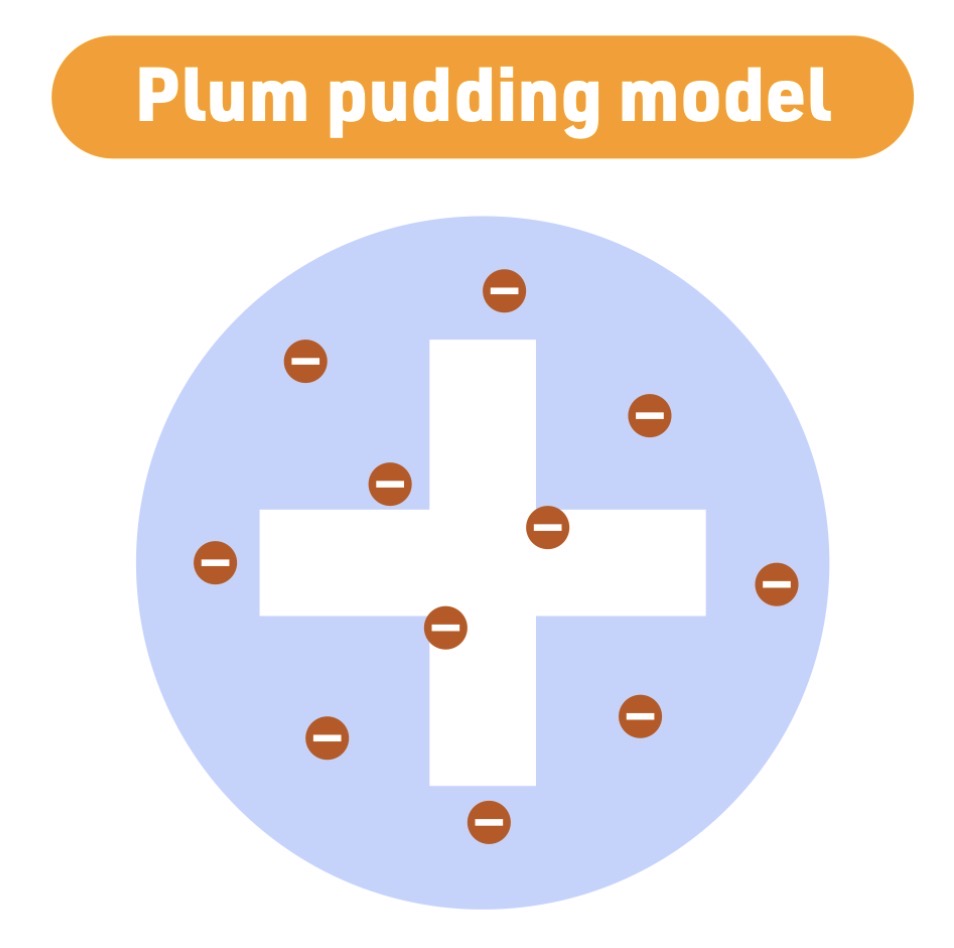
Atomic model - 1904
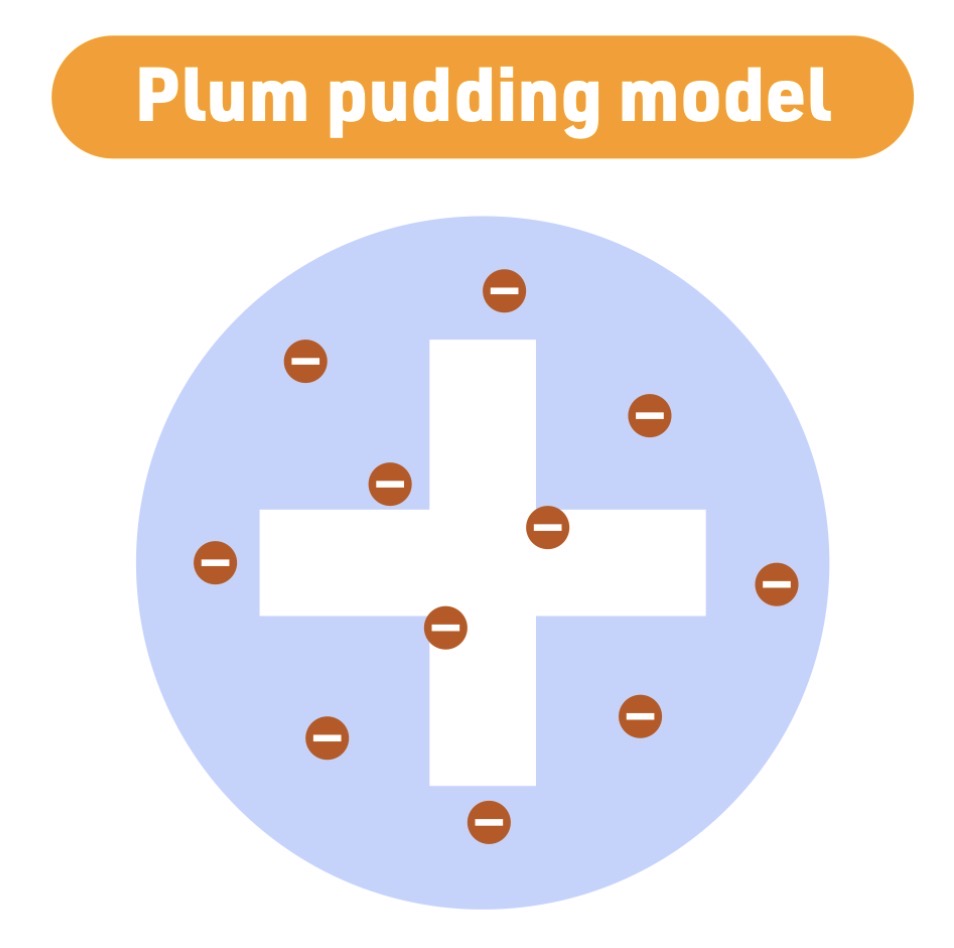
J.J. Thomson
Plum Pudding model
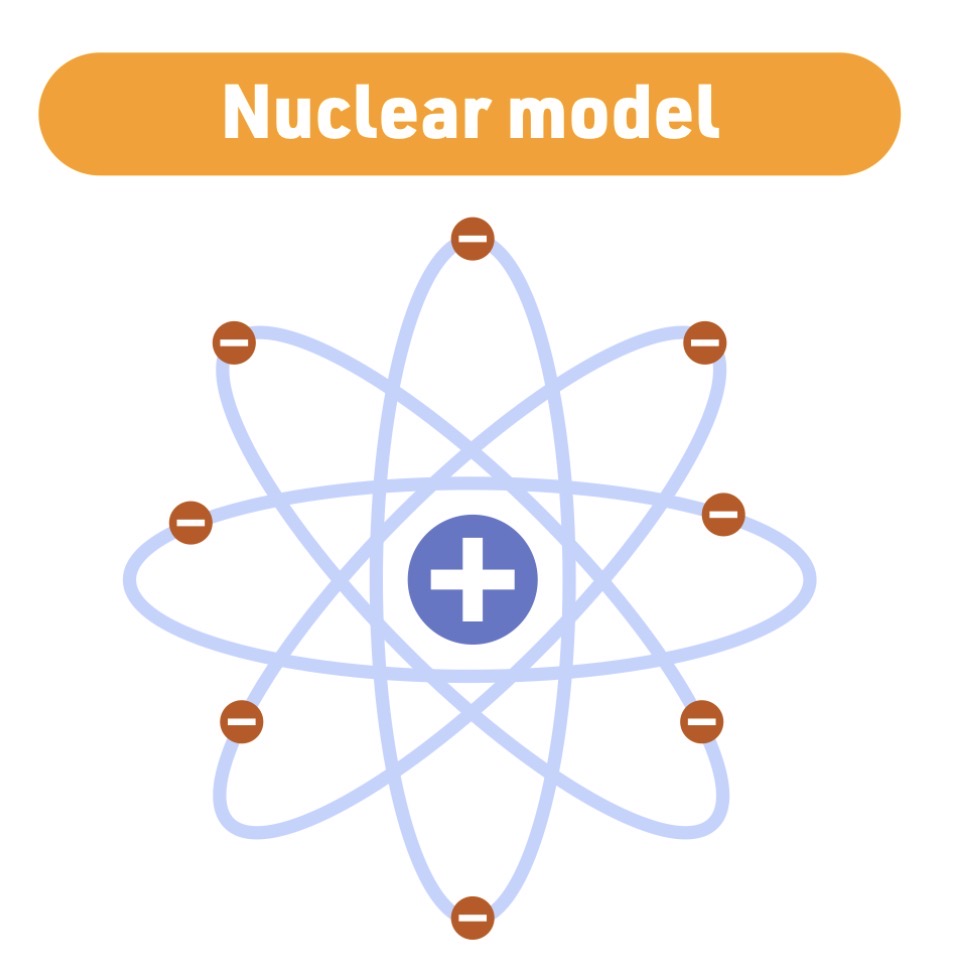
Atomic model - 1911
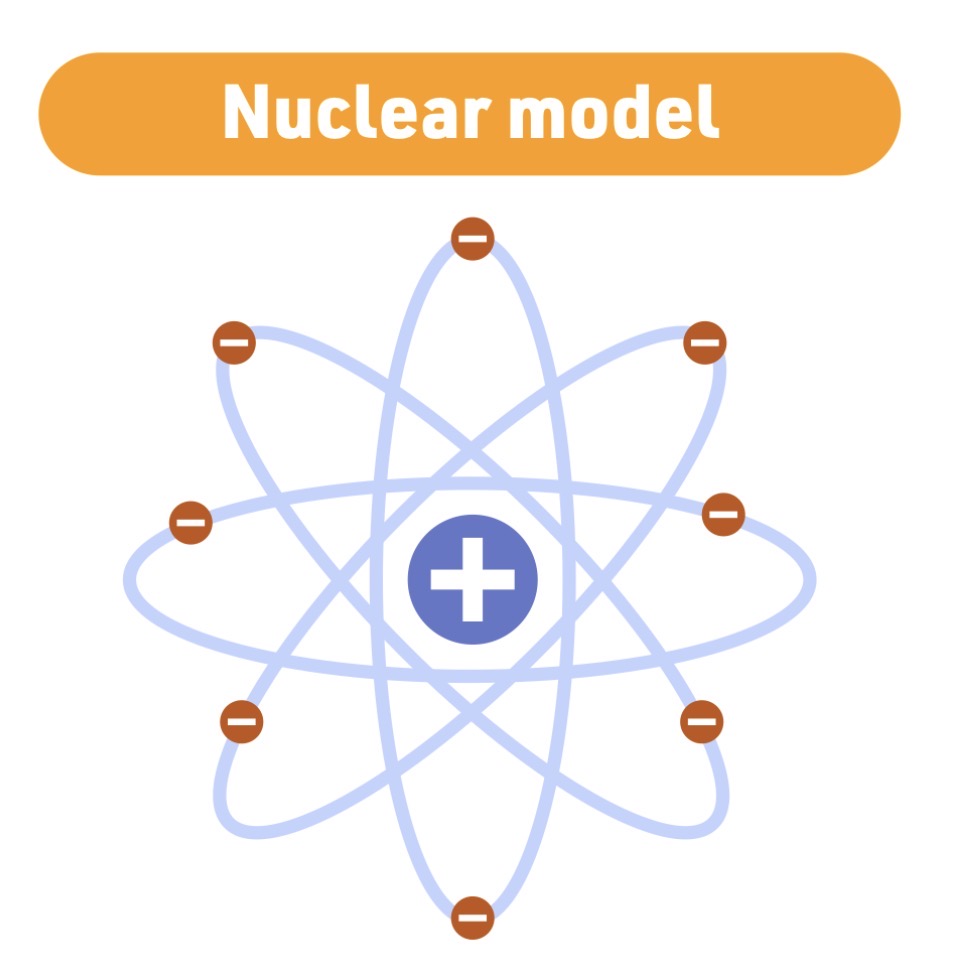
Ernest Rutherford
Nuclear model
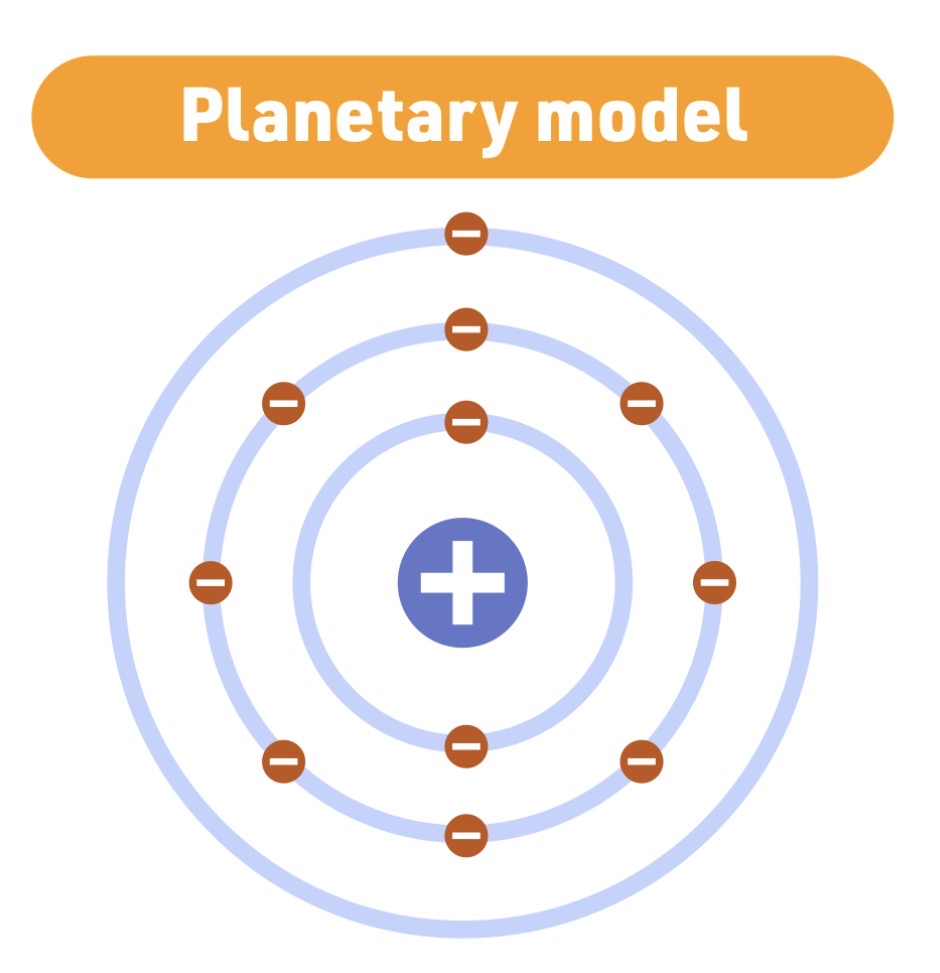
Atomic Model - 1913
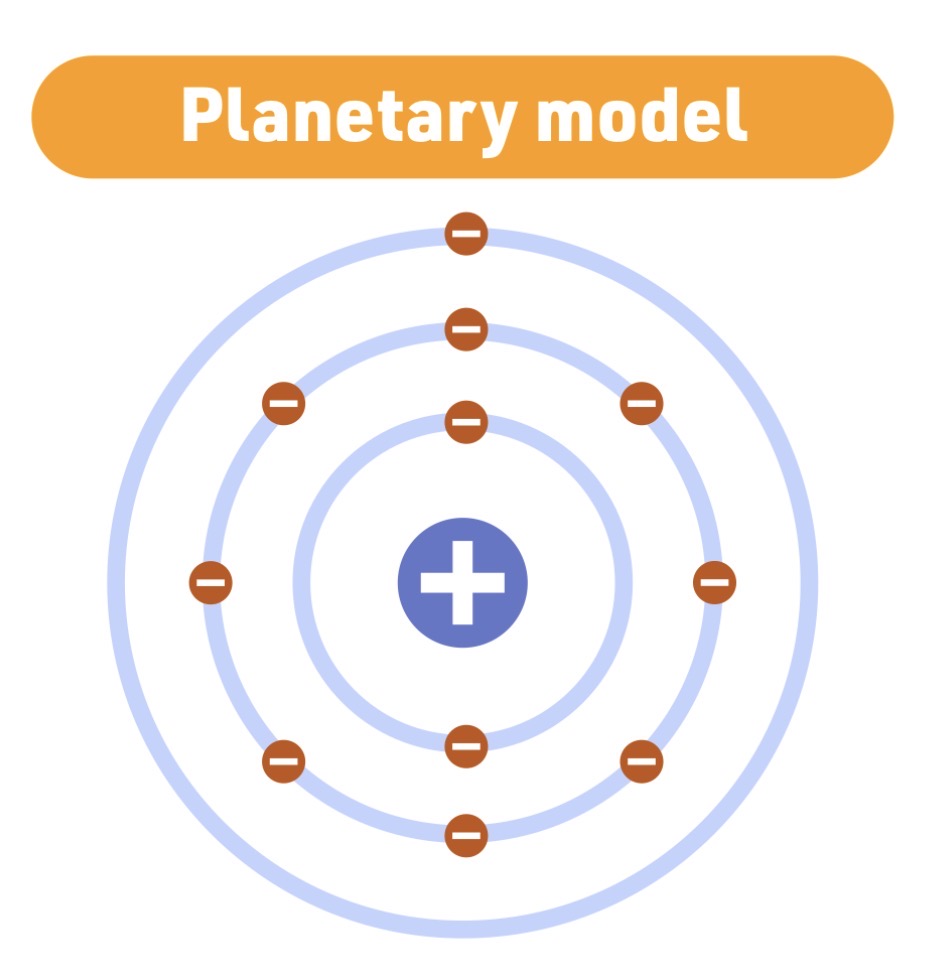
Niels Bohr
Planetary model
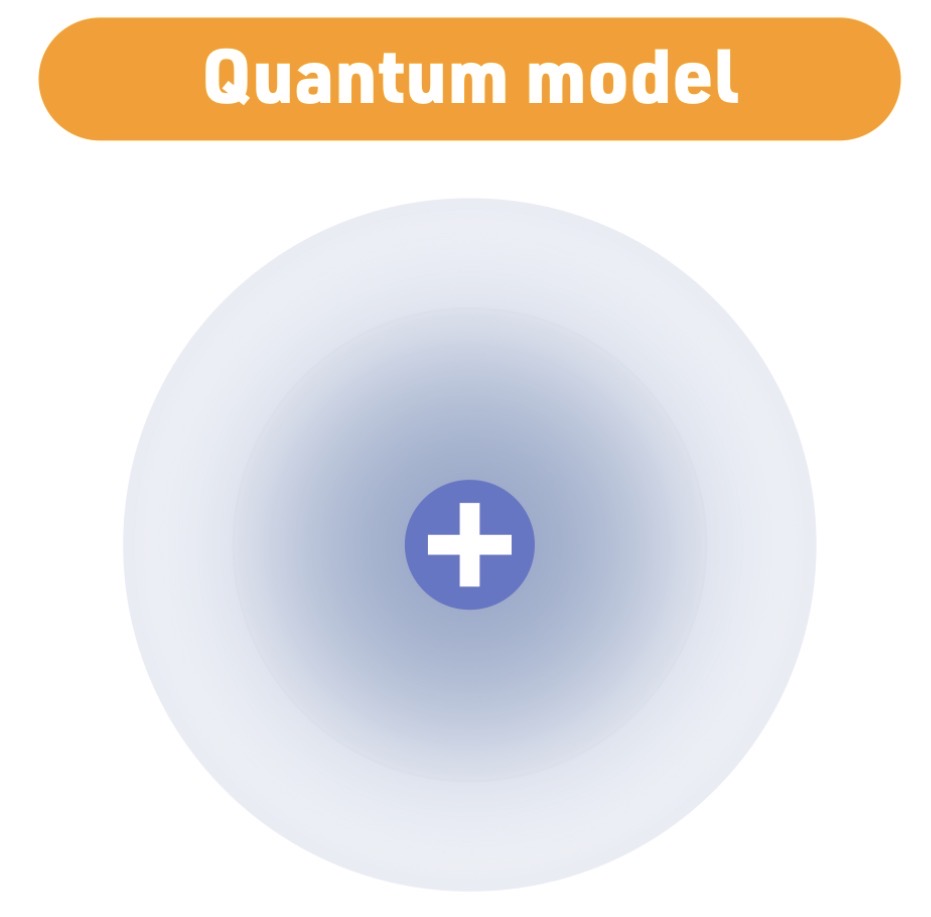
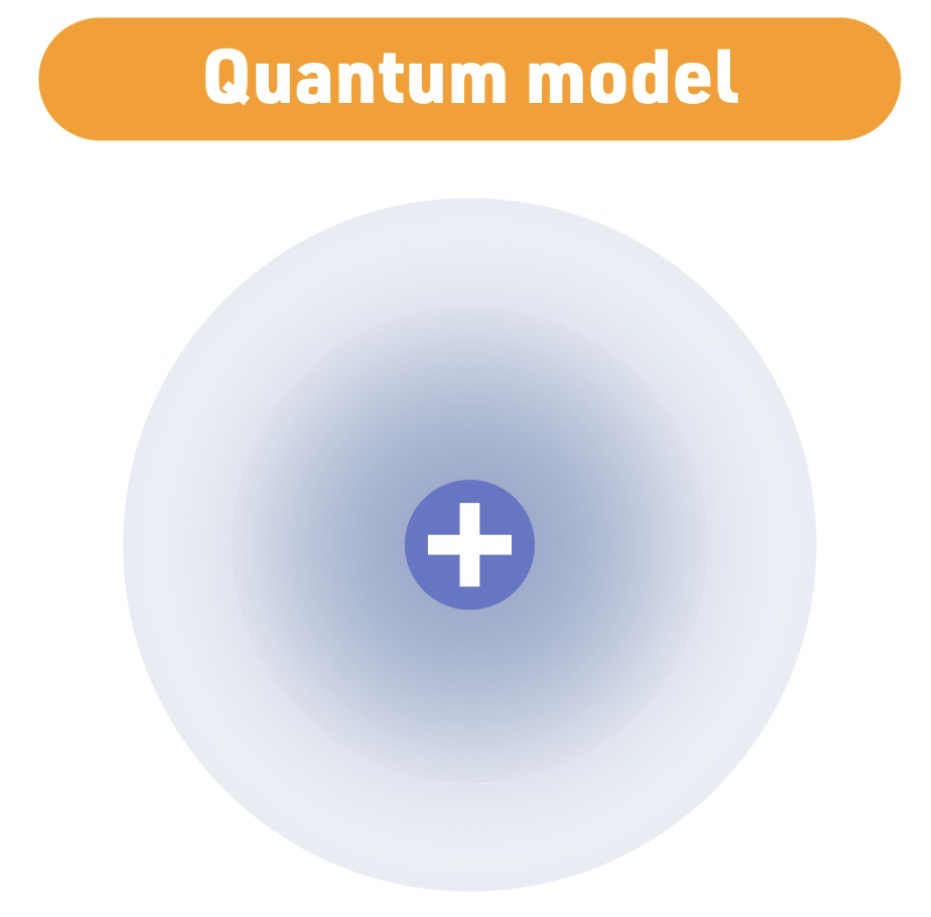
Atomic Model - 1926
Schrödinger
Quantum model
Dalton atomic model Characteristic
atoms are solid - indivisible - spheres identical for each element
Thomson model Characteristic
atom is a positive - sphere - with electrons embedded inside
Rutherford model Characteristic
atom is mostly empty space - with a small - dense - positive - nucleus
Bohr model Characteristic
electrons move in fixed - energy levels - around - the nucleus
Schrödinger model Characteristic
electrons exist in probability - clouds - not fixed - paths
Thomson Experiment Discovery
discovered electrons as negative - particles - in atoms
Rutherford Experiment Discovery
discovered a small - dense - positive nucleus
&
atoms are mostly empty space
Thomson Experiment
electricity passed through a vacuum tube
&
the beam was deflected by magnets -
showing it had particles with - negative - charges
Rutherford Experiment
alpha + particles were fired at - gold foil
&
most passed through while - some were - deflected or - bounced back
Identify Compound Type :
H…
Acid
Identify Compound Type :
metal + non-metal
Ionic
Identify Compound Type:
non-metal + non-metal
Covalent
Identify Compound Type:
… · nH₂O
Hydrate
Overall compound charge
charge = 0
Ionic compound name of
metal (fixed charge) + non metal
cation + anion [root + ide]
Ionic compound name of
metal (multiple charges) + non metal
{cation} metal (Roman #) - anion [root + ide]
Covalent compound name
prefix & 1st element + prefix & 2nd element
(NO “mono” for 1st element)
Prefixes
1 - mono
2 - di
3 - tri
4 - tetra
5 - penta
6 - hexa
7 - hepta
8 - octa
9 - nona
10 - deca
Acid naming for
No oxygen
hydro & element/compound [root + ic] + acid
Acid naming for
Oxyacids - with oxygen
oxyanion [ -ite ➞ ous] acid
oxyanion [ -ate ➞ ic] acid
Polyatomic ion compound name for:
cation + polyatomic ion {anion}
polyatomic cation + anion
polyatomic cation + polyatomic ion {anion}
cation + anion ( {if not polyatomic ion} root + ide)
Hydrate name of
compound · nH₂O
compound + prefix & hydrate
2 oxyanions name pattern
less O ➞ -ite
more O ➞ -ate
4 oxyanions name pattern
least O ➞ hypo … ite
less O ➞ -ite
more O ➞ -ate
most O ➞ per … ate